Abstract
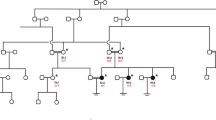
Oocyte maturation defects are major phenotypes resulting in female infertility. Although many genetic factors have been found to be responsible for these phenotypes, the underlying pathogenic genes and variants remain to be identified. The anaphase promoting complex or cyclosome (APC/C) is known to be essential in the metaphase-to-anaphase transition. In this study, we identified two homozygous missense variants (c.986A > G, p.Y329C and c.988C > T, p.R330C) in CDC23 that are responsible for female infertility characterized by oocyte maturation defects in three infertile individuals. CDC23 (cell division cycle 23) is one of the core subunits of the APC/C. In vitro experiments showed that the variant c.986A > G (p.Y329C) led to a decrease in CDC23 protein level and the variant c.988C > T (p.R330C) changed the localization of CDC23 in HeLa cells and mouse oocytes. In vivo studies showed that Cdc23Y329C/Y329C mice successfully mimicked the patients’ phenotype by causing low expression of CDC23 and APC4 and the accumulation of securin and cyclin B1 in oocytes. AZ3146 treatment was able to rescue the phenotype. Taken together, our findings reveal the important roles of CDC23 in human oocyte maturation and provide a new genetic marker for female infertility.

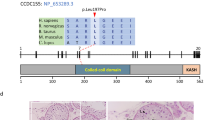
Source data of CDC23 protein feature are from UniProt



Similar content being viewed by others
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
References
Chen B, Zhang Z, Sun X et al (2017) Biallelic Mutations in PATL2 Cause Female Infertility Characterized by Oocyte Maturation Arrest. Am J Hum Genet 101:609–615. https://doi.org/10.1016/j.ajhg.2017.08.018
Fan H-Y, Sun Q-Y (2019) Chapter 12 - Oocyte Meiotic Maturation. In: Leung PCK, Adashi EY (eds) The Ovary (Third Edition). Academic Press, pp 181–203
Feng R, Sang Q, Kuang Y et al (2016a) Mutations in TUBB8 cause human oocyte meiotic arrest. N Engl J Med 374:223–232. https://doi.org/10.1056/NEJMoa1510791
Feng R, Yan Z, Li B et al (2016b) Mutations in TUBB8 cause a multiplicity of phenotypes in human oocytes and early embryos. J Med Genet 53:662–671. https://doi.org/10.1136/jmedgenet-2016-103891
Greaney J, Wei Z, Homer H (2018) Regulation of chromosome segregation in oocytes and the cellular basis for female meiotic errors. Hum Reprod Update 24:135–161. https://doi.org/10.1093/humupd/dmx035
Homer H (2013) The APC/C in female mammalian meiosis I. Reproduction 146:R61–R71. https://doi.org/10.1530/REP-13-0163
Jiao S-Y, Yang Y-H, Chen S-R (2021) Molecular genetics of infertility: loss-of-function mutations in humans and corresponding knockout/mutated mice. Hum Reprod Update 27:154–189. https://doi.org/10.1093/humupd/dmaa034
Jones KT (2011) Anaphase-Promoting Complex Control in Female Mouse Meiosis. In: Kubiak JZ (ed) Cell Cycle in Development. Springer, Berlin, Heidelberg, pp 343–363
Kudo NR, Anger M, Peters AHFM et al (2009) Role of cleavage by separase of the Rec8 kleisin subunit of cohesin during mammalian meiosis I. J Cell Sci 122:2686–2698. https://doi.org/10.1242/jcs.035287
Li Q, Zhao L, Zeng Y et al (2023) Large-scale analysis of de novo mutations identifies risk genes for female infertility characterized by oocyte and early embryo defects. Genome Biol 24:68. https://doi.org/10.1186/s13059-023-02894-0
Maddirevula S, Coskun S, Alhassan S et al (2017) Female Infertility Caused by Mutations in the Oocyte-Specific Translational Repressor PATL2. Am J Hum Genet 101:603–608. https://doi.org/10.1016/j.ajhg.2017.08.009
Maddirevula S, Coskun S, Al-Qahtani M et al (2022) ASTL is mutated in female infertility. Hum Genet 141:49–54. https://doi.org/10.1007/s00439-021-02388-8
Mansfeld J, Collin P, Collins MO et al (2011) APC15 drives the turnover of MCC-CDC20 to make the spindle assembly checkpoint responsive to kinetochore attachment. Nat Cell Biol 13:1234–1243. https://doi.org/10.1038/ncb2347
Marangos P, Carroll J (2004) The dynamics of cyclin B1 distribution during meiosis I in mouse oocytes. Reproduction 128:153–162. https://doi.org/10.1530/rep.1.00192
McGuinness BE, Anger M, Kouznetsova A et al (2009) Regulation of APC/C Activity in Oocytes by a Bub1-Dependent Spindle Assembly Checkpoint. Curr Biol 19:369–380. https://doi.org/10.1016/j.cub.2009.01.064
Mihajlović AI, FitzHarris G (2018) Segregating Chromosomes in the Mammalian Oocyte. Curr Biol 28:R895–R907. https://doi.org/10.1016/j.cub.2018.06.057
Mu J, Wang W, Chen B et al (2019) Mutations in NLRP2 and NLRP5 cause female infertility characterised by early embryonic arrest. J Med Genet 56:471–480. https://doi.org/10.1136/jmedgenet-2018-105936
Nabti I, Grimes R, Sarna H et al (2017) Maternal age-dependent APC/C-mediated decrease in securin causes premature sister chromatid separation in meiosis II. Nat Commun 8:15346. https://doi.org/10.1038/ncomms15346
Pines J (2011) Cubism and the cell cycle: the many faces of the APC/C. Nat Rev Mol Cell Biol 12:427–438. https://doi.org/10.1038/nrm3132
So C, Seres KB, Steyer AM et al (2019) A liquid-like spindle domain promotes acentrosomal spindle assembly in mammalian oocytes. Science. https://doi.org/10.1126/science.aat9557
Thomas C, Wetherall B, Levasseur MD et al (2021) A prometaphase mechanism of securin destruction is essential for meiotic progression in mouse oocytes. Nat Commun 12:4322. https://doi.org/10.1038/s41467-021-24554-2
Thornton BR, Ng TM, Matyskiela ME et al (2006) An architectural map of the anaphase-promoting complex. Genes Dev 20:449–460. https://doi.org/10.1101/gad.1396906
Uzunova K, Dye BT, Schutz H et al (2012) APC15 mediates CDC20 autoubiquitylation by APC/CMCC and disassembly of the mitotic checkpoint complex. Nat Struct Mol Biol 19:1116–1123. https://doi.org/10.1038/nsmb.2412
Wang W, Miyamoto Y, Chen B et al (2023) Karyopherin α deficiency contributes to human preimplantation embryo arrest. J Clin Invest. https://doi.org/10.1172/JCI159951
Wu L, Chen H, Li D et al (2019) Novel mutations in PATL2: expanding the mutational spectrum and corresponding phenotypic variability associated with female infertility. J Hum Genet 64:379–385. https://doi.org/10.1038/s10038-019-0568-6
Wu T, Dong J, Fu J et al (2022) The mechanism of acentrosomal spindle assembly in human oocytes. Science. https://doi.org/10.1126/science.abq7361
Xu R-Y, Xu J, Wang L et al (2019) The Arabidopsis anaphase-promoting complex/cyclosome subunit 8 is required for male meiosis. New Phytol 224:229–241. https://doi.org/10.1111/nph.16014
Zhang L, Rahbari R, He M, Kebebew E (2011) CDC23 regulates cancer cell phenotype and is overexpressed in papillary thyroid cancer. Endocr Relat Cancer 18:731–742. https://doi.org/10.1530/ERC-11-0181
Zhang Z, Li B, Fu J et al (2020) Bi-allelic Missense Pathogenic Variants in TRIP13 Cause Female Infertility Characterized by Oocyte Maturation Arrest. Am J Hum Genet 107:15–23. https://doi.org/10.1016/j.ajhg.2020.05.001
Zhao L, Xue S, Yao Z et al (2020) Biallelic mutations in CDC20 cause female infertility characterized by abnormalities in oocyte maturation and early embryonic development. Protein Cell 11:921–927. https://doi.org/10.1007/s13238-020-00756-0
Zhou Q, Li J, Yue W et al (2020) Cell division cycle 23 is required for mouse oocyte meiotic maturation. FASEB J 34:8990–9002. https://doi.org/10.1096/fj.202000131R
Zhou C-J, Wang X-Y, Dong Y-H et al (2022) CENP-F-dependent DRP1 function regulates APC/C activity during oocyte meiosis I. Nat Commun 13:7732. https://doi.org/10.1038/s41467-022-35461-5
Acknowledgements
We would like to thank the patients and their families and the female healthy volunteers who participated in this study.
Funding
This work was supported by the National Key Research and Development Program of China (2021YFC2700100), the National Natural Science Foundation of China (82288102, 32130029, 82171643, 81971450,81971382), New Cornerstone Science Foundation through the XPLORER PRIZE, the Project of the Shanghai Municipal Science and Technology Commission (21XD1420300) and the China Postdoctoral Science Fund (2022M712147).
Author information
Authors and Affiliations
Contributions
HF, ZZ, WZ, YG, and QM contributed equally to this work. LW and QS conceived and designed the study. WZ, YG, QM, LW, JZ, and GL contributed to the recruitment and characterization of the patients. BC organized the medical records and analyzed the whole-exome data. HF, ZZ, WW, JD, YZ, RL, and HG performed the experiments. HF and ZZ analyzed the data. HF, ZZ, and QS drafted the article and revised the manuscript. All authors approved the final manuscript to be published and were accounted for all aspects of the work.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
Our study was approved by the ethics committee of the Medical College of Fudan University and the Reproductive Study Ethics Committees of the hospitals, and written informed consent was obtained from the affected individuals.
Consent to participate
All contributors provided written informed consent to participate in this study.
Consent to publish
All contributors provided written informed consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fan, H., Zhou, Z., Zheng, W. et al. Homozygous variants in CDC23 cause female infertility characterized by oocyte maturation defects. Hum. Genet. 142, 1621–1631 (2023). https://doi.org/10.1007/s00439-023-02606-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-023-02606-5