Abstract
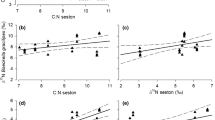
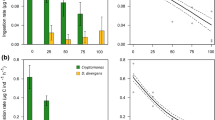
Dunaliella salina (microalgae), Fabrea salina (ciliate) and Artemia salina (crustacean) are the most abundant halophile Eukaryote organisms present in solar salterns at Sfax (Tunisia) when salinity is up to 150 PSU. We analysed the predator/prey relationship between the three organisms in laboratory conditions. In this study, aquatic food web relations were analysed by studying the grazing and energy transfer rates. In solar saltern, the dynamic population of Dunaliella displays a negative relationship with that of Fabrea and Artemia. Grazing experiments confirm that Fabrea and Artemia exercise a top-down control on Dunaliella populations. While Artemia and Fabrea occupy the same trophic level in food chain, the grazing rate of Fabrea on Dunaliella is strongly high reaching 0.85 × 106 ± 0.05 cells mL−1 day−1. The fatty acids (FAs) appeared as good tracers to define the energy transfer along the food chain studied. Effectively, the FA composition of consumers appeared correlated to the FA of their prey. In parallel, the saturated fatty acids (SFAs) content decreased according to the trophic levels, and the opposite was observed for monounsaturated fatty acids (MUFAs). Therefore, palmitic acid (C16:0) showed trends that rendered it useful for tracing trophic transfer to consumers (Artemia). The polyunsaturated fatty acid (PUFA) and especially linoleic acid (C18:2) and linolenic acid (C18:3) were able to be traced across the transfer to Fabrea. This study increased our knowledge of the energy transfer between the major halophile organisms living in the solar salterns at Sfax (Tunisia). We proposed also that the fatty acids are used in future investigations to understand the predator/prey ecological relationship in marine eukaryote organisms.










Similar content being viewed by others
Data availability
The data are available on request.
References
Aaser H, Jeppesen E, Søndergaard M (1995) Seasonal dynamics of the mysid Neomysis integer and its predation on the copepod Eurytemora affinis in a shallow hypertrophic brackish lake. Mar Ecol Prog Ser 127:47–56. https://doi.org/10.3354/meps127047
Abatzopoulos TJ, Kappas I, Bossier P et al (2002) Genetic characterization of Artemia tibetiana (Crustacea: Anostraca). Biol J Linn Soc 75:333–344. https://doi.org/10.1111/j.1095-8312.2002.tb02074.x
Abatzopoulos TJ, Amat F, Baxevanis AD et al (2009) Updating geographic distribution of Artemia urmiana Günther, 1890 (Branchiopoda: Anostraca) in Europe: an integrated and interdisciplinary approach. Int Rev Hydrobiol 94:560–579. https://doi.org/10.1002/iroh.200911147
Abid O, Sellami-Kammoun A, Ayadi H et al (2008) Biochemical adaptation of phytoplankton to salinity and nutrient gradients in a coastal solar saltern, Tunisia. Estuar Coast Shelf Sci 80:391–400. https://doi.org/10.1016/j.ecss.2008.09.007
Albouy C, Archambault P, Appeltans W et al (2019) The marine fish food web is globally connected. Nat Ecol Evol 3:1153–1161. https://doi.org/10.1038/s41559-019-0950-y
Aleya L (1991) Ecophysiological significance of the diel biochemical changes of particulates coupled with metabolic and environmental parameters in two trophically different lakes. Arch Für Hydrobiol pp 403–432
Anderson TR, Pond DW (2000) Stoichiometric theory extended to micronutrients: comparison of the roles of essential fatty acids, carbon, and nitrogen in the nutrition of marine copepods. Limnol Oceanogr 45:1162–1167. https://doi.org/10.4319/lo.2000.45.5.1162
Arendt KE, Jonasdottir SH, Hansen PJ, Gartner S (2005) Effects of dietary fatty acids on the reproductive success of the calanoid copepod Temora longicornis. Mar Biol 146:513–530. https://doi.org/10.1007/s00227-004-1457-9
Asencio AD (2013) Permanent salt evaporation ponds in a semi-arid mediterranean region as model systems to study primary production processes under hypersaline conditions. Estuar Coast Shelf Sci 124:24–33. https://doi.org/10.1016/j.ecss.2013.03.006
Avron M, Ben-Amotz A (1992) Dunaliella: physiology, biochemistry, and biotechnology. CRC Press, Boca Raton
Ayadi H, Abid O, Elloumi J et al (2004) Structure of the phytoplankton communities in two lagoons of different salinity in the Sfax saltern (Tunisia). J Plankton Res 26:669–679. https://doi.org/10.1093/plankt/fbh047
Ayadi H, Elloumi J, Guermazi W et al (2008) Fatty acid composition in relation to the micro-organisms in the Sfax Solar Saltern, Tunisia. Acta Protozool 47:189–203
Bell MV, Tocher DR (2009) Biosynthesis of polyunsaturated fatty acids in aquatic ecosystems: general pathways and new directions. In: Kainz M, Brett MT, Arts MT (eds) Lipids in aquatic ecosystems. Springer, New York, pp 211–236
Bellisario B, Cerfolli F, Nascetti G (2012) The interplay between network structure and functioning of detritus-based communities in patchy aquatic environment. Aquat Ecol 46:431–441. https://doi.org/10.1007/s10452-012-9412-1
Belovsky GE, Stephens D, Perschon C et al (2011) The Great Salt Lake Ecosystem (Utah, USA): long term data and a structural equation approach. Ecosphere 2(3):1–40. https://doi.org/10.1890/ES10-00091.1
Bhavya PS, Kim BK, Jo N et al (2019) A review on the macromolecular compositions of phytoplankton and the implications for aquatic biogeochemistry. Ocean Sci J 54:1–14. https://doi.org/10.1007/s12601-018-0061-8
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917. https://doi.org/10.1139/o59-099
Boëchat IG, Schuran S, Adrian R (2005) Supplementation of the protist Chilomonas paramecium with a highly unsaturated fatty acid enhances its nutritional quality for the rotifer Keratella quadrata. J Plankton Res 27:663–670. https://doi.org/10.1093/plankt/fbi040
Brito AC, Sá C, Brotas V et al (2015) Effect of phytoplankton size classes on bio-optical properties of phytoplankton in the Western Iberian coast: application of models. Remote Sens Environ 156:537–550. https://doi.org/10.1016/j.rse.2014.10.020
Brouard O (2012) Les communautés microbiennes des phytotelmes des Broméliacées: structure et influence de l’habitat, des conditions environnementales et des interactions biologiques. Clermont-Ferrand 2
Bruce LC, Imberger J (2009) The role of zooplankton in the ecological succession of plankton and benthic algae across a salinity gradient in the Shark Bay solar salt ponds. Hydrobiologia 626:111–128. https://doi.org/10.1007/s10750-009-9740-x
Butterwick C, Heaney SI, Talling JF (2005) Diversity in the influence of temperature on the growth rates of freshwater algae, and its ecological relevance. Freshw Biol 50:291–300
Carrasco NK, Perissinotto R (2012) Development of a Halotolerant community in the St. Lucia Estuary (South Africa) during a Hypersaline phase. PLoS One 7:e29927. https://doi.org/10.1371/journal.pone.0029927
Carta G, Murru E, Banni S, Manca C (2017) Palmitic acid: physiological role metabolism and nutritional implications. Front Physiol 8:902. https://doi.org/10.3389/fphys.2017.00902
Chen P, Pan D, Mao Z (2015) Application of a laser fluorometer for discriminating phytoplankton species. Opt Laser Technol 67:50–56. https://doi.org/10.1016/j.optlastec.2014.09.013
Copeman LA, Parrish CC, Brown JA, Harel M (2002) Effects of docosahexaenoic, eicosapentaenoic, and arachidonic acids on the early growth, survival, lipid composition and pigmentation of yellowtail flounder (Limanda ferruginea): a live food enrichment experiment. Aquaculture 210:285–304. https://doi.org/10.1016/S0044-8486(01)00849-3
Copeman L, Parrish C, Gregory R et al (2009) Fatty acid biomarkers in coldwater eelgrass meadows: elevated terrestrial input to the food web of age-0 Atlantic cod Gadus morhua. Mar Ecol Prog Ser 386:237–251. https://doi.org/10.3354/meps08063
Dalsgaard J, St. John M, Kattner G, et al (2003) Fatty acid trophic markers in the pelagic marine environment. Advances in marine biology. Elsevier, pp 225–340
DaSarma S, Arora P (2002) Halophiles. encyclopedia of life sciences. Lond Nat Publ Group 8:458–466
Davis JS (1993) Biological management for problem solving and biological concepts for a new generation of solar saltworks. Seventh symposium on salt. Elsevier, Amsterdam, pp 611–616
Davis JS (2000) Structure, function, and management of the biological system for seasonal solar saltworks. Glob Nest J 2:217–226
Davis JS, Giordano M (1995) Biological and physical events involved in the origin, effects, and control of organic matter in solar saltworks. Int J Salt Lake Res 4:335–347
Desvilettes Ch, Bourdier G, Amblard Ch, Barth B (1997) Use of fatty acids for the assessment of zooplankton grazing on bacteria, protozoans and microalgae. Freshw Biol 38:629–637. https://doi.org/10.1046/j.1365-2427.1997.00241.x
Di Nitto D, Erftemeijer PLA, Van Beek JKL et al (2013) Modelling drivers of mangrove propagule dispersal and restoration of abandoned shrimp farms. Biogeosciences 10:5095–5113. https://doi.org/10.5194/bg-10-5095-2013
DuBois M, Gilles KA, Hamilton JK et al (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356. https://doi.org/10.1021/ac60111a017
Ederington MC, McManus GB, Harvey HR (1995) Trophic transfer of fatty acids, sterols, and a triterpenoid alcohol between bacteria, a ciliate, and the copepod Acartia tonsa. Limnol Oceanogr 40:860–867. https://doi.org/10.4319/lo.1995.40.5.0860
Edis K, Perçin F (2018) Characteristics of biological systems in Çamalti solar saltworks (Izmir/ Turkey). Qual Stud 13:15–25
Elevi Bardavid R, Oren A (2008) Dihydroxyacetone metabolism in Salinibacter ruber and in Haloquadratum walsbyi. Extremophiles 12:125–131
Ellis JM (1937) The morphology, division, and conjugation of the salt-marsh ciliate Fabrea salina Henneguy. No Title
Elloumi J, Carrias J-F, Ayadi H et al (2009a) Communities structure of the planktonic halophiles in the solar saltern of Sfax, Tunisia. Estuar Coast Shelf Sci 81:19–26. https://doi.org/10.1016/j.ecss.2008.09.019
Elloumi J, Guermazi W, Ayadi H et al (2009b) Abundance and biomass of prokaryotic and eukaryotic microorganisms coupled with environmental factors in an arid multi-pond solar saltern (Sfax, Tunisia). J Mar Biol Assoc U K 89:243–253. https://doi.org/10.1017/S0025315408002269
Falkowski PG, Raven JA (2013) Aquatic photosynthesis. Princeton University Press
Falk-Petersen S, Pavlov V, Timofeev S, Sargent JR (2007) Climate variability and possible effects on arctic food chains: The role of Calanus. In: Ørbæk JB, Kallenborn R, Tombre I et al (eds) Arctic Alpine ecosystems and people in a changing environment. Springer, Berlin, Heidelberg, pp 147–166
Farnas MJ (1990) In situ growth rates of marine phytoplankton: approaches to measurement, community and species growth rate. J Plankton Res 12:117–121
Fazeli MR, Tofighi H, Samadi N et al (2006) Carotenoids accumulation by Dunaliella tertiolecta (Lake Urmia isolate) and Dunaliella salina (CCAP 19/18 & WT) under stress conditions. DARU J Pharm Sci 14:146–150
Feki W, Hamza A, Frossard V et al (2013) What are the potential drivers of blooms of the toxic dinoflagellate Karenia selliformis? A 10-year study in the Gulf of Gabes, Tunisia, southwestern Mediterranean Sea. Harmful Algae 23:8–18. https://doi.org/10.1016/j.hal.2012.12.001
Figueiredo J, Van Woesik R, Lin J, Narciso L (2009) Artemia franciscana enrichment model—how to keep them small, rich and alive? Aquaculture 294:212–220. https://doi.org/10.1016/j.aquaculture.2009.05.007
Finkel ZV, Follows MJ, Liefer JD et al (2016) Phylogenetic diversity in the macromolecular composition of microalgae. PLoS One 11:e0155977
Froneman PW (2001) Feeding ecology of the mysid, Mesopodopsis wooldridgei, in a temperate estuary along the eastern seaboard of South Africa. J Plankton Res 23:999–1008. https://doi.org/10.1093/plankt/23.9.999
Frost BW (1972) Effects of size and concentration of food particles on the feeding behavior of the marine planktonic copepod Calanus pacificus. 1: feeding behavior of Calanus. Limnol Oceanogr 17:805–815. https://doi.org/10.4319/lo.1972.17.6.0805
Gascuel D, Morissette L, Palomares MLD, Christensen V (2008) Trophic flow kinetics in marine ecosystems: toward a theoretical approach to ecosystem functioning. Ecol Model 217:33–47. https://doi.org/10.1016/j.ecolmodel.2008.05.012
Gillan FT, Johns RB, Verheyen TV et al (1981) trans -Monounsaturated acids in a marine bacterial isolate. Appl Environ Microbiol 41:849–856. https://doi.org/10.1128/aem.41.4.849-856.1981
Gudmundsdottir R, Palsson S, Hannesdottir ER et al (2013) Diatoms as indicators: the influences of experimental nitrogen enrichment on diatom assemblages in sub-Arctic streams. Ecol Indic 32:74–81. https://doi.org/10.1016/j.ecolind.2013.03.015
Guermazi W, Elloumi J, Ayadi H et al (2008a) Rearing of Fabrea salina Henneguy (Ciliophora, Heterotrichida) with three unicellular feeds. C R Biol 331:56–63. https://doi.org/10.1016/j.crvi.2007.10.006
Guermazi W, Elloumi J, Ayadi H et al (2008b) Coupling changes in fatty acid and protein composition of Artemia salina with environmental factors in the Sfax solar saltern (Tunisia). Aquat Living Resour 21:63–73. https://doi.org/10.1051/alr:2008013
Guermazi W, Ayadi H, Aleya L (2009) Correspondence of the seasonal patterns of the Brine Shrimp, Artemia Salina (Leach, 1819) (Anostraca) with several environmental factors in an arid solar saltern (Sfax, Southern Tunisia). Crustaceana 82:327–348. https://doi.org/10.1163/156854008X390227
Guermazi W, Masmoudi S, Boukhris S et al (2014) Under low irradiation, the light regime modifies growth and metabolite production in various species of microalgae. J Appl Phycol 26:2283–2293. https://doi.org/10.1007/s10811-014-0260-3
Guermazi W, Annabi-Trabelsi N, Belmonte G et al (2023a) Solar salterns and pollution: valorization of some endemic species as sentinels in ecotoxicology. Toxics 11:524. https://doi.org/10.3390/toxics11060524
Guermazi W, Masmoudi S, Trabelsi NA et al (2023b) Physiological and biochemical responses in microalgae Dunaliella salina, Cylindrotheca closterium and Phormidium versicolor NCC466 exposed to high salinity and irradiation. Life 13:313. https://doi.org/10.3390/life13020313
Gunde-Cimerman N, Oren A, Plemenitaš A (2005) Mikrosafari-Čudoviti Svet Mikroorganizmov Solin-The Beautiful World of Microorganisms in the Salterns. Bilingual Slovenian/English publication, Državna Založba Slovenije, Ljubljana
Hagen W, Auel H (2001) Seasonal adaptations and the role of lipids in oceanic zooplankton. Zoology 104:313–326. https://doi.org/10.1078/0944-2006-00037
Hall D, Lee SY, Meziane T (2006) Fatty acids as trophic tracers in an experimental estuarine food chain: tracer transfer. J Exp Mar Biol Ecol 336:42–53. https://doi.org/10.1016/j.jembe.2006.04.004
Harrison PJ, Waters RE, Taylor FJR (1980) A broad spectrum artificial sea water medium for coastal and open ocean phytoplankton. J Phycol 16:28–35. https://doi.org/10.1111/j.0022-3646.1980.00028.x
Haubert D, Häggblom MM, Langel R et al (2006) Trophic shift of stable isotopes and fatty acids in Collembola on bacterial diets. Soil Biol Biochem 38:2004–2007. https://doi.org/10.1016/j.soilbio.2005.11.031
Hillebrand H, Sommer U (1997) Response of epilithic microphytobenthos of the Western Baltic Sea to in situ experiments with nutrient enrichment. Mar Ecol Prog Ser 160:35–46. https://doi.org/10.3354/meps160035
Hinchcliffe PR, Riley JP (1972) The effect of diet on the component fatty-acid composition of Artemia Salina. J Mar Biol Assoc U K 52:203–211. https://doi.org/10.1017/S0025315400018658
Hong HP, Choi JK (2015) Can the halophilic ciliate Fabrea salina be used as a bio-control of microalgae blooms in solar salterns? Ocean Sci J 50:529–536. https://doi.org/10.1007/s12601-015-0048-7
Hotos GN (2019) Feeding with various microalgae the salt “loving” ciliate fabrea salina in normal salinity of 35 ppt. Int J Food Sci Agric. https://doi.org/10.26855/ijfsa.2019.09.001
Howe E, Simenstad C (2007) Restoration trajectories and food web linkages in San Francisco Bays estuarine marshes: a manipulative translocation experiment. Mar Ecol Prog Ser 351:65–76. https://doi.org/10.3354/meps07120
Huertas IE, Rouco M, López-Rodas V, Costas E (2011) Warming will affect phytoplankton differently: evidence through a mechanistic approach. Proc R Soc B Biol Sci 278:3534–3543. https://doi.org/10.1098/rspb.2011.0160
Javor BJ (1983) Planktonic standing crop and nutrients in a saltern ecosystem1. Limnol Oceanogr 28:153–159. https://doi.org/10.4319/lo.1983.28.1.0153
Javor B (1989) Dunaliella and other halophilic, eucaryotic algae. Hypersaline environments. Springer, Berlin, Heidelberg, pp 147–158
Kainz M, Arts MT, Mazumder A (2004) Essential fatty acids in the planktonic food web and their ecological role for higher trophic levels. Limnol Oceanogr 49:1784–1793. https://doi.org/10.4319/lo.2004.49.5.1784
Kelly J, Scheibling R (2012) Fatty acids as dietary tracers in benthic food webs. Mar Ecol Prog Ser 446:1–22. https://doi.org/10.3354/meps09559
Kleiber M (1961) The fire of life. An introduction to animal energetics. Fire Life Introd Anim Energ
Konoplya BI, Soares FS (2011) New geometric models for calculation of microalgal biovolume. Braz Arch Biol Technol 54:527–534. https://doi.org/10.1590/S1516-89132011000300014
Koussoroplis A-M, Bec A, Perga M-E et al (2011) Fatty acid transfer in the food web of a coastal Mediterranean lagoon: evidence for high arachidonic acid retention in fish. Estuar Coast Shelf Sci 91:450–461. https://doi.org/10.1016/j.ecss.2010.11.010
Lamers PP, Janssen M, De Vos RCH et al (2012) Carotenoid and fatty acid metabolism in nitrogen-starved Dunaliella salina, a unicellular green microalga. J Biotechnol 162:21–27. https://doi.org/10.1016/j.jbiotec.2012.04.018
Leu E, Falk-Petersen S, Kwaśniewski S et al (2006) Fatty acid dynamics during the spring bloom in a High Arctic fjord: importance of abiotic factors versus community changes. Can J Fish Aquat Sci 63:2760–2779. https://doi.org/10.1139/f06-159
Lichtenthaler HK (1987) [34] Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods in enzymology. Elsevier, pp 350–382
Litchfield CD, Oren A (2001) Polar lipids and pigments as biomarkers for the study of the microbial community structure of solar salterns. In: Melack JM, Jellison R, Herbst DB (eds) Saline Lakes. Springer, Netherlands, Dordrecht, pp 81–89
Litchfield CD, Irby A, Kis-Papo T, Oren A (2000) Comparisons of the polar lipid and pigment profiles of two solar salterns located in Newark, California, USA, and Eilat, Israel. Extremophiles 4:259–265
Lowry OH, NiraJ R, Farr AL, RoseJ R (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/S0021-9258(19)52451-6
Maciej Gliwicz Z (2002) On the different nature of top-down and bottom-up effects in pelagic food webs: top-down versus bottom-up effects. Freshw Biol 47:2296–2312. https://doi.org/10.1046/j.1365-2427.2002.00990.x
Madkour FF, Gaballah MM (2012) Phytoplankton assemblage of a solar saltern in Port Fouad. Egypt Oceanologia 54:687–700. https://doi.org/10.5697/oc.54-4.687
Marangoni R, Gobbi L, Verni F, et al. (1996) Pigment granules and hypericin-like fluorescence in the marine ciliate Fabrea salina. Acta Protozool 35
Masmoudi S, Tastard E, Guermazi W et al (2015) Salinity gradient and nutrients as major structuring factors of the phytoplankton communities in salt marshes. Aquat Ecol 49:1–19. https://doi.org/10.1007/s10452-014-9500-5
Maszczyk P, Wurtsbaugh WA (2017) Brine shrimp grazing and fecal production increase sedimentation to the deep brine layer (monimolimnion) of Great Salt Lake, Utah. Hydrobiologia 802:7–22. https://doi.org/10.1007/s10750-017-3235-y
Meiners K, Gradinger R, Fehling J et al (2003) Vertical distribution of exopolymer particles in sea ice of the Fram Strait (Arctic) during autumn. Mar Ecol Prog Ser 248:1–13
Mohebbi F (2010) The brine shrimp Artemia and hypersaline environments microalgal composition: a mutual interaction. Int J Aquat Sci 1:19–27
Moraiti-Ioannidou M, Castritsi-Catharios J, Miliou H, Kotzamanis YP (2007) Fatty acid composition and biometry of five Greek Artemia populations suitable for aquaculture purposes. Aquac Res 38:1664–1672. https://doi.org/10.1111/j.1365-2109.2007.01835.x
Moreno-Garrido I, Cañavate JP (2001) Assessing chemical compounds for controlling predator ciliates in outdoor mass cultures of the green algae Dunaliella salina. Aquac Eng 24:107–114. https://doi.org/10.1016/S0144-8609(00)00067-4
Olivotto I, Oliver MP, Turchi C (2017) Larval diets and nutrition. In: Calado R, Olivotto I, Oliver MP, Holt GJ (eds) Marine ornamental species aquaculture, 1st edn. Wiley, pp 125–137
Oren A (2005) A hundred years of Dunaliella research: 1905–2005. Saline Syst 1:2. https://doi.org/10.1186/1746-1448-1-2
Oren A (2009) Saltern evaporation ponds as model systems for the study of primary production processes under hypersaline conditions. Aquat Microb Ecol 56:193–204. https://doi.org/10.3354/ame01297
Oren A, Mana L (2002) Amino acid composition of bulk protein and salt relationships of selected enzymes of Salinibacter ruber, an extremely halophilic bacterium. Extremophiles 6:217–223
Pandey BD (2011) Characteristics of biological systems in solar saltworks. J Basic Appl Biol 5:53–59
Pandey BD, Yeragi SG (2004) Preliminary and mass culture experiments on a heterotrichous ciliate, Fabrea salina. Aquaculture 232:241–254. https://doi.org/10.1016/S0044-8486(03)00459-9
Pandey BD, Yeragi SG, Pal AK (2004) Nutritional value of a heterotrichous ciliate, Fabrea salina with emphasis on its fatty acid profile. Asian-Australas J Anim Sci 17:995–999. https://doi.org/10.5713/ajas.2004.995
Polle JE, Tran D, Ben-Amotz A (2009) History, distribution, and habitats of algae of the genus Dunaliella Teodoresco (Chlorophyceae). Alga Dunaliella, pp 1–14
Pollierer MM, Scheu S, Haubert D (2010) Taking it to the next level: trophic transfer of marker fatty acids from basal resource to predators. Soil Biol Biochem 42:919–925. https://doi.org/10.1016/j.soilbio.2010.02.008
Rebolloso Fuentes M, Acién Fernández CG, Sánchez Pérez JA, Guil Guerrero JL (2000) Biomass nutrient profiles of the microalga Porphyridium cruentum. Food Chem 70:345–353. https://doi.org/10.1016/S0308-8146(00)00101-1
Richmond A (2004) Handbook of microalgae culture: biotechnology and applied phycology. Black Well Science Ltd
Riemann B, Christoffersen K (1993) Microbial trophodynamics in temperate lakes. Mar Microb Food Webs 7:69–100
Rimet F, Bouchez A (2012) Biomonitoring river diatoms: Implications of taxonomic resolution. Ecol Indic 15:92–99. https://doi.org/10.1016/j.ecolind.2011.09.014
Ruess L, Müller-Navarra DC (2019) Essential biomolecules in food webs. Front Ecol Evol 7:269
Ruess L, Häggblom MM, Garcı́a Zapata EJ, Dighton J (2002) Fatty acids of fungi and nematodes—possible biomarkers in the soil food chain? Soil Biol Biochem 34:745–756. https://doi.org/10.1016/S0038-0717(01)00231-0
Ruess L, Hӓggblom MM, Langel R, Scheu S (2004) Nitrogen isotope ratios and fatty acid composition as indicators of animal diets in belowground systems. Oecologia 139:336–346. https://doi.org/10.1007/s00442-004-1514-6
Ruiz O, Medina G, Cohen R et al (2007) Diversity of the fatty acid composition of Artemia spp. cysts from Argentinean populations. Mar Ecol Prog Ser 335:155–165. https://doi.org/10.3354/meps335155
Rustan AC, Drevon CA (2005) Fatty acids: structures and properties, 1st edn. Wiley
Saint-Béat B, Dupuy C, Bocher P et al (2013) Key features of intertidal food webs that support migratory shorebirds. PLoS One 8:e76739. https://doi.org/10.1371/journal.pone.0076739
Sánchez MI, Varo N, Matesanz C et al (2013) Cestodes change the isotopic signature of brine shrimp, Artemia, hosts: Implications for aquatic food webs. Int J Parasitol 43:73–80. https://doi.org/10.1016/j.ijpara.2012.11.003
Shantha NC, Ackman RG (1990) Nervonic acid versus tricosanoic acid as internal standards in quantitative gas chromatographic analyses of fish oil longer-chain n—3 polyunsaturated fatty acid methyl esters. J Chromatogr B Biomed Sci App 533:1–10. https://doi.org/10.1016/S0378-4347(00)82182-9
Sommer U (1995) An experimental test of the intermediate disturbance hypothesis using cultures of marine phytoplankton. Limnol Oceanogr 40:1271–1277. https://doi.org/10.4319/lo.1995.40.7.1271
Sorgeloos P, Lavens P, Leger P, et al (1986) Manual for the culture and use of brine shrimp Artemia in aquaculture. Ghent (Belgium) State University of Ghent, Artemia Reference Center
Stott AW, Davies E, Evershed RP, Tuross N (1997) Monitoring the routing of dietary and biosynthesised lipids through compound - specific stable isotope (δ 13 C) measurements at natural abundance. Naturwissenschaften 84:82–86. https://doi.org/10.1007/s001140050354
Sun J, Feng Y, Zhou F et al (2013) Top-down control of spring surface phytoplankton blooms by microzooplankton in the Central Yellow Sea, China. Deep Sea Res Part II Top Stud Oceanogr 97:51–60. https://doi.org/10.1016/j.dsr2.2013.05.005
Sura SA, Herlihy NS, Mahon HK, Belovsky GE (2017) Environmental impacts on grazing of different brine shrimp (Artemia franciscana) life stages. Hydrobiologia 792:97–104. https://doi.org/10.1007/s10750-016-3047-5
Teodoresco EC (1905) Organisation et développement du Dunaliella, nouveau genre de Volvocacée-Polyblepharidée. Beih z Bot Centralbl. 1905, Bd. XVIII:215–232
Tewfik A, JosephB R, McCann KS (2005) Anthropogenic enrichment alters a marine benthic food web. Ecology 86:2726–2736. https://doi.org/10.1890/04-1537
Uthermöl H (1958) Zur Vervollkommung der quantitativen Phytoplankton Methodik. Mitt. int. Ver Theor
Wang S-J, Wu M-J, Chen X-J et al (2012) DsHsp90 is involved in the early response of Dunaliella salina to environmental stress. Int J Mol Sci 13:7963–7979. https://doi.org/10.3390/ijms13077963
Wurtsbaugh WA, Gliwicz ZM (2001) Limnological control of brine shrimp population dynamics and cyst production in the Great Salt Lake, Utah. In: Saline Lakes: publications from the 7th international conference on Salt Lakes, held in Death Valley National Park, California, USA, September 1999. Springer, pp 119–132
Yu Y, Zhang W, Wang S, Xiao T (2013) Abundance and biomass of planktonic ciliates in the sea area around Zhangzi Island, Northern Yellow Sea. Acta Ecol Sin 33:45–51. https://doi.org/10.1016/j.chnaes.2012.12.007
Zhukova NV, Imbs AB, Yi LF (1998) Diet-induced changes in lipid and fatty acid composition of Artemia salina. Comp Biochem Physiol B Biochem Mol Biol 120:499–506. https://doi.org/10.1016/S0305-0491(98)10036-6
Acknowledgements
This study was supported by the Tunisian Ministry of Scientific Research and Technology. We gratefully acknowledge support from the staff of the Sfax Saltern Company MARE ALB.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Handling editor: Man Xiao.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guermazi, W., Athmouni, K., Annabi-Trabelsi, N. et al. Energy transfers in a predator–prey context involving D. salina (microalga), F. salina (ciliate) and A. salina (crustacean), living in salterns of Sfax (Tunisia). Aquat Ecol 58, 429–449 (2024). https://doi.org/10.1007/s10452-023-10082-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-023-10082-2