Abstract
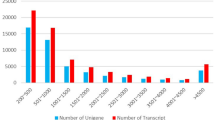
Cobia is an important cultured marine fish species in southern China. It is characterised by fast growth. Due to the effects of climate change on water oxygen levels and seawater temperatures, understanding the influence of environmental challenges on cobia culture has become very important. In this study, to explore the stress and adaptability of cobia (Rachycentron canadum) during hypoxia-reoxygenation conditions, the fish were exposed to an oxygen-deficient environment with dissolved oxygen (DO) level of 2.64 ± 0.25 mg/L. Liver tissue transcriptome sequencing was detected in the fish at acute hypoxia stress, after which fish were returned to normal DO levels (6.34 mg/L) for 8, 24, and 48 h. Comparative analysis of liver transcriptomes revealed that there were 1689, 651, 236, and 1150 differential genes in the hypoxia stress group (SC), reoxygenation-8h group (R8), reoxygenation-24h group (R24), and reoxygenation-48h group (R48), respectively. The differentially expressed genes were compared with the GO database. The main aggregated genes were related to gene ontology functional elements such as ribosome structural components, matrix-dependent cell migration, hormone activity, and oxidoreductase activity. The differentially expressed genes were compared with the Kyoto Encyclopedia of Genes and Genomes (KEGG) database, and a total of 43,054 differentially expressed genes were found to be enriched in 212 cases. For the first time, gene expression patterns in the liver of a juvenile’s cobia were examined in response to hypoxia. The results of this study contribute to further clarifying hypoxia’s effects on the liver of marine fish.










Similar content being viewed by others
References
Abdel-Tawwab M, Monier MN, Hoseinifar SH, Faggio C (2019) Fish response to hypoxia stress: growth, physiological, and immunological biomarkers. Fish Physiol Biochem 45(3):997–1013
Amenyogbe E, Huang J-S, Chen G, Wang W-Z (2021) Probiotic potential of indigenous (Bacillus sp. RCS1, Pantoea agglomerans RCS2, and Bacillus cereus strain RCS3) isolated from cobia fish (Rachycentron canadum) and their antagonistic effects on the growth of pathogenic Vibrio alginolyticus, Vibrio harveyi, Streptococcus iniae, and Streptococcus agalactiae. Front Mar Sci 8:672213. https://doi.org/10.3389/fmars.2021.672213
Amenyogbe E, Yang E-J, Xie R-T, Huang J-S, Chen G (2022) Influences of indigenous isolates Pantoea agglomerans RCS2 on growth, proximate analysis, haematological parameters, digestive enzyme activities, serum biochemical parameters, antioxidants activities, intestinal morphology, disease resistance, and molecular immune response in juvenile’s cobia fish (Rachycentron canadum). Aquaculture 737942:0044–8486. https://doi.org/10.1016/j.aquaculture.2022.737942
Cai X-H, Huang Y-T, Zhang Z-P, Wang Y-L (2014) Hypoxia inducible factor-1 (HIF-1) and its research advance in aquatic animals. J Agri Biotechnol 22(1):119–132
Cameron JS, Dewitt JP, Ngo TT, Yajnik T, Chan S, Chung E, Kang E (2013) Cardiac KATP channel alterations associated with acclimation to hypoxia in goldfish (Carassius auratus L.). Comp Biochem Physiol A Mol Integr Physiol. 164(4):554–64. https://doi.org/10.1016/j.cbpa.2012.12.020
Casey TM (2002) Hypoxia causes downregulation of protein and RNA synthesis in noncontracting mammalian cardiomyocytes. Circ Res 90(7):777–783. https://doi.org/10.1161/01.res.0000015592.959
Chee NT, Lohse I, Brothers SP (2019) mRNA-to-protein translation in hypoxia. Mol Cancer 18:49. https://doi.org/10.1186/s12943-019-0968-4
Chen B.-X (2016) Identification and application of hypoxia-associated molecular markers in Megalobrama amblycephala[D]; Huazhong Agricultural University
Chi S, Tan B, Dong X, Yang Q, Liu H (2014) Effects of supplemental coated or crystallinemethionine in low-fishmeal diet on the growth performance and body composition of juvenile cobia Rachycentron canadum (Linnaeus). Chin J Oceanol Limnol 32:1297–1306
Chi S, Tan B, Dong X, Yang Q, Liu H, Xu Y, Huang H (2011) Effect of supplementation microcapsule or crystalline methionine in diets on related enzyme activity of cobia (Rachycentron canadum). J. Fish. Sci. China (Zhongguo Shuichan Kexue) 18
Cnaani A, McLean E (2009) Time-course response of cobia (Rachycentron canadum) to acute stress. Aquaculture 289(1–2):140–142. https://doi.org/10.1016/j.aquaculture.2008.12.016
Crear DP, Watkins BE, Friedrichs MAM, St-Laurent P, Weng KC (2020) Estimating shifts in phenology and habitat use of cobia in Chesapeake Bay under climate change. Front Mar Sci 7:579135. https://doi.org/10.3389/fmars.2020.579135
Detmar M, Brown LF, Berse B, Jackman RW, Elicker BM, Dvorak HF, Claffey KP (1997) Hypoxia regulates the expression of vascular permeability factor/vascular endothelial growth factor (VPF/VEGF) and its receptors in human skin. J Invest Dermatol 108(3):263–8. https://doi.org/10.1111/1523-1747.ep12286453
Di Z-C, Zhou T, Xu Q-H (2018) Expression comparisons of heat shock proteins gene in gills of zebrafish exposed to both normoxia and hypoxia. J Dalian Ocean Univ 33(6):11–16
Diaz RJ (2001) Overview of hypoxia around the world. J Environ Quality 30(2):275
Gong D, Xu L, Li W, Shang R, Chen J, Hu F, Wang S, Liua Q, Wu C, Zhou R, Zhang C, Tao M, Wang Y, Liu S (2020) Comparative analysis of liver transcriptomes associated with hypoxia tolerance in the gynogenetic blunt snout bream. Aquaculture, 735163https://doi.org/10.1016/j.aquaculture.2020.735163
Guo H-Y, Cheng G-H, Li Y-J, Zhang H, Qin K-L (2016) The high throughput sequencing technology and its application in biology. Contemporary Animal Husbandry 12:61–65
Guo X (2005). Hemoglobin mechanism of hypoxia toleration in Tibetan chick embryo [D]; China Agricultural University
Heath AG (1988) Anaerobic and aerobic energy metabolism in brain and liver tissue from rainbow trout (Salmo gairdneri) and bullhead catfish (Ictalurus nebulosus). J Exp Zool 248(2):140–146
Huang C-Y, Lin H-C, Lin C-H (2015) Effects of hypoxia on ionic regulation, glycogen utilisation and antioxidative ability in the gills and liver of the aquatic air-breathing fish Trichogastermicrolepis. Comparative Biochem Physiol Part A: Mole Integr Physiol 179:25–34
Huang J-S, Guo Z-X, Zhang J-D, Wang W-Z, Wang Z-L, Amenyogbe E, Chen G (2021) Effects of hypoxia-reoxygenation conditions on serum chemistry indicators and gill and liver tissues of cobia (Rachycentroncanadum). Aquaculture Rep 20:2352–5134. https://doi.org/10.1016/j.aqrep.2021.100692
Huang JS, Zhi L, Chen G, Zhang J, Guo Z, Hong Y (2019) Acute hypoxia stress on blood biochemical indexes of large-sized juvenile cobia (Rachycentron canadum). Haiyang Xuebao 41(6):76–84
Kang J, Ma X, He S (2017) Evidence of high-altitude adaptation in the glyptosternoid fish, Creteuchiloglanis macropterus from the Nujiang River obtained through transcriptome analysis. BMC Evol Biol 17:229. https://doi.org/10.1186/s12862-017-1074-0
Lai KP, Li J-W, Tse AC-K, Chan T-F, Wu RS-S (2016) Hypoxia alters steroidogenesis in female marine medaka through miRNAs regulation. Aquatic Toxicol 172:1–8. https://doi.org/10.1016/j.aquatox.2015.12.012
Lando D (2002) Asparagine hydroxylation of the HIF transactivation domain: a hypoxic switch. Science 295(5556):858–861
Lang KJD, Kappel A, Goodall GJ (2002) Hypoxia-inducible factor-1α mRNA contains an internal ribosome entry site that allows efficient translation during normoxia and hypoxia. 13(5): 1792-1801
Li HL, Lin HR, Xia JH (2017) Differential gene expression profiles and alternative isoform regulations in gill of Nile tilapia in response to acute hypoxia. Marine Biotechnol 19(6):551–562
Li FG, Chen J, Jiang XY, Zou SM (2015) Transcriptome analysis of blunt snout bream (Megalobrama amblycephala) reveals putative differential expression genes related to growth and hypoxia. PLoS One 10:e0142801
Li J-G, Wang S-Y, Wu X-N (2014) Whole transcriptome sequencing technology and its application in leukemia and lymphoma research. Int J Gene 37(3):124–128
Li W, Tan H, Liu J, Hu J, Cui J, Wang S, Liu Q, Hu F, Ren L, Tao M, Zhao R, Yang C, Qin Q, Liu S (2019) Comparative analysis of testis transcriptomes associated with male infertility in triploid cyprinid fish. Reprod Fertil Dev 31:248–260
Li X-L, Shi H-M, Xia H-Y, Zhou Y-P, Qiu Y-W (2014) Seasonal hypoxia and its potential forming mechanisms in the Mirs Bay, the northern South China Sea. Continental Shelf Res 80:1–7. https://doi.org/10.1016/j.csr.2014.03.003
Liao X, Lei C, Peng X, Guoqing L, Michael W, Xiaowen S, Songlin C, Marc R-R (2013) Transcriptome analysis of crucian carp (Carassius auratus), an important aquaculture and hypoxia-tolerant species. PLoS One 8:e62308
Liu J-W, Mai K-S, Xu W, Zhang Y-J, Zhou H-H, Ai Q-H (2016) Effects of dietary glutamine on activities of nonspecific immune related enzymes and HIF-1α expression after hypoxia of half-smooth tongue sole post larvae. Acta Hydrobiologica Sinica 40(4):736–743
Liu Y, Li E, Xu C, Su Y, Qin JG, Chen L, Wang X (2018) Brain transcriptome profiling analysis of Nile tilapia (Oreochromis niloticus) under long-term hypersaline stress. Frontiers in Physiology, 9https://doi.org/10.3389/fphys.2018.00219
Luo Y (2013) Nutrition requirement of dietary protein and optimal replacement of fish meal protein by rapeseed meal and corn gluten meal in cobia, Rachycentron canadum L. Ocean University of China, China ((MA. Sc. dissertation), in Chinese with English abstract)
Mandic M, Todgham AE, Richards JG (2009) Mechanisms and evolution of hypoxia tolerance in fish. Proceed Royal Soc B: Biol Sci 276(1657):735–744
Martinez LM (2006) Effects of long-term hypoxia on enzymes of carbohydrate metabolism in the Gulf killifish. Fundulus grandis J Exp Biol 209(19):3851–3861
Mohindra V, Tripathi RK, Singh RK (2013) Molecular characterisation and expression analysis of PPP1R3C in hypoxia-tolerant Indian catfish, Clarias batrachus (Linnaeus, 1758) under hypoxia. Gene 530(1):127–133
Mu Y, Li W, Wei Z, He L, Zhang W, Chen X (2020) Transcriptome analysis reveals molecular strategies in gills and heart of large yellow croaker (Larimichthys crocea) under hypoxia stress. Fish & Shellfish Immunol. https://doi.org/10.1016/j.fsi.2020.06.028
Nicholas SA, Sumbayev VV (2010) The role of redox-dependent mechanisms in the downregulation of ligand-induced Toll-like receptors 7, 8 and 4-mediated HIF-1α prolyl hydroxylation. Immunol Cell Biol 88(2):180–186
Olsvik PA, Vibeke V, Lie KK, Hevrøy EM (2013) Transcriptional responses to temperature and low oxygen stress in Atlantic salmon studied with next-generation sequencing technology. BMC Genomics 14:817–837
Pan Y, Chen D-X, Song X-H, Zhang X, Li L-Y (2017) Transcriptome characterisation for Scrophularia ningpoensis based on high-throughput sequencing technology and related genes for synthesis of terpenoid compounds. China J Chinese Materia Medica 42(13):2460–2466
Qi D, Chao Y, Wu R, Xia M, Chen Q, Zheng Z (2018) Transcriptome analysis provides insights into the adaptive responses to hypoxia of a schizothoracine fish (Gymnocypris eckloni). Front Physiol 9. https://doi.org/10.3389/fphys.2018.01326
Qi Y-X, Liu Y-B, Rong W-H (2011) RNA-Seq and its applications: a new technology for transcriptomics. Hereditas 33(11):1191–1202
Qu W-D, Zhao H-Q (2009) Research progress on the role of HIF-1 in promoting angiogenesis under hypoxic conditions. J Oral Sci Res 25(2):234–236
Rankin EB, Rha J, Selak MA, Unger TL, Keith B, Liu Q, Haase VH (2009) Hypoxia-inducible factor 2 regulates hepatic lipid metabolism. Mol Cell Biol 29:4527–4538
Rojas DA, Perez-Munizaga DA, Centanin,L, Antonelli M, Wappner P, Allende ML, Reyes AE (2007). Cloning of hif-1alpha and hif-2alpha and mRNA expression pattern during development in zebrafish. 7(3): 339-345. https://doi.org/10.1016/j.modgep.2006.08.002
Schofield CJ, Ratcliffe PJ (2004) Oxygen sensing by HIF hydroxylases. Nature Rev Mole Cell Biol 5(5):343–354
Shang EH, Wu RS (2004) Aquatic hypoxia is a teratogen and affects fish embryonic development. Environ Sci Technol 38:4763–4767. https://doi.org/10.1021/es0496423
Smith RW, Houlihan DF, Nilsson GN, Alexandre J (1996) Tissue-specific changes in protein synthesis rates in vivo during anoxia in crucian carp. Am J Physiol-Regulatory, Integr Comparative Physiol 271(4):897–904
Stein I, Itin A, Einat P, Skaliter R, Grossman Z, Keshet E (1998) Translation of vascular endothelial growth factor mRNA by internal ribosome entry: implications for translation under hypoxia. Mole Cell Biol 18(6):3112–3119. https://doi.org/10.1128/MCB.18.6.3112
Storz JF, Scott GR, Cheviron ZA (2010) Phenotypic plasticity and genetic adaptation to high-altitude hypoxia in vertebrates. J Exp Biol 213(Pt 24):4125–4136. https://doi.org/10.1242/jeb.048181
Sun L, Liu S, Bao L, Li Y, Feng J, Liu Z (2015) Claudin multigene family in channel catfish and their expression profiles in response to bacterial infection and hypoxia as revealed by meta-analysis of RNA-Seq datasets. Comp Biochem Physiol Part D Genomics Proteomics 13:60–69
Sun H, Li X-B, Fang J (2013) Hypoxia and tumors. Chinese Bullet Life Sci 25(2):206–217
Sun H-C (2006) The influence of HIF-1α and microvessel density as well as apoptosis associated index on the prognosis of pancreatic adenocarcinoma [D]; Zhe Jiang University
Sun S, Xuan F, Fu H, Zhu J, Ge X, Gu Z (2015) Transciptomic and histological analysis of hepatopancreas, muscle and gill tissues of oriental river prawn (Macrobrachium nipponense) in response to chronic hypoxia. BMC Genomics 16:491
Ton C, Stamatiou D, Liew C-C (2003) Gene expression profile of zebrafish exposed to hypoxia during development. Physiol Genomics 13(2):97–106
Trasvi A-Arenas CH, Garcia-Triana A, Peregrino-Uriarte AB, Yepiz-Plascencia G (2013) White shrimp Litopenaeusvannamei catalase: gene structure, expression and activity under hypoxia and reoxygenation. Comp Biochem Physiol Part B: Biochem Mole Biol 164(1):44–52
Vaquer-Sunyer R, Duarte CM (2008) Thresholds of hypoxia for marine biodiversity. Proc Natl Acad Sci USA 105:15452–15457
Wang C-Y, Guo M-Z, Liu X-Y, Liu Y (2015) Structural variation detection algorithm in high-throughput sequencing data. Intelligent Comput App 5(1):1–4
Wang RN, Yoshida K, Toki T, Sawada T, Uechi T, Okuno Y, Sato-Otsubo A, Kudo K, Kamimaki I, Kanezaki R, Shiraishi Y, Chiba K, Tanaka H, Terui K, Sato T, Iribe Y, Ohga S, Kuramitsu M, Hamaguchi I, Ohara A, Hara J, Goi K, Matsubara K, Koike K, Ishiguro A, Okamoto Y, Watanabe K, Kanno H, Kojima S, Miyano S, Kenmochi N, Ogawa S, Ito E (2015) Loss of function mutations in RPL27 and RPS27 identified by whole-exome sequencing in Diamond-Blackfan anaemia. Br J Haematol 168(6):854–64. https://doi.org/10.1111/bjh.13229
Wang Y, Yang L, Wu B, Song Z, He S (2015) Transcriptome analysis of the plateau fish (Triplophysa dalaica): implications for adaptation to hypoxia in fishes. Gene 565:211–220
Wang Z, Zhang S-Y (2017) Recent studies on the evolution of mammalian flight using high-throughput sequencing technology. Chinese Sci Bullet 62(7):631–634
Xiao W, Yang S, Zhu Z, Feng S, Li K (2007) Characterisation of the full-length cDNA, chromosomal localization, and polymorphism of the porcine RPLP0 gene. J Genet Genomics. 34(2):104–108. https://doi.org/10.1016/S1673-8527(07)60011-3
Xu K, Wen M, Duan W, Ren L, Hu F, Xiao J, Wang J, Tao M, Zhang C, Wang J, Zhou Y, Zhang Y, Liu Y, Liu S (2015) Comparative analysis of testis transcriptomes from triploid and fertile diploid cyprinid fish. Biol Reprod 92:95
Xu S, Ying T, Li S (2011) Advance on PPAR-γ function. J Northeast Agri Univ 42(9):1–6
Yasuda S, Sato T, Maekawa S, Aoyama S, Fukao Y, Yamaguchi J (2014) Phosphorylation of Arabidopsis ubiquitin ligase ATL31 is critical for plant carbon/nitrogen nutrient balance response and controls the stability of 14-3-3 proteins. J Biol Chem 289(22):15179–15193
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11(2):R14. https://doi.org/10.1186/gb-2010-11-2-r14
Yu M-C (2017) Molecular adaption of the schizothoracine fishes in the phased uplifting of Qinghai-Tibetan Plateau revealed by transcriptome comparisons. Shanghai Ocean University
Zhang G, Zhao C, Wang Q, Gu Y, Li Z, Tao P, Chen J, Yin S (2017) Identification of HIF-1 signaling pathway in Pelteobagrus vachelli using RNA-Seq: effects of acute hypoxia and reoxygenation on oxygen sensors, respiratory metabolism, and hematology indices. J Comp Physiol B 187(7):931–943. https://doi.org/10.1007/s00360-017-1083-8
Zhang C, Lin C, Fu F, Zhong X, Peng B, Yan H, Zhang J, Zhang W, Wang P, Ding X, Zhang W, Zhao L (2017) Comparative transcriptome analysis of flower heterosis in two soybean F1 hybrids by RNA-seq. PLoS One 12:e0181061
Zhang J, Chen H, Zhou Q, Zhu F (2016) Effects of glutamine on antioxidant capacity and nonspecific immunity of juvenile yellow catfish (Pelteoobagrusfulvidraco). Chinese J Animal Nutrition 28(3):759–765
Zhang Q-Y (2000) The structure and function of PPAR and its biological role. J Environ Hygiene 27(5):284–288
Zhang W-J, Qing G-L (2013) Glutamine metabolism and cancer. Chinese Bullet Life Sci 11:73–78
Zhong XP, Wang D, Zhang YB, Gui JF (2009) Identification and characterisation of hypoxia-induced genes in Carassius auratus blastulae embryonic cells using suppression subtractive hybridisation. Comp Biochem Physiol B Biochem Mol Biol 152:161–170. https://doi.org/10.1016/j.cbpb.2008.10.013
Zhou Q-C, Wu Z-H, Tan B-P, Chi S-Y, Yang Q-H (2006) Optimal dietary methionine requirement for juvenile cobia (Rachycentron canadum). Aquaculture 258:551–557
Zhu L, Lu Y-L, Feng L-S, Zhang S-X (2019) Effects of hypoxia exercise induced miR-27/PPARγ on fatty acids metabolism in gastrocnemius of obese rat. China Sport Sci 39(6):55–61
Funding
This work was supported by grants from Southern Marine Science and Engineering Guangdong Laboratory (Zhanjiang) (ZJW-2019-06) and the China Agriculture Research System of MOF and MARA (CARS-47).
Author information
Authors and Affiliations
Contributions
Jian-Sheng Huang participated in the data curation, project administration, data analysis, and original article writing. Zhi-Xiong Guo participated in the data collection, Jian-Dong Zhang participated in the data collection, Wei-Zheng Wang participated in the data collection, Zhong-Liang Wang supervised the project, and Rui-Tao Xie supervised the project. Eric Amenyogbe did the writing, review, and edited the original article. Gang Chen planned and designed the experiments and acquired funding.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
This study was conducted in accordance with the guidelines of Guangdong Ocean University Research Council for the care and use of laboratory animals (approval number: GDOU-LAE-2020-013).
Consent to participate
All authors voluntarily consent to participate.
Consent for publication
All authors voluntarily consent for publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Pierre Boudry
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, JS., Guo, ZX., Zhang, JD. et al. Transcriptomic analysis of juvenile cobia in response to hypoxic stress. Aquacult Int 31, 931–955 (2023). https://doi.org/10.1007/s10499-022-01007-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-022-01007-1