Abstract
Purpose
In biliary tract cancer (BTC), malignancy is strongest at the invasion front. To improve the BTC prognosis, the invasion front should be controlled. We evaluated tumor–stroma crosstalk at the tumor center and at the invasion front of BTC lesions. We investigated the expression of SPARC, a marker of cancer-associated fibroblasts, and determined its ability to predict BTC prognosis after neoadjuvant chemoradiotherapy (NAC-RT).
Methods
We performed immunohistochemistry to evaluate SPARC expression in resected specimens from patients that underwent BTC surgery. We established highly invasive (HI) clones in two BTC cell lines (NOZ, CCLP1), and performed mRNA microarrays to compare gene expression in parental and HI cells.
Results
Among 92 specimens, stromal SPARC expression was higher at the invasion front than at the lesion center (p = 0.014). Among 50 specimens from patients treated with surgery alone, high stromal SPARC expression at the invasion front was associated with a poor prognosis (recurrence-free survival: p = 0.033; overall survival: p = 0.017). Coculturing fibroblasts with NOZ-HI cells upregulated fibroblast SPARC expression. mRNA microarrays showed that connective tissue growth factor (CTGF) was upregulated in NOZ-HI and CCLP1-HI cells. A CTGF knockdown suppressed cell invasion in NOZ-HI cells. Exogeneous CTGF upregulated SPARC expression in fibroblasts. SPARC expression at the invasion front was significantly lower after NAC-RT, compared to surgery alone (p = 0.003).
Conclusion
CTGF was associated with tumor–stroma crosstalk in BTC. CTGF activated stromal SPARC expression, which promoted tumor progression, particularly at the invasion front. SPARC expression at the invasion front after NAC-RT may serve as a prognosis predictor.





Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Affo S, Nair A, Brundu F et al (2021) Promotion of cholangiocarcinoma growth by diverse cancer-associated fibroblast subpopulations. Cancer Cell 39:866-882.e811
Alford AI, Hankenson KD (2006) Matricellular proteins: extracellular modulators of bone development, remodeling, and regeneration. Bone 38:749–757
Aoki S, Inoue K, Klein S et al (2022) Placental growth factor promotes tumour desmoplasia and treatment resistance in intrahepatic cholangiocarcinoma. Gut 71:185–193
Bloomston M, Ellison EC, Muscarella P et al (2006) Stromal osteonectin overexpression is associated with poor outcome in patients with ampullary cancer. Ann Surg Oncol 14:211–217
Chen P-S, Wang M-Y, Wu S-N et al (2007) CTGF enhances the motility of breast cancer cells via an integrin-αvβ3–ERK1/2-dependent S100A4-upregulated pathway. J Cell Sci 120:2053–2065
Galván JA, Wiprächtiger J, Slotta-Huspenina J et al (2020) Immunohistochemical analysis of the expression of cancer-associated fibroblast markers in esophageal cancer with and without neoadjuvant therapy. Virchows Arch 476:725–734
Guweidhi A, Kleeff J, Adwan H et al (2005) Osteonectin influences growth and invasion of pancreatic cancer cells. Ann Surg 242:224–234
Homma S, Hasumura S, Nagamori S, Kameda H (1988) Establishment and characterization of a human gall bladder carcinoma cell line NOZ. Hum Cell 1:95–97
Infante JR, Matsubayashi H, Sato N et al (2007) Peritumoral fibroblast SPARC expression and patient outcome with resectable pancreatic adenocarcinoma. J Clin Oncol 25:319–325
Jiang C-G, Lv L, Liu F-R et al (2013) Connective tissue growth factor is a positive regulator of epithelial–mesenchymal transition and promotes the adhesion with gastric cancer cells in human peritoneal mesothelial cells. Cytokine 61:173–180
Jun J-I, Lau LF (2011) Taking aim at the extracellular matrix: CCN proteins as emerging therapeutic targets. Nat Rev Drug Discov 10:945–963
Kim H, Son S (2019) Therapeutic potential of connective tissue growth factor (CTGF) in triple-negative breast cancer. Ann Oncol 30:8–8
Kim B, Kim H, Jung S et al (2020) A CTGF–RUNX2–RANKL axis in breast and prostate cancer cells promotes tumor progression in bone. J Bone Miner Res 35:155–166
Kinoshita M, Kobayashi S, Gotoh K et al (2020) Heterogeneity of Treg/Th17 according to cancer progression and modification in biliary tract cancers via self-producing cytokines. Dig Dis Sci 65:2937–2948
Kobayashi S, Gotoh K, Takahashi H et al (2016) Clinicopathological features of surgically-resected biliary tract cancer following chemo-radiation therapy. Anticancer Res 36:335–342
Kobayashi S, Tomokuni A, Gotoh K et al (2017) A retrospective analysis of the clinical effects of neoadjuvant combination therapy with full-dose gemcitabine and radiation therapy in patients with biliary tract cancer. EJSO 43:763–771
Kular L, Pakradouni J, Kitabgi P, Laurent M, Martinerie C (2011) The CCN family: a new class of inflammation modulators? Biochimie 93:377–388
Liu B-C, Zhang J-D, Zhang X-L, Wu G-Q, Li M-X (2006) Role of connective tissue growth factor (CTGF) module 4 in regulating epithelial mesenchymal transition (EMT) in HK-2 cells. Clin Chim Acta 373:144–150
Lobe C, Vallette M, Arbelaiz A et al (2021) ZEB1 promotes cholangiocarcinoma progression through tumor dedifferentiation and tumor–stroma paracrine signaling. Hepatology 74:3194–3212
Makino Y, Hikita H, Kodama T et al (2018) CTGF mediates tumor–stroma interactions between hepatoma cells and hepatic stellate cells to accelerate HCC progression. Cancer Res 78:4902–4914
Mazzocca A, Fransvea E, Dituri F, Lupo L, Antonaci S, Giannelli G (2010) Down-regulation of connective tissue growth factor by inhibition of transforming growth factor β blocks the tumor–stroma cross-talk and tumor progression in hepatocellular carcinoma. Hepatology 51:523–534
McMasters KM, Tuttle TM, Leach SD, Rich T, Cleary KR, Curley SA (1997) Neoadjuvant chemoradiation for extrahepatic cholangiocarcinoma. Am J Surg 174:605–609
Miyakawa S, Ishihara S, Horiguchi A, Takada T, Miyazaki M, Nagakawa T (2009) Biliary tract cancer treatment: 5,584 results from the Biliary Tract Cancer Statistics Registry from 1998 to 2004 in Japan. J Hepatobiliary Pancreat Surg 16:1–7
Morizane C, Okusaka T, Mizusawa J et al (2019) Combination gemcitabine plus S-1 versus gemcitabine plus cisplatin for advanced/recurrent biliary tract cancer: the FUGA-BT (JCOG1113) randomized phase III clinical trial. Ann Oncol 30:1950–1958
Nakeeb A, Pitt HA, Sohn TA et al (1996) Cholangiocarcinoma—a spectrum of intrahepatic, perihilar, and distal tumors. Ann Surg 224:463–473
Ouyang G, Liu Q, Wu Y et al (2021) The global, regional, and national burden of gallbladder and biliary tract cancer and its attributable risk factors in 195 countries and territories, 1990 to 2017: a systematic analysis for the Global Burden of Disease Study 2017. Cancer 127:2238–2250
Picozzi V, Alseidi A, Winter J et al (2020) Gemcitabine/nab-paclitaxel with pamrevlumab: a novel drug combination and trial design for the treatment of locally advanced pancreatic cancer. Esmo Open 5:e000668
Rachna TS, Katherine A, Aaron JS et al (2023) SWOG 1815: a phase III randomized trial of gemcitabine, cisplatin, and nab-paclitaxel versus gemcitabine and cisplatin in newly diagnosed, advanced biliary tract cancers. J Clin Oncol 41(4 suppl):LBA490
Shi Q, Bao S, Song L et al (2007) Targeting SPARC expression decreases glioma cellular survival and invasion associated with reduced activities of FAK and ILK kinases. Oncogene 26:4084–4094
Sumiyoshi T, Shima Y, Okabayashi T et al (2018) Chemoradiotherapy for initially unresectable locally advanced cholangiocarcinoma. World J Surg 42:2910–2918
Takagi K, Imura J, Shimomura A et al (2020) Establishment of highly invasive pancreatic cancer cell lines and the expression of IL-32. Oncol Lett 20:2888–2896
Valle JW (2010) Advances in the treatment of metastatic or unresectable biliary tract cancer. Ann Oncol 21:345–348
Valle J, Wasan H, Palmer DH et al (2010) Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med 362:1273–1281
Wang SJ, Lemieux A, Kalpathy-Cramer J et al (2011) Nomogram for predicting the benefit of adjuvant chemoradiotherapy for resected gallbladder cancer. J Clin Oncol 29:4627–4632
Wang YZ, Li J, Xia Y et al (2013) Prognostic nomogram for intrahepatic cholangiocarcinoma after partial hepatectomy. J Clin Oncol 31:1188–1195
Yamada D, Kobayashi S, Wada H et al (2013) Role of crosstalk between interleukin-6 and transforming growth factor-beta 1 in epithelial–mesenchymal transition and chemoresistance in biliary tract cancer. Eur J Cancer 49:1725–1740
Acknowledgements
The authors would like to thank San Francisco Edit (https://www.sfedit.net/) for the English language editing.
Funding
The authors declare that no funds, grants, or other support was received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
HT, SK, KG, YI and HE were involved in study design and data interpretation. HT, SK, KG, KS, YI, DY, YT, TN, HT, HA, HW, TA, MT, YD and HE were involved in the data analysis. All authors critically revised the report, commented on drafts of the manuscript, and approved the final report.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Human Ethics Review Committee of the Graduate School of Medicine, Osaka University (No. 20493).
Consent to participate
Written informed consent was obtained from all patients included in the study.
Consent to publish
Not applicable as all data are anonymized.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
432_2023_4835_MOESM1_ESM.tif
Supplementary fig. 1 SPARC is primarily expressed in fibroblast cells and rarely in biliary tract cancer (BTC) cells. (a) qRT-PCR results show SPARC mRNA expression and (b) Western blot shows SPARC protein expression in normal human dermal fibroblasts (NHDFs), BTC cells (NOZ-parental), and highly invasive BTC cells (NOZ-HI) (TIF 677 KB)
432_2023_4835_MOESM2_ESM.tif
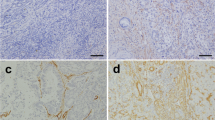
Supplementary fig. 2 Evaluation of CTGF and SPARC immunostaining in BTC resected specimens. Immunohistochemistry of CTGF and SPARC in BTC resected specimens. (a) Representative figure of positive staining for CTGF (b) Representative figure of positive staining for SPARC (c) Representative figure of negative staining for CTGF (d) Representative figure of negative staining for SPARC. (a) and (b), (c) and (d) are the same field of the same specimen (TIF 8460 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takayama, H., Kobayashi, S., Gotoh, K. et al. SPARC accelerates biliary tract cancer progression through CTGF-mediated tumor–stroma interactions: SPARC as a prognostic marker of survival after neoadjuvant therapy. J Cancer Res Clin Oncol 149, 10935–10950 (2023). https://doi.org/10.1007/s00432-023-04835-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04835-7