Abstract
Purpose
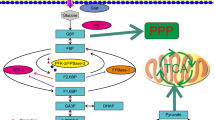
This study was to investigate the biological effect of 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 2 (PFKFB2) in colorectal cancer (CRC).
Methods
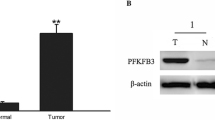
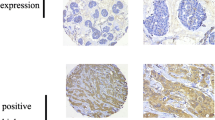
PFKFB2 was selected by metabolism polymerase chain reaction (PCR) array from CRC cells under alkaline culture medium (pH 7.4) and acidic culture medium (pH 6.8). The expression of PFKFB2 mRNA and protein was detected by quantitative real-time PCR and immunohistochemistry in 70 paired fresh and 268 paired paraffin-embedded human CRC tissues, respectively, and then the prognostic value of PFKFB2 was investigated. The effects of PFKFB2 on CRC cells were also verified in vitro, which were through detecting the change of migration, invasion, sphere formation, proliferation, colony formation, and extracellular acidification rate of CRC cells after PFKFB2 knockdown in alkaline culture medium (pH 7.4) and overexpression in acidic culture medium (pH 6.8).
Results
PFKFB2 expression was downregulated in acidic culture medium (pH 6.8). In addition, we found PFKFB2 expression decreased in human CRC tissues compared with the adjacent normal tissues. Furthermore, the OS and DFS rate of CRC patients with low PFKFB2 expression was significantly shorter than those of patients with high PFKFB2 expression. Multivariate analysis indicated that low PFKFB2 expression was an independent prognostic factor for both OS and DFS in CRC patients. Moreover, the abilities of migration, invasion, spheroidizing ability, proliferation, and colony formation of CRC cells were significantly increased after depletion of PFKFB2 in alkaline culture medium (pH 7.4) and decreased after overexpression of PFKFB2 in acidic culture medium (pH 6.8) in vitro. Epithelial–mesenchymal transition (EMT) pathway was found and verified involved in the PFKFB2-mediated regulation of metastatic function in CRC cells. Further, glycolysis of CRC cells was significantly elevated after knockdown of PFKFB2 in alkaline culture medium (pH 7.4) and decreased after overexpression of PFKFB2 in acidic culture medium (pH 6.8).
Conclusion
PFKFB2 expression is downregulated in CRC tissues and associated with worse survival for CRC patients. PFKFB2 could inhibit metastasis and the malignant progression of CRC cells by suppressing EMT and glycolysis.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abadjian MZ, Edwards WB, Anderson CJ (2017) Imaging the tumor microenvironment. Adv Exp Med Biol 1036:229–257. https://doi.org/10.1007/978-3-319-67577-0_15
Barar J, Omidi Y (2013) Dysregulated pH in tumor microenvironment checkmates cancer therapy. Bioimpacts 3:149–162. https://doi.org/10.5681/bi.2013.036
Boedtkjer E, Pedersen SF (2020) The acidic tumor microenvironment as a driver of cancer. Annu Rev Physiol 82:103–126. https://doi.org/10.1146/annurev-physiol-021119-034627
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Brown KGM, Solomon MJ, Mahon K, O’Shannassy S (2019) Management of colorectal cancer. BMJ 366:14561. https://doi.org/10.1136/bmj.l4561
Calorini L, Peppicelli S, Bianchini F (2012) Extracellular acidity as favouring factor of tumor progression and metastatic dissemination. Exp Oncol 34:79–84
Chen W et al (2018) Cancer incidence and mortality in China, 2014. Chin J Cancer Res 30:1–12. https://doi.org/10.21147/j.issn.1000-9604.2018.01.01
Ellis R et al (2019) Increased expression and phosphorylation of 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase isoforms in urinary exosomes in pre-eclampsia. J Transl Med 17:60. https://doi.org/10.1186/s12967-019-1806-6
Gallagher FA et al (2008) Magnetic resonance imaging of pH in vivo using hyperpolarized 13C-labelled bicarbonate. Nature 453:940–943. https://doi.org/10.1038/nature07017
Guraya SY (2019) Pattern, stage, and time of recurrent colorectal cancer after curative surgery. Clin Colorectal Cancer 18:e223–e228. https://doi.org/10.1016/j.clcc.2019.01.003
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. https://doi.org/10.1016/j.cell.2011.02.013
He Z, You C, Zhao D (2018) Long non-coding RNA UCA1/miR-182/PFKFB2 axis modulates glioblastoma-associated stromal cells-mediated glycolysis and invasion of glioma cells. Biochem Biophys Res Commun 500:569–576. https://doi.org/10.1016/j.bbrc.2018.04.091
Houles T et al (2018) RSK regulates PFK-2 activity to promote metabolic rewiring in melanoma. Cancer Res 78:2191–2204. https://doi.org/10.1158/0008-5472.CAN-17-2215
Hu PS et al (2020) VDR-SOX2 signaling promotes colorectal cancer stemness and malignancy in an acidic microenvironment. Signal Transduct Target Ther 5:183. https://doi.org/10.1038/s41392-020-00230-7
Ji D et al (2014) MACC1 expression correlates with PFKFB2 and survival in hepatocellular carcinoma. Asian Pac J Cancer Prev 15:999–1003. https://doi.org/10.7314/apjcp.2014.15.2.999
Ju HQ et al (2019) Modulation of redox homeostasis by inhibition of MTHFD2 in colorectal cancer: mechanisms and therapeutic implications. J Natl Cancer Inst 111:584–596. https://doi.org/10.1093/jnci/djy160
Kotowski K et al (2021) Role of PFKFB3 and PFKFB4 in cancer: genetic basis, impact on disease development/progression, and potential as therapeutic targets. Cancers (Basel) 13. https://doi.org/10.3390/cancers13040909
Liu H et al (2019) miR-613 inhibits Warburg effect in gastric cancer by targeting PFKFB2. Biochem Biophys Res Commun 515:37–43. https://doi.org/10.1016/j.bbrc.2019.05.001
Liu J et al (2020) Long noncoding RNA AGPG regulates PFKFB3-mediated tumor glycolytic reprogramming. Nat Commun 11:1507. https://doi.org/10.1038/s41467-020-15112-3
Lu YX et al (2018) Pharmacological ascorbate suppresses growth of gastric cancer cells with GLUT1 overexpression and enhances the efficacy of oxaliplatin through redox modulation. Theranostics 8:1312–1326. https://doi.org/10.7150/thno.21745
Lucien F et al (2017) Hypoxia-induced mobilization of NHE6 to the plasma membrane triggers endosome hyperacidification and chemoresistance. Nat Commun 8:15884. https://doi.org/10.1038/ncomms15884
Minchenko O, Opentanova I, Caro J (2003) Hypoxic regulation of the 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase gene family (PFKFB-1-4) expression in vivo. FEBS Lett 554:264–270. https://doi.org/10.1016/s0014-5793(03)01179-7
Pascale RM, Calvisi DF, Simile MM, Feo CF, Feo F (2020) The Warburg effect 97 years after its discovery. Cancers (Basel) 12. https://doi.org/10.3390/cancers12102819
Peppicelli S, Bianchini F, Calorini L (2014) Extracellular acidity, a “reappreciated” trait of tumor environment driving malignancy: perspectives in diagnosis and therapy. Cancer Metastasis Rev 33:823–832. https://doi.org/10.1007/s10555-014-9506-4
Rider MH, Bertrand L, Vertommen D, Michels PA, Rousseau GG, Hue L (2004) 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase: head-to-head with a bifunctional enzyme that controls glycolysis. Biochem J 381:561–579. https://doi.org/10.1042/BJ20040752
Riemann A, Rauschner M, Giesselmann M, Reime S, Haupt V, Thews O (2019) Extracellular acidosis modulates the expression of epithelial-mesenchymal transition (EMT) markers and adhesion of epithelial and tumor cells. Neoplasia 21:450–458. https://doi.org/10.1016/j.neo.2019.03.004
Ros S, Schulze A (2013) Balancing glycolytic flux: the role of 6-phosphofructo-2-kinase/fructose 2,6-bisphosphatases in cancer metabolism. Cancer Metab 1:8. https://doi.org/10.1186/2049-3002-1-8
Sanders E, Diehl S (2015) Analysis and interpretation of transcriptomic data obtained from extended Warburg effect genes in patients with clear cell renal cell carcinoma. Oncoscience 2:151–186. https://doi.org/10.18632/oncoscience.128
Schwartz L, Supuran CT, Alfarouk KO (2017) The Warburg effect and the hallmarks of cancer. Anticancer Agents Med Chem 17:164–170. https://doi.org/10.2174/1871520616666161031143301
Sharma V, Kaur R, Bhatnagar A, Kaur J (2015) Low-pH-induced apoptosis: role of endoplasmic reticulum stress-induced calcium permeability and mitochondria-dependent signaling. Cell Stress Chaperones 20:431–440. https://doi.org/10.1007/s12192-014-0568-6
Stuwe L et al (2007) pH dependence of melanoma cell migration: protons extruded by NHE1 dominate protons of the bulk solution. J Physiol 585:351–360. https://doi.org/10.1113/jphysiol.2007.145185
Sutoo S, Maeda T, Suzuki A, Kato Y (2020) Adaptation to chronic acidic extracellular pH elicits a sustained increase in lung cancer cell invasion and metastasis. Clin Exp Metastasis 37:133–144. https://doi.org/10.1007/s10585-019-09990-1
Tian XP et al (2019) Acidic microenvironment up-regulates exosomal miR-21 and miR-10b in early-stage hepatocellular carcinoma to promote cancer cell proliferation and metastasis. Theranostics 9:1965–1979. https://doi.org/10.7150/thno.30958
Uhlen M et al (2015) Proteomics. Tissue-based map of the human proteome. Science 347:1260419. https://doi.org/10.1126/science.1260419
van der Stok EP, Spaander MCW, Grunhagen DJ, Verhoef C, Kuipers EJ (2017) Surveillance after curative treatment for colorectal cancer. Nat Rev Clin Oncol 14:297–315. https://doi.org/10.1038/nrclinonc.2016.199
Yang H et al (2019) 6-Phosphofructo-2-kinase/fructose-2,6-biphosphatase-2 regulates TP53-dependent paclitaxel sensitivity in ovarian and breast cancers. Clin Cancer Res 25:5702–5716. https://doi.org/10.1158/1078-0432.CCR-18-3448
Yi M, Ban Y, Tan Y, Xiong W, Li G, Xiang B (2019) 6-Phosphofructo-2-kinase/fructose-2,6-biphosphatase 3 and 4: a pair of valves for fine-tuning of glucose metabolism in human cancer. Mol Metab 20:1–13. https://doi.org/10.1016/j.molmet.2018.11.013
Zhang J, Zhang Q (2019) Using seahorse machine to measure OCR and ECAR in cancer cells. Methods Mol Biol 1928:353–363. https://doi.org/10.1007/978-1-4939-9027-6_18
Zhao L et al (2017) Long noncoding RNA LINC00092 acts in cancer-associated fibroblasts to drive glycolysis and progression of ovarian cancer. Cancer Res 77:1369–1382. https://doi.org/10.1158/0008-5472.CAN-16-1615
Zhao SJ et al (2018) SLIT2/ROBO1 axis contributes to the Warburg effect in osteosarcoma through activation of SRC/ERK/c-MYC/PFKFB2 pathway. Cell Death Dis 9:390. https://doi.org/10.1038/s41419-018-0419-y
Acknowledgements
The authors would like to acknowledge the helpful comments regarding this paper received from the reviewers.
Funding
This work was supported by the Precision Oncology Research Program of Guangdong Province (2019/1–2020/12), Medical Scientific Research Foundation of Guangdong Province (A2022054), Natural Science Foundation of Guangdong Province (No. 2021A1515011767) and Fundamental Research Funds for the Central Universities, Sun Yat-sen University (22qntd3601).
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by LFR, WXL and CZH. CYX performed the bioinformatics analysis. The first draft of the manuscript was written by LFR. All the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, F., Wei, X., Chen, Z. et al. PFKFB2 is a favorable prognostic biomarker for colorectal cancer by suppressing metastasis and tumor glycolysis. J Cancer Res Clin Oncol 149, 10737–10752 (2023). https://doi.org/10.1007/s00432-023-04946-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04946-1