Abstract
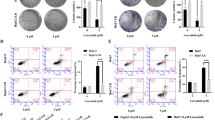
MHY1485 is an mTOR activator that inhibits the autophagy process by inhibiting the fusion between autophagosomes and lysosomes. This study aimed to explore the role and mechanism of MHY1485 in hepatocellular carcinoma (HCC) and to provide an in-depth understanding of the mechanisms of autophagy regulation in relation to adriamycin (ADM) resistance, as well as the development of a molecularly targeted autophagy-modulating approach. Here, ADM was used to treat HepG2 cells and construct an ADM-resistant cell model. The HepG2/ADM cell line and HepG2 cells were treated with MHY1485 and ADM, respectively, and the proliferation and apoptosis of HCC cells were detected using CCK8, clone formation, flow cytometry, and 5-ethynyl-2′-deoxyuridine staining assays. Ki-67, mTOR phosphorylation, and LC3A expression were detected by IF staining; the expression or phosphorylation levels of autophagy-related proteins (i.e., GLUT1, PGI, PFK, END, and MTHFD2) and apoptosis-related proteins (caspase-3, caspase-8, and caspase-9) were detected by qPCR and western blotting. The number of autophagosomes was determined by monodansylcadaverine staining. Our results showed that MHY1485 can inhibit the proliferation and growth of liver cancer cells, and that MHY1485 combined with ADM can effectively inhibit the tolerance of HepG2/ADM cells to ADM and enhance the efficacy of ADM. The results of the detection of the autophagy-related protein LC3A also indicated that MHY1485 activates mTOR and can affect the phosphorylation level of ULK1, inhibit autophagy, and enhance the sensitivity of liver cancer cells to adriamycin. In summary, MHY1485 can enhance the sensitivity of adriamycin-resistant cells to adriamycin by activating mTOR and blocking the autophagy process in cells; therefore, mTOR may become a potential target for the treatment of liver cancer.




Similar content being viewed by others
Data availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
References
Ben-Sahra I, Hoxhaj G, Ricoult SJH, Asara JM, Manning BD (2016) mTORC1 induces purine synthesis through control of the mitochondrial tetrahydrofolate cycle. Science 351:728–733. https://doi.org/10.1126/science.aad0489
Chamoto K, Chowdhury PS, Kumar A, Sonomura K, Matsuda F, Fagarasan S, Honjo T (2017) Mitochondrial activation chemicals synergize with surface receptor PD-1 blockade for T cell-dependent antitumor activity. Proc Natl Acad Sci U S A 114:E761–E770. https://doi.org/10.1073/pnas.1620433114
Chen C, Lu L, Yan S et al (2018) Autophagy and doxorubicin resistance in cancer. Anticancer Drugs 29:1–9. https://doi.org/10.1097/CAD.0000000000000572
Chen S, Zhu X, Qiao H et al (2016) Protective autophagy promotes the resistance of HER2-positive breast cancer cells to lapatinib. Tumour Biol 37:2321–2331. https://doi.org/10.1007/s13277-015-3800-9
Ding ZB, Hui B, Shi YH et al (2011) Autophagy activation in hepatocellular carcinoma contributes to the tolerance of oxaliplatin via reactive oxygen species modulation. Clin Cancer Res 17:6229–6238. https://doi.org/10.1158/1078-0432.CCR-11-0816
Eichhorn PJ, Gili M, Scaltriti M et al (2008) Phosphatidylinositol 3-kinase hyperactivation results in lapatinib resistance that is reversed by the mTOR/phosphatidylinositol 3-kinase inhibitor NVP-BEZ235. Cancer Res 68:9221–9230. https://doi.org/10.1158/0008-5472.CAN-08-1740
Han J, Li J, Tang K, Zhang H, Guo B, Hou N, Huang C (2017) miR-338-3p confers 5-fluorouracil resistance in p53 mutant colon cancer cells by targeting the mammalian target of rapamycin. Exp Cell Res 360:328–336. https://doi.org/10.1016/j.yexcr.2017.09.023
Hu YT, Shu ZY, Jiang JH, Xie QF, Zheng SS (2020) Torin2 overcomes sorafenib resistance via suppressing mTORC2-AKT-BAD pathway in hepatocellular carcinoma cells. Hepatobiliary Pancreat Dis Int 19:547–554. https://doi.org/10.1016/j.hbpd.2020.09.010
Liang Y, Wang X, Wang H et al (2022) IL-33 activates mTORC1 and modulates glycolytic metabolism in CD8(+) T cells. Immunology 165:61–73. https://doi.org/10.1111/imm.13404
Lin M, Hua R, Ma J, Zhou Y, Li P, Xu X, Yu Z, Quan S (2021) Bisphenol A promotes autophagy in ovarian granulosa cells by inducing AMPK/mTOR/ULK1 signalling pathway. Environ Int 147:106298. https://doi.org/10.1016/j.envint.2020.106298
Milani M, Rzymski T, Mellor HR, Pike L, Bottini A, Generali D, Harris AL (2009) The role of ATF4 stabilization and autophagy in resistance of breast cancer cells treated with Bortezomib. Cancer Res 69:4415–4423. https://doi.org/10.1158/0008-5472.CAN-08-2839
Oh J, Kim GD, Kim S, Lee SK (2017) Antofine, a natural phenanthroindolizidine alkaloid, suppresses angiogenesis via regulation of AKT/mTOR and AMPK pathway in endothelial cells and endothelial progenitor cells derived from mouse embryonic stem cells. Food Chem Toxicol 107:201–207. https://doi.org/10.1016/j.fct.2017.06.036
Raje N, Kumar S, Hideshima T et al (2004) Combination of the mTOR inhibitor rapamycin and CC-5013 has synergistic activity in multiple myeloma. Blood 104:4188–4193. https://doi.org/10.1182/blood-2004-06-2281
Sandow TA, Arndt SE, Albar AA et al (2018) Assessment of response to transcatheter arterial chemoembolization with doxorubicin-eluting microspheres: tumor biology and hepatocellular carcinoma recurrence in a 5-year transplant cohort. Radiology 286:1072–1083. https://doi.org/10.1148/radiol.2017170731
Sun L, Morikawa K, Sogo Y, Sugiura Y (2021a) MHY1485 enhances X-irradiation-induced apoptosis and senescence in tumor cells. J Radiat Res 62:782–792. https://doi.org/10.1093/jrr/rrab057
Sun M, Zhao S, Duan Y, Ma Y, Wang Y, Ji H, Zhang Q (2021b) GLUT1 participates in tamoxifen resistance in breast cancer cells through autophagy regulation. Naunyn Schmiedebergs Arch Pharmacol 394:205–216. https://doi.org/10.1007/s00210-020-01893-3
Thorn CF, Oshiro C, Marsh S, Hernandez-Boussard T, McLeod H, Klein TE, Altman RB (2011) Doxorubicin pathways: pharmacodynamics and adverse effects. Pharmacogenet Genomics 21:440–446. https://doi.org/10.1097/FPC.0b013e32833ffb56
Tian T, Li X, Zhang J (2019) mTOR signaling in cancer and mTOR inhibitors in solid tumor targeting therapy. Int J Mol Sci 20. https://doi.org/10.3390/ijms20030755
Umemura A, Park EJ, Taniguchi K et al (2014) Liver damage, inflammation, and enhanced tumorigenesis after persistent mTORC1 inhibition. Cell Metab 20:133–144. https://doi.org/10.1016/j.cmet.2014.05.001
Vogel A, Meyer T, Sapisochin G, Salem R, Saborowski A (2022) Hepatocellular carcinoma. Lancet 400:1345–1362. https://doi.org/10.1016/S0140-6736(22)01200-4
Wang J, He XD, Yao N, Liang WJ, Zhang YC (2013) A meta-analysis of adjuvant therapy after potentially curative treatment for hepatocellular carcinoma. Can J Gastroenterol 27:351–363. https://doi.org/10.1155/2013/417894
Wang Y, Zenkoh J, Gerelchuluun A, Sun L, Cai S, Li X, Tsuboi K (2019) Administration of Dendritic Cells and Anti-PD-1 Antibody Converts X-ray Irradiated Tumors Into Effective In situ Vaccines. Int J Radiat Oncol Biol Phys 103:958–969. https://doi.org/10.1016/j.ijrobp.2018.11.019
Wu S, Wang Y, Ding H et al (2020) The efficacy and safety of the mTOR signaling pathway activator, MHY1485, for in vitro activation of human ovarian tissue. Front Genet 11:603683. https://doi.org/10.3389/fgene.2020.603683
Yang LY, Trujillo JM (1990) Biological characterization of multidrug-resistant human colon carcinoma sublines induced/selected by two methods. Cancer Res 50:3218–3225
Yu L, Shi Q, Jin Y, Liu Z, Li J, Sun W (2021) Blockage of AMPK-ULK1 pathway mediated autophagy promotes cell apoptosis to increase doxorubicin sensitivity in breast cancer (BC) cells: an in vitro study. BMC Cancer 21:195. https://doi.org/10.1186/s12885-021-07901-w
Zhang M, Yue H, Huang X, Wang J, Li Z, Deng X (2022) Novel platinum nanoclusters activate PI3K/AKT/mTOR signaling pathway-mediated autophagy for cisplatin-resistant ovarian cancer therapy. ACS Appl Mater Interfaces 14:48502–48514. https://doi.org/10.1021/acsami.2c15143
Zhang P, Liu X, Li H, Chen Z, Yao X, Jin J, Ma X (2017) TRPC5-induced autophagy promotes drug resistance in breast carcinoma via CaMKKβ/AMPKα/mTOR pathway. Sci Rep 7:3158. https://doi.org/10.1038/s41598-017-03230-w
Zhu K, Yuan Y, Wen J, Chen D, Zhu W, Ouyang Z, Wang W (2020) LncRNA Sox2OT-V7 promotes doxorubicin-induced autophagy and chemoresistance in osteosarcoma via tumor-suppressive miR-142/miR-22. Aging (Albany NY) 12:6644–6666. https://doi.org/10.18632/aging.103004
Zhu L, Lin M (2021) The Synthesis of Nano-Doxorubicin and its Anticancer Effect. Anticancer Agents Med Chem 21:2466–2477. https://doi.org/10.2174/1871520621666201229115612
Author information
Authors and Affiliations
Contributions
JG and YL conceived the study. YL, LL, and ZW performed the experiments. BZ and JF analyzed the data. GL and QG data reviewing and writing. JP review and editing. All authors read and approved the final version of manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was reviewed and approved by the Ethic Committee of the He Xian Memorial Affiliated Hospital of Southern Medical University. No patient was involved in this study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, J., Lei, Y., Liu, L. et al. MHY1485 promotes adriamycin sensitivity in HepG2 cells by inhibiting autophagy. Funct Integr Genomics 24, 22 (2024). https://doi.org/10.1007/s10142-024-01304-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10142-024-01304-3