Abstract
Objectives
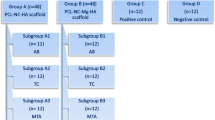
To compare the regenerative properties of human stem cells of the apical papilla (SCAPs) embedded in a platelet-rich plasma (PRP) scaffold, when implanted in vivo using an organotypic model composed of human root segments, with or without the presence of the bioactive cements – ProRoot MTA or Biodentine.
Material and methods
SCAPs were isolated from third molars with incomplete rhizogenesis and expanded and characterized in vitro using stem cell and surface markers. The pluripotency of these cells was also assessed using adipogenic, chondrogenic, and osteogenic differentiation protocols. SCAPs together with a scaffold of PRP were added to the root segment lumen and the organotypic model implanted on the dorsal region of immunodeficient rats for a period of 4 months.
Results
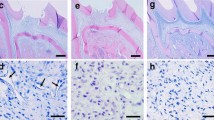
Presence of SCAPs induced de novo formation of dentin-like and pulp-like tissue. A barrier of either ProRoot MTA or Biodentine did not significantly affect the fraction of sections from roots segments observed to contain deposition of hard material (P > 0.05). However, the area of newly deposited dentin was significantly greater in segments containing a barrier of Biodentine compared with ProRoot MTA (P < 0.001).
Conclusions and clinical relevance
SCAPs offer a viable alternative to other dental stem cells (DSCs) in their regenerative properties when enclosed in the microenvironment of human tooth roots. The present study also suggests that the presence of bioactive materials does not hinder or impede the formation of new hard tissues, but the presence of Biodentine may promote greater mineralized tissue deposition.





Similar content being viewed by others
References
Till JE, Mc CE (1961) A direct measurement of the radiation sensitivity of normal mouse bone marrow cells. Radiat Res 14:213–222
Li L, Xie T (2005) Stem cell niche: structure and function. Annu Rev Cell Dev Biol 21:605–631. https://doi.org/10.1146/annurev.cellbio.21.012704.131525
Morrison SJ, Spradling AC (2008) Stem cells and niches: mechanisms that promote stem cell maintenance throughout life. Cell 132:598–611. https://doi.org/10.1016/j.cell.2008.01.038
Gronthos S, Mankani M, Brahim J, Robey PG, Shi S (2000) Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A 97:13625–13630. https://doi.org/10.1073/pnas.240309797
Miura M, Gronthos S, Zhao M, Lu B, Fisher LW, Robey PG, Shi S (2003) SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci U S A 100:5807–5812. https://doi.org/10.1073/pnas.0937635100
Seo BM, Miura M, Gronthos S, Bartold PM, Batouli S, Brahim J, Young M, Robey PG, Wang CY, Shi S (2004) Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 364:149–155. https://doi.org/10.1016/S0140-6736(04)16627-0
Morsczeck C, Gotz W, Schierholz J, Zeilhofer F, Kuhn U, Mohl C, Sippel C, Hoffmann KH (2005) Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biol 24:155–165. https://doi.org/10.1016/j.matbio.2004.12.004
Sonoyama W, Liu Y, Fang D, Yamaza T, Seo BM, Zhang C, Liu H, Gronthos S, Wang CY, Wang S et al (2006) Mesenchymal stem cell-mediated functional tooth regeneration in swine. PLoS One 1:e79. https://doi.org/10.1371/journal.pone.0000079
Sonoyama W, Liu Y, Yamaza T, Tuan RS, Wang S, Shi S, Huang GT (2008) Characterization of the apical papilla and its residing stem cells from human immature permanent teeth: a pilot study. J Endod 34:166–171. https://doi.org/10.1016/j.joen.2007.11.021
Volponi AA, Pang Y, Sharpe PT (2010) Stem cell-based biological tooth repair and regeneration. Trends Cell Biol 20:715–722. https://doi.org/10.1016/j.tcb.2010.09.012
Zhang W, Walboomers XF, Shi S, Fan M, Jansen JA (2006) Multilineage differentiation potential of stem cells derived from human dental pulp after cryopreservation. Tissue Eng 12:2813–2823. https://doi.org/10.1089/ten.2006.12.2813
Kim BC, Bae H, Kwon IK, Lee EJ, Park JH, Khademhosseini A, Hwang YS (2012) Osteoblastic/cementoblastic and neural differentiation of dental stem cells and their applications to tissue engineering and regenerative medicine. Tissue Eng Part B Rev 18:235–244. https://doi.org/10.1089/ten.TEB.2011.0642
Nuti N, Corallo C, Chan BM, Ferrari M, Gerami-Naini B (2016) Multipotent differentiation of human dental pulp stem cells: a literature review. Stem Cell Rev Rep 12:511–523. https://doi.org/10.1007/s12015-016-9661-9
Chalisserry EP, Nam SY, Park SH, Anil S (2017) Therapeutic potential of dental stem cells. J Tissue Eng 8:2041731417702531. https://doi.org/10.1177/2041731417702531
Mita T, Furukawa-Hibi Y, Takeuchi H, Hattori H, Yamada K, Hibi H, Ueda M, Yamamoto A (2015) Conditioned medium from the stem cells of human dental pulp improves cognitive function in a mouse model of Alzheimer's disease. Behav Brain Res 293:189–197. https://doi.org/10.1016/j.bbr.2015.07.043
Yang KL, Chen MF, Liao CH, Pang CY, Lin PY (2009) A simple and efficient method for generating Nurr1-positive neuronal stem cells from human wisdom teeth (tNSC) and the potential of tNSC for stroke therapy. Cytotherapy 11:606–617. https://doi.org/10.1080/14653240902806994
Gandia C, Arminan A, Garcia-Verdugo JM, Lledo E, Ruiz A, Minana MD, Sanchez-Torrijos J, Paya R, Mirabet V, Carbonell-Uberos F et al (2008) Human dental pulp stem cells improve left ventricular function, induce angiogenesis, and reduce infarct size in rats with acute myocardial infarction. Stem Cells 26:638–645. https://doi.org/10.1634/stemcells.2007-0484
Ishikawa J, Takahashi N, Matsumoto T, Yoshioka Y, Yamamoto N, Nishikawa M, Hibi H, Ishigro N, Ueda M, Furukawa K et al (2016) Factors secreted from dental pulp stem cells show multifaceted benefits for treating experimental rheumatoid arthritis. Bone 83:210–219. https://doi.org/10.1016/j.bone.2015.11.012
Chen YJ, Zhao YH, Zhao YJ, Liu NX, Lv X, Li Q, Chen FM, Zhang M (2015) Potential dental pulp revascularization and odonto-/osteogenic capacity of a novel transplant combined with dental pulp stem cells and platelet-rich fibrin. Cell Tissue Res 361:439–455. https://doi.org/10.1007/s00441-015-2125-8
Hayashi Y, Murakami M, Kawamura R, Ishizaka R, Fukuta O, Nakashima M (2015) CXCL14 and MCP1 are potent trophic factors associated with cell migration and angiogenesis leading to higher regenerative potential of dental pulp side population cells. Stem Cell Res Ther 6:111. https://doi.org/10.1186/s13287-015-0088-z
Gronthos S, Brahim J, Li W, Fisher LW, Cherman N, Boyde A, DenBesten P, Robey PG, Shi S (2002) Stem cell properties of human dental pulp stem cells. J Dent Res 81:531–535. https://doi.org/10.1177/154405910208100806
Cordeiro MM, Dong Z, Kaneko T, Zhang Z, Miyazawa M, Shi S, Smith AJ, Nor JE (2008) Dental pulp tissue engineering with stem cells from exfoliated deciduous teeth. J Endod 34:962–969. https://doi.org/10.1016/j.joen.2008.04.009
Rosa V, Zhang Z, Grande RH, Nor JE (2013) Dental pulp tissue engineering in full-length human root canals. J Dent Res 92:970–975. https://doi.org/10.1177/0022034513505772
Nakashima M, Iohara K, Murakami M, Nakamura H, Sato Y, Ariji Y, Matsushita K (2017) Pulp regeneration by transplantation of dental pulp stem cells in pulpitis: a pilot clinical study. Stem Cell Res Ther 8:61. https://doi.org/10.1186/s13287-017-0506-5
Xuan K, Li B, Guo H, Sun W, Kou X, He X, Zhang Y, Sun J, Liu A, Liao L et al (2018) Deciduous autologous tooth stem cells regenerate dental pulp after implantation into injured teeth. Sci Transl Med 10. https://doi.org/10.1126/scitranslmed.aaf3227
Na S, Zhang H, Huang F, Wang W, Ding Y, Li D, Jin Y (2016) Regeneration of dental pulp/dentine complex with a three-dimensional and scaffold-free stem-cell sheet-derived pellet. J Tissue Eng Regen Med 10:261–270. https://doi.org/10.1002/term.1686
Palma PJ, Ramos JC, Martins JB, Diogenes A, Figueiredo MH, Ferreira P, Viegas C, Santos JM (2017) Histologic evaluation of regenerative endodontic procedures with the use of chitosan scaffolds in immature dog teeth with apical periodontitis. J Endod 43:1279–1287. https://doi.org/10.1016/j.joen.2017.03.005
Chen X, Bao ZF, Liu Y, Liu M, Jin XQ, Xu XB (2013) Regenerative endodontic treatment of an immature permanent tooth at an early stage of root development: a case report. J Endod 39:719–722. https://doi.org/10.1016/j.joen.2012.12.023
Xu J, Gou L, Zhang P, Li H, Qiu S (2020) Platelet-rich plasma and regenerative dentistry. Aust Dent J 65:131–142. https://doi.org/10.1111/adj.12754
Meschi N, Castro AB, Vandamme K, Quirynen M, Lambrechts P (2016) The impact of autologous platelet concentrates on endodontic healing: a systematic review. Platelets 27:613–633. https://doi.org/10.1080/09537104.2016.1226497
Alsousou J, Ali A, Willett K, Harrison P (2013) The role of platelet-rich plasma in tissue regeneration. Platelets 24:173–182. https://doi.org/10.3109/09537104.2012.684730
Gong T, Heng BC, Lo EC, Zhang C (2016) Current advance and future prospects of tissue engineering approach to dentin/pulp regenerative therapy. Stem Cells Int 2016:9204574. https://doi.org/10.1155/2016/9204574
Sequeira DB, Seabra CM, Palma PJ, Cardoso AL, Peça J, Santos JM (2018) Effects of a New Bioceramic Material on Human Apical Papilla Cells. J Funct Biomater 9:74. https://doi.org/10.3390/jfb9040074
Saberi EA, Karkehabadi H, Mollashahi NF (2016) Cytotoxicity of various endodontic materials on stem cells of human apical papilla. Iran Endod J 11:17–22. https://doi.org/10.7508/iej.2016.01.004
Araujo LB, Cosme-Silva L, Fernandes AP, Oliveira TM, Cavalcanti BDN, Gomes Filho JE, Sakai VT (2018) Effects of mineral trioxide aggregate, BiodentineTM and calcium hydroxide on viability, proliferation, migration and differentiation of stem cells from human exfoliated deciduous teeth. J Appl Oral Sci 26:e20160629. https://doi.org/10.1590/1678-7757-2016-0629
Paranjpe A, Zhang H, Johnson JD (2010) Effects of mineral trioxide aggregate on human dental pulp cells after pulp-capping procedures. JEndod 36:1042–1047. https://doi.org/10.1016/j.joen.2010.02.013
D'Anto V, Di Caprio MP, Ametrano G, Simeone M, Rengo S, Spagnuolo G (2010) Effect of mineral trioxide aggregate on mesenchymal stem cells. J Endod 36:1839–1843. https://doi.org/10.1016/j.joen.2010.08.010
Pelepenko LE, Saavedra F, Antunes TBM, Bombarda GF, Gomes B, Zaia AA, Camilleri J, Marciano MA (2021) Physicochemical, antimicrobial, and biological properties of White-MTAFlow. Clin Oral Investig 25:663–672. https://doi.org/10.1007/s00784-020-03543-7
Palma PJ, Marques JA, Falacho RI, Correia E, Vinagre A, Santos JM, Ramos JC (2019) Six-Month Color Stability Assessment of Two Calcium Silicate-Based Cements Used in Regenerative Endodontic Procedures. J Funct Biomater 10. https://doi.org/10.3390/jfb10010014
Sequeira D, Palma P, Diogo P, Cardoso A, Santos JM (2016) The effect of calcium silicate-based cements on viability of stem cells within apical papilla. Int Endod J 0:1–90
Galler KM, Widbiller M, Buchalla W, Eidt A, Hiller KA, Hoffer PC, Schmalz G (2016) EDTA conditioning of dentine promotes adhesion, migration and differentiation of dental pulp stem cells. Int Endod J 49:581–590. https://doi.org/10.1111/iej.12492
Huang GT, Sonoyama W, Liu Y, Liu H, Wang S, Shi S (2008) The hidden treasure in apical papilla: the potential role in pulp/dentin regeneration and bioroot engineering. J Endod 34:645–651. https://doi.org/10.1016/j.joen.2008.03.001
Smith AJ, Scheven BA, Takahashi Y, Ferracane JL, Shelton RM, Cooper PR (2012) Dentine as a bioactive extracellular matrix. Arch Oral Biol 57:109–121. https://doi.org/10.1016/j.archoralbio.2011.07.008
Trevino EG, Patwardhan AN, Henry MA, Perry G, Dybdal-Hargreaves N, Hargreaves KM, Diogenes A (2011) Effect of irrigants on the survival of human stem cells of the apical papilla in a platelet-rich plasma scaffold in human root tips. J Endod 37:1109–1115. https://doi.org/10.1016/j.joen.2011.05.013
Ring KC, Murray PE, Namerow KN, Kuttler S, Garcia-Godoy F (2008) The comparison of the effect of endodontic irrigation on cell adherence to root canal dentin. J Endod 34:1474–1479. https://doi.org/10.1016/j.joen.2008.09.001
da Rosa WLO, Piva E, da Silva AF (2018) Disclosing the physiology of pulp tissue for vital pulp therapy. Int Endod J 51:829–846. https://doi.org/10.1111/iej.12906
Hilkens P, Driesen RB, Wolfs E, Gervois P, Vangansewinkel T, Ratajczak J, Dillen Y, Bronckaers A, Lambrichts I (2016) Cryopreservation and Banking of Dental Stem Cells. Adv Exp Med Biol 951:199–235. https://doi.org/10.1007/978-3-319-45457-3_17
Palma PJ, Martins J, Diogo P, Sequeira D, Ramos JC, Diogenes A, Santos JM (2019) Does apical papilla survive and develop in apical periodontitis presence after regenerative endodontic procedures? Appl Sci 9:3942
Jazayeri HE, Lee SM, Kuhn L, Fahimipour F, Tahriri M, Tayebi L (2020) Polymeric scaffolds for dental pulp tissue engineering: A review. Dent Mater 36:e47–e58. https://doi.org/10.1016/j.dental.2019.11.005
Galler KM, D'Souza RN, Hartgerink JD, Schmalz G (2011) Scaffolds for dental pulp tissue engineering. Adv Dent Res 23:333–339. https://doi.org/10.1177/0022034511405326
Galler KM, Brandl FP, Kirchhof S, Widbiller M, Eidt A, Buchalla W, Gopferich A, Schmalz G (2018) Suitability of different natural and synthetic biomaterials for dental pulp tissue engineering. Tissue Eng A 24:234–244. https://doi.org/10.1089/ten.TEA.2016.0555
Blair P, Flaumenhaft R (2009) Platelet alpha-granules: basic biology and clinical correlates. Blood Rev 23:177–189. https://doi.org/10.1016/j.blre.2009.04.001
Anitua E, Alkhraisat MH, Orive G (2012) Perspectives and challenges in regenerative medicine using plasma rich in growth factors. J Control Release 157:29–38. https://doi.org/10.1016/j.jconrel.2011.07.004
Jeanneau C, Lundy FT, El Karim IA, About I (2017) Potential therapeutic strategy of targeting pulp fibroblasts in dentin-pulp regeneration. J Endod 43:S17–S24. https://doi.org/10.1016/j.joen.2017.06.007
Chmilewsky F, Jeanneau C, Laurent P, About I (2014) Pulp fibroblasts synthesize functional complement proteins involved in initiating dentin-pulp regeneration. Am J Pathol 184:1991–2000. https://doi.org/10.1016/j.ajpath.2014.04.003
Mathieu S, Jeanneau C, Sheibat-Othman N, Kalaji N, Fessi H, About I (2013) Usefulness of controlled release of growth factors in investigating the early events of dentin-pulp regeneration. J Endod 39:228–235. https://doi.org/10.1016/j.joen.2012.11.007
Chmilewsky F, Jeanneau C, Laurent P, Kirschfink M, About I (2013) Pulp progenitor cell recruitment is selectively guided by a C5a gradient. J Dent Res 92:532–539. https://doi.org/10.1177/0022034513487377
Tran-Hung L, Laurent P, Camps J, About I (2008) Quantification of angiogenic growth factors released by human dental cells after injury. Arch Oral Biol 53:9–13. https://doi.org/10.1016/j.archoralbio.2007.07.001
Rombouts C, Giraud T, Jeanneau C, About I (2017) Pulp vascularization during tooth development, regeneration, and therapy. Dent Res 96:137–144. https://doi.org/10.1177/0022034516671688
Chmilewsky F, About I, Chung SH (2016) pulp fibroblasts control nerve regeneration through complement activation. J Dent Res 95:913–922. https://doi.org/10.1177/0022034516643065
Moss-Salentijn L, Applebaum E, Lammé AT (1972) Orofacial histology and embryology: a visual integration. F. A. Davis, Philadelphia, p xii 75 p. and slide set (180 col. slides. 182 × 182 in.)
Kucukkaya S, Gorduysus MO, Zeybek ND, Muftuoglu SF (2016) In Vitro Cytotoxicity of Calcium Silicate-Based Endodontic Cement as Root-End Filling Materials. Scientifica 2016:9203932. https://doi.org/10.1155/2016/9203932
Luo Z, Li D, Kohli MR, Yu Q, Kim S, He WX (2014) Effect of Biodentine on the proliferation, migration and adhesion of human dental pulp stem cells. J Dent 42:490–497. https://doi.org/10.1016/j.jdent.2013.12.011
Zanini M, Sautier JM, Berdal A, Simon S (2012) Biodentine induces immortalized murine pulp cell differentiation into odontoblast-like cells and stimulates biomineralization. J Endod 38:1220–1226. https://doi.org/10.1016/j.joen.2012.04.018
Erakovic M, Duka M, Bekic M, Tomic S, Ismaili B, Vucevic D, Colic M (2020) Anti-inflammatory and immunomodulatory effects of Biodentine on human periapical lesion cells in culture. Int Endod J. https://doi.org/10.1111/iej.13351
Acknowledgments
The authors thank Cláudia Brites, dedicated technician of the Hard Tissues Histology Laboratory, Faculty of Medicine, University of Coimbra, for assisting with the histological processing and staining. This project received funding from GAI, Gabinete Apoio à Investigação, Faculty of Medicine, University of Coimbra and the European Union’s Horizon 2020 research and innovation programme under grant agreement Number 799164. This work was financed by the European Regional Development Fund (ERDF), through the Centro 2020 and through the COMPETE 2020 - OperationalProgramme for Competitiveness and Internationalisation and Portuguese national funds via FCT – Fundação para a Ciência e a Tecnologia, under project UIDB/04539/2020.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
The procedures and study protocol described here were approved by the Ethics Committee of the Faculty of Medicine, University of Coimbra (Project CE-028/2016) and following the guidelines of the Declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sequeira, D.B., Oliveira, A.R., Seabra, C.M. et al. Regeneration of pulp-dentin complex using human stem cells of the apical papilla: in vivo interaction with two bioactive materials. Clin Oral Invest 25, 5317–5329 (2021). https://doi.org/10.1007/s00784-021-03840-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-021-03840-9