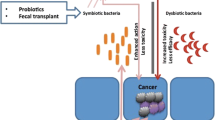
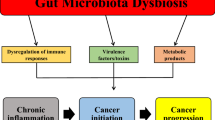
Abstract
Purpose
Colorectal cancer (CRC) is the third most common cancer worldwide, and its incidence and mortality rates are increasing every year. The intestinal microbiota has been called the "neglected organ" and there is growing evidence that the intestinal microbiota and its metabolites can be used in combination with immunotherapy, radiotherapy and chemotherapy to greatly enhance the treatment of colorectal cancer and to address some of the side effects and adverse effects of these therapies. Antibiotics have great potential to eliminate harmful microbiota, control infection, and reduce colorectal cancer side effects. However, the use of antibiotics has been a highly controversial issue, and numerous retrospective studies have shown that the use of antibiotics affects the effectiveness of treatment (especially immunotherapy). Understanding the bi-directional role of the gut microbiota and antibiotics will further enhance our research into the diagnosis and treatment of cancer.
Methods
We searched the "PubMed" database and selected the following keywords "intestinal microbiota, antibiotics, treatment, prevention, colorectal cancer". In this review, we discuss the role of the intestinal microbiota in immunotherapy, radiotherapy, chemotherapy, diagnosis, and prevention of CRC. We also conclude that the intestinal microbiota and antibiotics work together to promote the treatment of CRC through a bidirectional effect.
Results
We found that the intestinal microbiota plays a key role in promoting immunotherapy, chemotherapy, radiotherapy, diagnosis and prevention of CRC. In addition, gut microbiota and antibiotic interactions could be a new strategy for CRC treatment.
Conclusion
The bi-directional role of the intestinal microbiota and antibiotics plays a key role in the prevention, diagnosis, and treatment of colorectal cancer.


Similar content being viewed by others
Abbreviations
- ACT:
-
Adoptive T cell
- APC:
-
Antigen-presenting cells
- BLI:
-
β-Lactamase inhibitors
- CAR:
-
Chimeric antigen receptors
- CAR-T:
-
Chimeric antigen receptor T
- CD133:
-
Cluster of differentiation 133
- CIN:
-
Chromosomal instability
- CIMP:
-
CpG island methylator phenotype
- CRC:
-
Colorectal cancer
- CTLA4:
-
Cytotoxic-T-lymphocyte- associated antigen 4
- DC:
-
Intestinal dendritic cells
- EpCAM:
-
Epithelial cell adhesion molecule
- EPIs:
-
Efflux pump inhibitors
- FMT:
-
Fecal microbiota transplantation
- FXR:
-
Farnesoid X receptor
- GUCY2C:
-
Guanylyl cyclase 2 C
- HER2:
-
Human epidermal growth factor receptor 2
- ICIs:
-
Immune checkpoint inhibitors
- JAX:
-
Jackson laboratories
- MPE:
-
Molecular pathological epidemiology
- MSI:
-
Microsatellite instability
- MSLN:
-
Mesothelin
- MUC-1:
-
Mucin-1
- NMR:
-
Nuclear Magnetic Resonance
- TME:
-
Tumor microenvironment
- TLR:
-
Toll-like receptor
- TLS:
-
Tumor-infiltrating lymphocytes
- PFS:
-
Progression-free survival
- PD-1:
-
Programmed cell death-ligand 1
- OS:
-
Overall survival
- SCFAs:
-
Short-chain fatty acids
- TCR:
-
T cell receptors
- Tregs:
-
Regulatory T cells
- TNF-α:
-
Tumor necrosis factor alpha
- VDR:
-
Vitamin D receptor
References
Abdulamir AS, Hafidh RR, Mahdi LK et al (2009) Investigation into the controversial association of Streptococcus gallolyticus with colorectal cancer and adenoma. BMC Cancer 9:403. https://doi.org/10.1186/1471-2407-9-403
Ahmed J, Kumar A, Parikh K et al (2018) Use of broad-spectrum antibiotics impacts outcome in patients treated with immune checkpoint inhibitors. Oncoimmunology 7(11):e1507670. https://doi.org/10.1080/2162402x.2018.1507670
Akbari M, Shomali N, Faraji A et al (2020) CD133: An emerging prognostic factor and therapeutic target in colorectal cancer. Cell Biol Int 44(2):368–80. https://doi.org/10.1002/cbin.11243
Aneke-Nash C, Yoon G, Du M et al (2021) Antibiotic use and colorectal neoplasia: a systematic review and meta-analysis. BMJ Open Gastroenterol. https://doi.org/10.1136/bmjgast-2021-000601
Aparicio C, Belver M, Enríquez L (2021) Cell therapy for colorectal cancer: the promise of chimeric antigen receptor (CAR)-T cells. Int J Mol Sci. https://doi.org/10.3390/ijms222111781
Arnold M, Sierra MS, Laversanne M et al (2017) Global patterns and trends in colorectal cancer incidence and mortality. Gut 66(4):683–91. https://doi.org/10.1136/gutjnl-2015-310912
Avril M, DePaolo RW (2021) “Driver-passenger” bacteria and their metabolites in the pathogenesis of colorectal cancer. Gut Microbes 13(1):1941710. https://doi.org/10.1080/19490976.2021.1941710
Barclay J, Creswell J, León J (2018) Cancer immunotherapy and the PD-1/PD-L1 checkpoint pathway. Arch Esp Urol 71(4):393–399
Barker HE, Paget JT, Khan AA et al (2015) The tumour microenvironment after radiotherapy: mechanisms of resistance and recurrence. Nat Rev Cancer 15(7):409–25. https://doi.org/10.1038/nrc3958
Bashir A, Miskeen AY, Bhat A et al (2015) Fusobacterium nucleatum: an emerging bug in colorectal tumorigenesis. Eur J Cancer Prevent 24(5):373–85. https://doi.org/10.1097/cej.0000000000000116
Becattini S, Taur Y, Pamer EG (2016) Antibiotic-induced changes in the intestinal microbiota and disease. Trends Mol Med 22(6):458–78. https://doi.org/10.1016/j.molmed.2016.04.003
Bezabeh T, Somorjai R, Dolenko B et al (2009) Detecting colorectal cancer by 1H magnetic resonance spectroscopy of fecal extracts. NMR Biomed 22(6):593–600. https://doi.org/10.1002/nbm.1372
Bingham SA, Day NE, Luben R et al (2003) Dietary fibre in food and protection against colorectal cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC): an observational study. Lancet (London, England) 361(9368):1496–501. https://doi.org/10.1016/s0140-6736(03)13174-1
Borgo C, Ruzzene M (2019) Role of protein kinase CK2 in antitumor drug resistance. J Exp Clin Cancer Res 38(1):287. https://doi.org/10.1186/s13046-019-1292-y
Bray F, Ferlay J, Soerjomataram I et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424. https://doi.org/10.3322/caac.21492
Bullman S, Pedamallu CS, Sicinska E et al (2017) Analysis of Fusobacterium persistence and antibiotic response in colorectal cancer. Science 358(6369):1443–48. https://doi.org/10.1126/science.aal5240
Campbell C, McKenney PT (2020) Bacterial metabolism of bile acids promotes generation of peripheral regulatory T cells. Nature 581(7809):475–79. https://doi.org/10.1038/s41586-020-2193-0
Campbell C, Marchildon F, Michaels AJ et al (2020) FXR mediates T cell-intrinsic responses to reduced feeding during infection. Proc Natl Acad Sci 117(52):33446–54. https://doi.org/10.1073/pnas.2020619117
Cekic C, Linden J (2014) Adenosine A2A receptors intrinsically regulate CD8+ T cells in the tumor microenvironment. Cancer Res 74(24):7239–49. https://doi.org/10.1158/0008-5472.can-13-3581
Chang CY, Pan TM (2018) Anticancer and antimigration effects of a combinatorial treatment of 5-fluorouracil and Lactobacillus paracasei subsp. paracasei NTU 101 fermented skim milk extracts on colorectal cancer cells. J Agric Food Chem 66(22):5549–55. https://doi.org/10.1021/acs.jafc.8b01445
Chen W, Liu F, Ling Z et al (2012a) Human intestinal lumen and mucosa-associated microbiota in patients with colorectal cancer. PloS One 7(6):e39743. https://doi.org/10.1371/journal.pone.0039743
Chen CC, Lin WC, Kong MS et al (2012b) Oral inoculation of probiotics Lactobacillus acidophilus NCFM suppresses tumour growth both in segmental orthotopic colon cancer and extra-intestinal tissue. Br J Nutr 107(11):1623–34. https://doi.org/10.1017/s0007114511004934
Chen L, Zou Y, Peng J et al (2015) Lactobacillus acidophilus suppresses colitis-associated activation of the IL-23/Th17 axis. J Immunol Res 2015:909514. https://doi.org/10.1155/2015/909514
Cho J, Kim KM, Kim HC et al (2019) The prognostic role of tumor associated glycoprotein 72 (TAG-72) in stage II and III colorectal adenocarcinoma. Pathol Res Pract 215(1):171–76. https://doi.org/10.1016/j.prp.2018.10.024
Chung L, Orberg ET, Geis AL et al (2018) Bacteroides fragilis toxin coordinates a pro-carcinogenic inflammatory cascade via targeting of colonic epithelial cells. Cell Host Microbe 23(3):421. https://doi.org/10.1016/j.chom.2018.02.004
Ciorba MA, Riehl TE, Rao MS et al (2012) Lactobacillus probiotic protects intestinal epithelium from radiation injury in a TLR-2/cyclo-oxygenase-2-dependent manner. Gut 61(6):829–38. https://doi.org/10.1136/gutjnl-2011-300367
Coppenhagen-Glazer S, Sol A, Abed J et al (2015) Fap2 of Fusobacterium nucleatum is a galactose-inhibitable adhesin involved in coaggregation, cell adhesion, and preterm birth. Infect Immun 83(3):1104–13. https://doi.org/10.1128/iai.02838-14
Cui B, Li P, Xu L et al (2015) Step-up fecal microbiota transplantation strategy: a pilot study for steroid-dependent ulcerative colitis. J Transl Med 13:298. https://doi.org/10.1186/s12967-015-0646-2
Cui M, Xiao H, Li Y et al (2017) Faecal microbiota transplantation protects against radiation-induced toxicity. EMBO Mol Med 9(4):448–61. https://doi.org/10.15252/emmm.201606932
Dahm CC, Keogh RH, Spencer EA et al (2010) Dietary fiber and colorectal cancer risk: a nested case-control study using food diaries. J Natl Cancer Inst 102(9):614–26. https://doi.org/10.1093/jnci/djq092
De Filippo C, Cavalieri D, Di Paola M et al (2010) Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci USA 107(33):14691–6. https://doi.org/10.1073/pnas.1005963107
Dejea CM, Fathi P, Craig JM (2018) Patients with familial adenomatous polyposis harbor colonic biofilms containing tumorigenic bacteria. Science 359(6375):592–97. https://doi.org/10.1126/science.aah3648
Deng X, Gao F, Li N et al (2019) Antitumor activity of NKG2D CAR-T cells against human colorectal cancer cells in vitro and in vivo. Am J Cancer Res 9(5):945–958
DeStefano Shields CE, Van Meerbeke SW, Housseau F et al (2016) Reduction of murine colon tumorigenesis driven by enterotoxigenic bacteroides fragilis using cefoxitin treatment. J Infect Dis 214(1):122–9. https://doi.org/10.1093/infdis/jiw069
Dutta R, Khalil R, Mayilsamy K et al (2021) Combination therapy of mithramycin A and immune checkpoint inhibitor for the treatment of colorectal cancer in an orthotopic murine model. Front Immunolo 12:706133. https://doi.org/10.3389/fimmu.2021.706133
Fesnak AD, June CH, Levine BL (2016) Engineered T cells: the promise and challenges of cancer immunotherapy. Nat Rev Cancer 16(9):566–81. https://doi.org/10.1038/nrc.2016.97
Fong W, Li Q, Yu J (2020) Gut microbiota modulation: a novel strategy for prevention and treatment of colorectal cancer. Oncogene 39(26):4925–43. https://doi.org/10.1038/s41388-020-1341-1
Frankel AE, Coughlin LA, Kim J et al (2017) Metagenomic shotgun sequencing and unbiased metabolomic profiling identify specific human Gut microbiota and metabolites associated with immune checkpoint therapy efficacy in melanoma patients. Neoplasia 19(10):848–55. https://doi.org/10.1016/j.neo.2017.08.004
Ganesh K, Stadler ZK, Cercek A et al (2019) Immunotherapy in colorectal cancer: rationale, challenges and potential. Nat Rev Gastroenterol Hepatol 16(6):361–75. https://doi.org/10.1038/s41575-019-0126-x
Geller LT, Barzily-Rokni M (2017) Potential role of intratumor bacteria in mediating tumor resistance to the chemotherapeutic drug gemcitabine. Science 357(6356):1156–60. https://doi.org/10.1126/science.aah5043
Genaro SC, de Souza Lima, Reis LS, Reis SK et al (2019) Probiotic supplementation attenuates the aggressiveness of chemically induced colorectal tumor in rats. Life Sci 237:116895. https://doi.org/10.1016/j.lfs.2019.116895
Ghadimi D, Helwig U, Schrezenmeir J et al (2012) Epigenetic imprinting by commensal probiotics inhibits the IL-23/IL-17 axis in an in vitro model of the intestinal mucosal immune system. J Leukocyte Biol 92(4):895–911. https://doi.org/10.1189/jlb.0611286
Gilsing AM, Fransen F, de Kok TM et al (2013) Dietary heme iron and the risk of colorectal cancer with specific mutations in KRAS and APC. Carcinogenesis 34(12):2757–66. https://doi.org/10.1093/carcin/bgt290
Gross G, Waks T, Eshhar Z (1989) Expression of immunoglobulin-T-cell receptor chimeric molecules as functional receptors with antibody-type specificity. Proc Natl Acad Sci USA 86(24):10024–8. https://doi.org/10.1073/pnas.86.24.10024
Guedan S, Calderon H, Posey AD Jr et al (2019) Engineering and design of chimeric antigen receptors. Mol Therapy Methods Clin Dev 12:145–56. https://doi.org/10.1016/j.omtm.2018.12.009
Hamada T, Cao Y, Qian ZR et al (2017) Aspirin use and colorectal cancer survival according to tumor CD274 (programmed cell death 1 Ligand 1) expression status. J Clin Oncol 35(16):1836–44. https://doi.org/10.1200/jco.2016.70.7547
Han ZW, Lyv ZW, Cui B et al (2020) The old CEACAMs find their new role in tumor immunotherapy. Investig New Drugs 38(6):1888–98. https://doi.org/10.1007/s10637-020-00955-w
Hang S, Paik D, Yao L et al (2019) Bile acid metabolites control T(H)17 and T(reg) cell differentiation. Nature 576(7785):143–48. https://doi.org/10.1038/s41586-019-1785-z
He B, Hoang TK, Wang T (2017) Resetting microbiota by Lactobacillus reuteri inhibits T reg deficiency-induced autoimmunity via adenosine A2A receptors. J Exp Med 214(1):107–23. https://doi.org/10.1084/jem.20160961
Herrera FG, Bourhis J, Coukos G (2017) Radiotherapy combination opportunities leveraging immunity for the next oncology practice. CA Cancer J Clinic 67(1):65–85. https://doi.org/10.3322/caac.21358
Huang XZ, Gao P, Song YX et al (2019) Antibiotic use and the efficacy of immune checkpoint inhibitors in cancer patients: a pooled analysis of 2740 cancer patients. Oncoimmunology 8(12):e1665973. https://doi.org/10.1080/2162402x.2019.1665973
Hughes LA, Williamson EJ, van Engeland M et al (2012) Body size and risk for colorectal cancers showing BRAF mutations or microsatellite instability: a pooled analysis. Int J Epidemiol 41(4):1060–72. https://doi.org/10.1093/ije/dys055
Hughes LAE, Simons C, van den Brandt PA et al (2017) Lifestyle, diet, and colorectal cancer risk according to (Epi)genetic instability: current evidence and future directions of molecular pathological epidemiology. Curr Colorect Cancer Rep 13(6):455–69. https://doi.org/10.1007/s11888-017-0395-0
Huycke MM, Abrams V, Moore DR (2002) Enterococcus faecalis produces extracellular superoxide and hydrogen peroxide that damages colonic epithelial cell DNA. Carcinogenesis 23(3):529–36. https://doi.org/10.1093/carcin/23.3.529
Ijssennagger N, Belzer C, Hooiveld GJ (2015) Gut microbiota facilitates dietary heme-induced epithelial hyperproliferation by opening the mucus barrier in colon. Proc Natl Acad Sci 112(32):10038–43. https://doi.org/10.1073/pnas.1507645112
Inamura K (2018) Colorectal cancers: an update on their molecular pathology. Cancers. https://doi.org/10.3390/cancers10010026
Jan G, Belzacq AS, Haouzi D et al (2002) Propionibacteria induce apoptosis of colorectal carcinoma cells via short-chain fatty acids acting on mitochondria. Cell Death Differ 9(2):179–88. https://doi.org/10.1038/sj.cdd.4400935
Jayasekara H, MacInnis RJ, Williamson EJ et al (2017) Lifetime alcohol intake is associated with an increased risk of KRAS+ and BRAF-/KRAS− but not BRAF+ colorectal cancer. Int J Cancer 140(7):1485–93. https://doi.org/10.1002/ijc.30568
Jia M, Gao X, Zhang Y et al (2016) Different definitions of CpG island methylator phenotype and outcomes of colorectal cancer: a systematic review. Clin Epigenet 8:25. https://doi.org/10.1186/s13148-016-0191-8
Johdi NA, Sukor NF (2020) Colorectal cancer immunotherapy: options and strategies. Front Immunol 11:1624. https://doi.org/10.3389/fimmu.2020.01624
Kalathur M, Toso A, Chen J et al (2015) A chemogenomic screening identifies CK2 as a target for pro-senescence therapy in PTEN-deficient tumours. Nat Commun 6:7227. https://doi.org/10.1038/ncomms8227
Kaushik M, Kumar S, Kapoor RK et al (2019) Integrons and antibiotic resistance genes in water-borne pathogens: threat detection and risk assessment. J Med Microbiol 68(5):679–92. https://doi.org/10.1099/jmm.0.000972
Klampatsa A, Dimou V (2021) Mesothelin-targeted CAR-T cell therapy for solid tumors. Expert Opin Biol Therapy 21(4):473–86. https://doi.org/10.1080/14712598.2021.1843628
Koliarakis I, Psaroulaki A, Nikolouzakis TK et al (2018) Intestinal microbiota and colorectal cancer: a new aspect of research. J BUON 23(5):1216–1234
Kostic AD, Gevers D, Pedamallu CS et al (2012) Genomic analysis identifies association of Fusobacterium with colorectal carcinoma. Genome Res 22(2):292–8. https://doi.org/10.1101/gr.126573.111
Kostic AD, Howitt MR, Garrett WS (2013) Exploring host-microbiota interactions in animal models and humans. Genes Dev 27(7):701–18. https://doi.org/10.1101/gad.212522.112
Lasek W, Janyst M, Wolny R et al (2015) Immunomodulatory effects of inosine pranobex on cytokine production by human lymphocytes. Acta pharmaceutica (Zagreb, Croatia) 65(2):171–80. https://doi.org/10.1515/acph-2015-0015
Laws M, Shaaban A, Rahman KM (2019) Antibiotic resistance breakers: current approaches and future directions. FEMS Microbiol Rev 43(5):490–516. https://doi.org/10.1093/femsre/fuz014
Le Leu RK, Brown IL, Hu Y et al (2003) Effect of resistant starch on genotoxin-induced apoptosis, colonic epithelium, and lumenal contents in rats. Carcinogenesis 24(8):1347–52. https://doi.org/10.1093/carcin/bgg098
Li CJ, Elsasser TH (2005) Butyrate-induced apoptosis and cell cycle arrest in bovine kidney epithelial cells: involvement of caspase and proteasome pathways. J Anim Sci 83(1):89–97. https://doi.org/10.2527/2005.83189x
Li J, Jie HB, Lei Y et al (2015) PD-1/SHP-2 inhibits Tc1/Th1 phenotypic responses and the activation of T cells in the tumor microenvironment. Cancer Res 75(3):508–18. https://doi.org/10.1158/0008-5472.can-14-1215
Li S, Peppelenbosch MP, Smits R (2019) Bacterial biofilms as a potential contributor to mucinous colorectal cancer formation. Biochimica et biophysica acta Rev Cancer 1872(1):74–79. https://doi.org/10.1016/j.bbcan.2019.05.009
Li H, Yang C, Cheng H et al (2021) CAR-T cells for colorectal cancer: target-selection and strategies for improved activity and safety. J Cancer 12(6):1804–14. https://doi.org/10.7150/jca.50509
Lioux T, Mauny MA, Lamoureux A et al (2016) Design, synthesis, and biological evaluation of novel cyclic adenosine-inosine monophosphate (cAIMP) analogs that activate stimulator of interferon genes (STING). J Med Chem 59(22):10253–67. https://doi.org/10.1021/acs.jmedchem.6b01300
Long X, Wong CC, Tong L et al (2019) Peptostreptococcus anaerobius promotes colorectal carcinogenesis and modulates tumour immunity. Nat Microbiol 4(12):2319–30. https://doi.org/10.1038/s41564-019-0541-3
Lu R, Wu S, Liu X et al (2010) Chronic effects of a Salmonella type III secretion effector protein AvrA in vivo. PloS one 5(5):e10505. https://doi.org/10.1371/journal.pone.0010505
Ma W, Mao Q, Xia W et al (2019) Gut microbiota shapes the efficiency of cancer therapy. Front Microbiol 10:1050. https://doi.org/10.3389/fmicb.2019.01050
Magee MS, Abraham TS, Baybutt TR et al (2018) Human GUCY2C-targeted chimeric antigen receptor (CAR)-expressing T cells eliminate colorectal cancer metastases. Cancer Immunol Res 6(5):509–16. https://doi.org/10.1158/2326-6066.cir-16-0362
Mager LF, Burkhard R (2020) Microbiome-derived inosine modulates response to checkpoint inhibitor immunotherapy. Science 369(6510):1481–89. https://doi.org/10.1126/science.abc3421
Maier L, Goemans CV (2021) Unravelling the collateral damage of antibiotics on gut bacteria. Nature 599(7883):120–24. https://doi.org/10.1038/s41586-021-03986-2
Makaremi S, Asadzadeh Z, Hemmat N (2021) Immune checkpoint inhibitors in colorectal cancer: challenges and future prospects. Biomedicines. https://doi.org/10.3390/biomedicines9091075
Markowiak P, Śliżewska K (2017) Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients. https://doi.org/10.3390/nu9091021
Masteller EL, Chuang E, Mullen AC et al (2000) Structural analysis of CTLA-4 function in vivo. J Immunol (Baltimore, Md: 1950) 164(10):5319–27. https://doi.org/10.4049/jimmunol.164.10.5319
McFarland LV (2010) Systematic review and meta-analysis of Saccharomyces boulardii in adult patients. World J Gastroenterol 16(18):2202–22. https://doi.org/10.3748/wjg.v16.i18.2202
Meira LB, Bugni JM, Green SL et al (2008) DNA damage induced by chronic inflammation contributes to colon carcinogenesis in mice. J Clin Investig 118(7):2516–25. https://doi.org/10.1172/jci35073
Michot JM, Bigenwald C, Champiat S et al (2016) Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Eur J Cancer (Oxford, England: 1990) 54:139–48. https://doi.org/10.1016/j.ejca.2015.11.016
Mills JP, Rao K, Young VB (2018) Probiotics for prevention of Clostridium difficile infection. Curr Opin Gastroenterol 34(1):3–10. https://doi.org/10.1097/mog.0000000000000410
Mima K, Kosumi K, Baba Y et al (2021) The microbiome, genetics, and gastrointestinal neoplasms: the evolving field of molecular pathological epidemiology to analyze the tumor-immune-microbiome interaction. Human Genet 140(5):725–46. https://doi.org/10.1007/s00439-020-02235-2
Montalban-Arques A, Scharl M (2019) Intestinal microbiota and colorectal carcinoma: implications for pathogenesis, diagnosis, and therapy. EBioMedicine 48:648–55. https://doi.org/10.1016/j.ebiom.2019.09.050
Müller MF, Ibrahim AE, Arends MJ (2016) Molecular pathological classification of colorectal cancer. Virchows Archiv 469(2):125–34. https://doi.org/10.1007/s00428-016-1956-3
Myers G (2018) Immune-related adverse events of immune checkpoint inhibitors: a brief review. Curr Oncol (Toronto, Ont) 25(5):342–47. https://doi.org/10.3747/co.25.4235
Nabavinia MS, Gholoobi A, Charbgoo F et al (2017) Anti-MUC1 aptamer: a potential opportunity for cancer treatment. Med Res Rev 37(6):1518–39. https://doi.org/10.1002/med.21462
Nithianantham S, Xu M, Yamada M et al (2009) Crystal structure of FadA adhesin from Fusobacterium nucleatum reveals a novel oligomerization motif, the leucine chain. J Biol Chem 284(6):3865–72. https://doi.org/10.1074/jbc.M805503200
Ogino S, Nowak JA, Hamada T et al (2019) Insights into pathogenic interactions among environment, host, and tumor at the crossroads of molecular pathology and epidemiology. Annu Rev Pathol 14:83–103. https://doi.org/10.1146/annurev-pathmechdis-012418-012818
Omar Al-Hassi H, Ng O, Brookes M (2018) Tumour-associated and non-tumour-associated microbiota in colorectal cancer. Gut 67(2):395. https://doi.org/10.1136/gutjnl-2017-314219
Ooki A, Shinozaki E, Yamaguchi K (2021) Immunotherapy in colorectal cancer: current and future strategies. J Anus, Rectum Colon 5(1):11–24. https://doi.org/10.23922/jarc.2020-064
Paliany AS, Ho GF, Wan Ishak WZ et al (2018). Asia-Pacific J Clin Oncol. https://doi.org/10.1111/ajco.12758
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12(4):252–64. https://doi.org/10.1038/nrc3239
Paulos CM, Wrzesinski C, Kaiser A et al (2007) Microbial translocation augments the function of adoptively transferred self/tumor-specific CD8+ T cells via TLR4 signaling. J Clin Investig 117(8):2197–204. https://doi.org/10.1172/jci32205
Phua LC, Chue XP, Koh PK et al (2014) Non-invasive fecal metabonomic detection of colorectal cancer. Cancer Biol Therapy 15(4):389–97. https://doi.org/10.4161/cbt.27625
Piewngam P, Zheng Y, Nguyen TH et al (2018) Pathogen elimination by probiotic Bacillus via signalling interference. Nature 562(7728):532–37. https://doi.org/10.1038/s41586-018-0616-y
Poole K (2007) Efflux pumps as antimicrobial resistance mechanisms. Annals Med 39(3):162–76. https://doi.org/10.1080/07853890701195262
Purcell RV, Visnovska M, Biggs PJ et al (2017) Distinct gut microbiome patterns associate with consensus molecular subtypes of colorectal cancer. Sci Rep 7(1):11590. https://doi.org/10.1038/s41598-017-11237-6
Ray D, Kidane D (2016) Gut microbiota imbalance and base excision repair dynamics in colon cancer. J Cancer 7(11):1421–30. https://doi.org/10.7150/jca.15480
Reis Ferreira M, Andreyev HJN, Mohammed K et al (2019) Microbiota- and radiotherapy-induced gastrointestinal side-effects (MARS) study: a large pilot study of the microbiome in acute and late-radiation enteropathy. Clin Cancer Res 25(21):6487–500. https://doi.org/10.1158/1078-0432.ccr-19-0960
Robak OH, Heimesaat MM, Kruglov AA et al (2018) Antibiotic treatment-induced secondary IgA deficiency enhances susceptibility to Pseudomonas aeruginosa pneumonia. J Clin Investig 128(8):3535–45. https://doi.org/10.1172/jci97065
Rosshart SP, Vassallo BG, Angeletti D et al (2017) Wild mouse gut microbiota promotes host fitness and improves disease resistance. Cell 171(5):1015–28.e13. https://doi.org/10.1016/j.cell.2017.09.016
Rothhammer V, Quintana FJ (2019) The aryl hydrocarbon receptor: an environmental sensor integrating immune responses in health and disease. Nat Rev Immunol 19(3):184–97. https://doi.org/10.1038/s41577-019-0125-8
Routy B, Gopalakrishnan V, Daillère R et al (2018) The gut microbiota influences anticancer immunosurveillance and general health. Nat Rev Clin Oncol 15(6):382–96. https://doi.org/10.1038/s41571-018-0006-2
Sahin IH, Akce M, Alese O et al (2019) Immune checkpoint inhibitors for the treatment of MSI-H/MMR-D colorectal cancer and a perspective on resistance mechanisms. Br J Cancer 121(10):809–18. https://doi.org/10.1038/s41416-019-0599-y
Salerno F, Freen-van Heeren JJ (2019) Costimulation through TLR2 drives polyfunctional CD8(+) T cell responses. J Immunol 202(3):714–23. https://doi.org/10.4049/jimmunol.1801026
Schubert ML, Rohrbach R, Schmitt M et al (2021) The potential role of the intestinal micromilieu and individual microbes in the immunobiology of chimeric antigen receptor T-cell therapy. Front Immunol 12:670286. https://doi.org/10.3389/fimmu.2021.670286
Shah MS, DeSantis TZ, Weinmaier T et al (2018) Leveraging sequence-based faecal microbial community survey data to identify a composite biomarker for colorectal cancer. Gut 67(5):882–91. https://doi.org/10.1136/gutjnl-2016-313189
Sharma P, Allison JP (2015) The future of immune checkpoint therapy. Science (New York, NY) 348(6230):56–61. https://doi.org/10.1126/science.aaa8172
Shen J, Li P, Liu S et al (2020) The chemopreventive effects of Huangqin-tea against AOM-induced preneoplastic colonic aberrant crypt foci in rats and omics analysis. Food Funct 11(11):9634–50. https://doi.org/10.1039/d0fo01731k
Shiao SL, Kershaw KM, Limon JJ et al (2021) Commensal bacteria and fungi differentially regulate tumor responses to radiation therapy. Cancer Cell 39(9):1202–13.e6. https://doi.org/10.1016/j.ccell.2021.07.002
Shimizu T, Tolcher AW, Papadopoulos KP et al (2012) The clinical effect of the dual-targeting strategy involving PI3K/AKT/mTOR and RAS/MEK/ERK pathways in patients with advanced cancer. Clin Cancer Res 18(8):2316–25. https://doi.org/10.1158/1078-0432.ccr-11-2381
Shindo Y, Yoshimura K, Kuramasu A et al (2015) Combination immunotherapy with 4–1BB activation and PD-1 blockade enhances antitumor efficacy in a mouse model of subcutaneous tumor. Anticancer Res 35(1):129–136
Siena S, Sartore-Bianchi A, Marsoni S et al (2018) Targeting the human epidermal growth factor receptor 2 (HER2) oncogene in colorectal cancer. Annals Oncol 29(5):1108–19. https://doi.org/10.1093/annonc/mdy100
Simin J, Fornes R, Liu Q et al (2020) Antibiotic use and risk of colorectal cancer: a systematic review and dose-response meta-analysis. Br J Cancer 123(12):1825–32. https://doi.org/10.1038/s41416-020-01082-2
Singhi SC, Kumar S (2016) Probiotics in critically ill children. Research. https://doi.org/10.12688/f1000research.7630.1
Sivan A, Corrales L, Hubert N et al (2015) Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science (New York, NY) 350(6264):1084–9. https://doi.org/10.1126/science.aac4255
Soltan Dallal MM, Davoodabadi A, Abdi M et al (2017) Inhibitory effect of Lactobacillus plantarum and Lb. fermentum isolated from the faeces of healthy infants against nonfermentative bacteria causing nosocomial infections. New Microbes New Infect 15:9–13. https://doi.org/10.1016/j.nmni.2016.09.003
Song M, Nishihara R, Wu K et al (2015) Marine ω-3 polyunsaturated fatty acids and risk of colorectal cancer according to microsatellite instability. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djv007
Song W, Tiruthani K, Wang Y et al (2018) Trapping of lipopolysaccharide to promote immunotherapy against colorectal cancer and attenuate liver metastasis. Adv Mater (Deerfield Beach, Fla) 30(52):e1805007. https://doi.org/10.1002/adma.201805007
Spranger S, Koblish HK, Horton B et al (2014) Mechanism of tumor rejection with doublets of CTLA-4, PD-1/PD-L1, or IDO blockade involves restored IL-2 production and proliferation of CD8(+) T cells directly within the tumor microenvironment. J Immunother Cancer 2:3. https://doi.org/10.1186/2051-1426-2-3
Sun S, Luo L (2020) Bifidobacterium alters the gut microbiota and modulates the functional metabolism of T regulatory cells in the context of immune checkpoint blockade. Proc Natl Acade Sci 117(44):27509–15. https://doi.org/10.1073/pnas.1921223117
Taieb F, Nougayrède JP, Oswald E (2011) Cycle inhibiting factors (cifs): cyclomodulins that usurp the ubiquitin-dependent degradation pathway of host cells. Toxins 3(4):356–68. https://doi.org/10.3390/toxins3040356
Taper HS, Roberfroid MB (2005) Possible adjuvant cancer therapy by two prebiotics–inulin or oligofructose. In Vivo (Athens, Greece) 19(1):201–204
Tilg H, Adolph TE, Gerner RR et al (2018) The Intestinal Microbiota in Colorectal Cancer. Cancer Cell 33(6):954–64. https://doi.org/10.1016/j.ccell.2018.03.004
Tinsley N, Zhou C, Tan G et al (2020) Cumulative antibiotic use significantly decreases efficacy of checkpoint inhibitors in patients with advanced cancer. Oncologist 25(1):55–63. https://doi.org/10.1634/theoncologist.2019-0160
Tjalsma H, Boleij A, Marchesi JR et al (2012) A bacterial driver-passenger model for colorectal cancer: beyond the usual suspects. Nat Rev Microbiol 10(8):575–82. https://doi.org/10.1038/nrmicro2819
Toumazi D, Constantinou C (2020) A fragile balance: the important role of the intestinal microbiota in the prevention and management of colorectal cancer. Oncology 98(9):593–602. https://doi.org/10.1159/000507959
Triner D, Devenport SN, Ramakrishnan SK et al (2019) Neutrophils restrict tumor-associated microbiota to reduce growth and invasion of colon tumors in mice. Gastroenterology 156(5):1467–82. https://doi.org/10.1053/j.gastro.2018.12.003
Tsai HF, Hsu PN (2017) Cancer immunotherapy by targeting immune checkpoints: mechanism of T cell dysfunction in cancer immunity and new therapeutic targets. J Biomed Sci 24(1):35. https://doi.org/10.1186/s12929-017-0341-0
Tsoi H, Chu ESH, Zhang X et al (2017) Peptostreptococcus anaerobius induces intracellular cholesterol biosynthesis in colon cells to induce proliferation and causes dysplasia in mice. Gastroenterology 152(6):1419–33.e5. https://doi.org/10.1053/j.gastro.2017.01.009
Ueda K, Yonekura S, Ogasawara N et al (2019) The impact of antibiotics on prognosis of metastatic renal cell carcinoma in Japanese patients treated with immune checkpoint inhibitors. Anticancer Res 39(11):6265–71. https://doi.org/10.21873/anticanres.13836
Uribe-Herranz M, Bittinger K, Rafail S et al (2018) Gut microbiota modulates adoptive cell therapy via CD8α dendritic cells and IL-12. JCI Insight. https://doi.org/10.1172/jci.insight.94952
Vétizou M, Pitt JM, Daillère R et al (2015) Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science (New York, NY) 350(6264):1079–84. https://doi.org/10.1126/science.aad1329
Veziant J, Gagnière J, Jouberton E et al (2016) Association of colorectal cancer with pathogenic Escherichia coli: Focus on mechanisms using optical imaging. World J Clin Oncol 7(3):293–301. https://doi.org/10.5306/wjco.v7.i3.293
Villéger R, Lopès A, Veziant J et al (2018) Microbial markers in colorectal cancer detection and/or prognosis. World J Gastroenterol 24(22):2327–47. https://doi.org/10.3748/wjg.v24.i22.2327
Wang X, Allen TD, May RJ et al (2008) Enterococcus faecalis induces aneuploidy and tetraploidy in colonic epithelial cells through a bystander effect. Cancer Res 68(23):9909–17. https://doi.org/10.1158/0008-5472.can-08-1551
Wang Y, Wu Y, Wang Y et al (2017) Antioxidant properties of probiotic bacteria. Nutrients. https://doi.org/10.3390/nu9050521
Wang F, Yin Q, Chen L et al (2018) Bifidobacterium can mitigate intestinal immunopathology in the context of CTLA-4 blockade. Proc Natl Acad Sci USA 115(1):157–61. https://doi.org/10.1073/pnas.1712901115
Wei Z, Cao S, Liu S et al (2016) Could gut microbiota serve as prognostic biomarker associated with colorectal cancer patients’ survival? A pilot study on relevant mechanism. Oncotarget 7(29):46158–72. https://doi.org/10.18632/oncotarget.10064
Wu S, Feng B, Li K et al (2012) Fish consumption and colorectal cancer risk in humans: a systematic review and meta-analysis. Am J Med 125(6):551–9.e5. https://doi.org/10.1016/j.amjmed.2012.01.022
Xie J, Han X, Zhao C et al (2019) Phosphotyrosine-dependent interaction between the kinases PKCθ and Zap70 promotes proximal TCR signaling. Sci Signal. https://doi.org/10.1126/scisignal.aar3349
Yan HHN, Lai JCW, Ho SL et al (2017) RNF43 germline and somatic mutation in serrated neoplasia pathway and its association with BRAF mutation. Gut 66(9):1645–56. https://doi.org/10.1136/gutjnl-2016-311849
Yang J, Yu J (2018) The association of diet, gut microbiota and colorectal cancer: what we eat may imply what we get. Protein Cell 9(5):474–87. https://doi.org/10.1007/s13238-018-0543-6
Yang Y, Weng W, Peng J et al (2017) Fusobacterium nucleatum increases proliferation of colorectal cancer cells and tumor development in mice by activating toll-like receptor 4 signaling to nuclear factor-κB, and up-regulating expression of microRNA-21. Gastroenterology 152(4):851–66.e24. https://doi.org/10.1053/j.gastro.2016.11.018
Ye X, Wang R, Bhattacharya R et al (2017) Fusobacterium nucleatum subspecies animalis influences proinflammatory cytokine expression and monocyte activation in human colorectal tumors. Cancer Prevent Res (Philadelphia, Pa) 10(7):398–409. https://doi.org/10.1158/1940-6207.capr-16-0178
Yu T, Guo F, Yu Y et al (2017) Fusobacterium nucleatum promotes chemoresistance to colorectal cancer by modulating autophagy. Cell 170(3):548–63.e16. https://doi.org/10.1016/j.cell.2017.07.008
Zabawa TP, Pucci MJ, Parr TR Jr et al (2016) Treatment of Gram-negative bacterial infections by potentiation of antibiotics. Curr Opin Microbiol 33:7–12. https://doi.org/10.1016/j.mib.2016.05.005
Zackular JP, Baxter NT, Iverson KD et al (2013) The gut microbiome modulates colon tumorigenesis. MBio 4(6):e00692-13. https://doi.org/10.1128/mBio.00692-13
Zhang C, Wang Z, Yang Z et al (2017) Phase I Escalating-Dose Trial of CAR-T Therapy Targeting CEA(+) Metastatic Colorectal Cancers. Mol Therapy 25(5):1248–58. https://doi.org/10.1016/j.ymthe.2017.03.010
Zhang F, Cui B, He X et al (2018) Microbiota transplantation: concept, methodology and strategy for its modernization. Protein Cell 9(5):462–73. https://doi.org/10.1007/s13238-018-0541-8
Zhang S, Wang Q, Zhou C et al (2019) Colorectal cancer, radiotherapy and gut microbiota. Chin J Cancer Res 31(1):212–22. https://doi.org/10.21147/j.issn.1000-9604.2019.01.16
Zhang J, Dai Z, Yan C et al (2021) A new biological triangle in cancer: intestinal microbiota, immune checkpoint inhibitors and antibiotics. Clin Transl Oncol 23(12):2415–30. https://doi.org/10.1007/s12094-021-02659-w
Zhuo Q, Yu B, Zhou J et al (2019) Lysates of Lactobacillus acidophilus combined with CTLA-4-blocking antibodies enhance antitumor immunity in a mouse colon cancer model. Sci Rep 9(1):20128. https://doi.org/10.1038/s41598-019-56661-y
Żółkiewicz J, Marzec A, Ruszczyński M (2020) Postbiotics-a step beyond pre- and probiotics. Nutrients. https://doi.org/10.3390/nu12082189
Funding
This work was supported by grants from the Training Project of Key Talents of Youth Medicine in Jiangsu province, China [No. QNRC2016330], the Graduate Research- Innovation Project in Jiangsu province [No. SJCX21_1644], the Social Development-Health Care Project of Yangzhou, Jiangsu Province [No. YZ2018087], the Social Development-Health Care Project of Yangzhou, Jiangsu Province [No. YZ2021075], and High-level talent “six one projects” top talent scientific research project of Jiangsu Province [No. LGY2019034]. The funding bodies had no role in the design of the study; in the collection, analysis, and interpretation of the data; and in writing the manuscript.
Author information
Authors and Affiliations
Contributions
WZ and TL drafted the manuscript in detail. WZ and JZ researched the literatures and drew figures. JX and HZ counted and plotted the diagram and table. DT and DW critically revised the article for important intellectual content. All authors read and approved the final manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that there is no conflict of interest.
Ethical Approval and Consent to participate
Not applicable
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, W., Zhang, J., Liu, T. et al. Bidirectional effects of intestinal microbiota and antibiotics: a new strategy for colorectal cancer treatment and prevention. J Cancer Res Clin Oncol 148, 2387–2404 (2022). https://doi.org/10.1007/s00432-022-04081-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04081-3