Abstract
Background
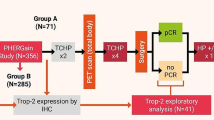
Trophoblast cell-surface antigen 2 (TROP2) is expressed on the surface of trophoblast cells and many malignant tumor cells. However, data on TROP2 expression in advanced lung cancer are insufficient, and its changes have not been fully evaluated.
Methods
We assessed the prevalence and changes in TROP2 expression in patients with lung cancer who received anti-cancer treatments using immunohistochemical (IHC) analysis with an anti-TROP2 antibody (clone: SP295). IHC scores were graded from 0 to 3; grade ≥ 2 was considered positive for TROP2 expression. We defined a difference in IHC score, before and after anti-cancer treatments, as the change in TROP2 expression.
Results
Before anti-cancer treatment, TROP2 expression was observed in 89% (143/160) of the patients and was significantly more common in adenocarcinoma and squamous cell carcinoma than in neuroendocrine carcinoma (P < 0.001). After anti-cancer treatment, TROP2 expression was observed in 87% (139/160) of the patients. The distribution of TROP2 expression in post-treatment samples was analogous to that in pre-treatment samples when compared using the Wilcoxon signed-rank test (P = 0.509). However, an increase in TROP2 expression was seen in 19 (12%), and a decrease in 20 (13%) patients. Patients treated with targeted therapy showed significantly higher changes in TROP2 expression (P = 0.019) and thoracic radiotherapy was more likely to increase TROP2 expression than chemotherapy alone.
Conclusion
Although some anti-cancer treatments might alter the TROP2 expression, TROP2 was expressed in most lung cancer specimens before and after anti-cancer treatments. These results support the development of TROP2-directed therapy against advanced lung cancer in various treatment lines.



Similar content being viewed by others
Availability of data and materials
The remaining data that support the findings of this study are available from the corresponding author upon reasonable request. The data are not publicly available due to privacy or ethical restrictions.
Code availability
Not applicable.
References
Bardia A et al (2017) Efficacy and safety of anti-trop-2 antibody drug conjugate Sacituzumab Govitecan (IMMU-132) in heavily pretreated patients with metastatic triple-negative breast cancer. J Clin Oncol 35:2141–2148. https://doi.org/10.1200/JCO.2016.70.8297
Bardia A et al (2019) Sacituzumab Govitecan-hziy in refractory metastatic triple-negative breast cancer. N Engl J Med 380:741–751. https://doi.org/10.1056/NEJMoa1814213
Bartley AN et al (2017) HER2 testing and clinical decision making in gastroesophageal adenocarcinoma: guideline from the College of American Pathologists, American Society for Clinical Pathology, and the American Society of Clinical Oncology. J Clin Oncol 35:446–464. https://doi.org/10.1200/JCO.2016.69.4836
Boutsikou E, Domvri K, Hardavella G, Tsiouda D, Zarogoulidis K, Kontakiotis T (2018) Tumour necrosis factor, interferon-gamma and interleukins as predictive markers of antiprogrammed cell-death protein-1 treatment in advanced non-small cell lung cancer: a pragmatic approach in clinical practice. Ther Adv Med Oncol 10:1758835918768238. https://doi.org/10.1177/1758835918768238
Cubas R, Li M, Chen C, Yao Q (2009) Trop2: a possible therapeutic target for late stage epithelial carcinomas. Biochim Biophys Acta 1796:309–314. https://doi.org/10.1016/j.bbcan.2009.08.001
Cubas R, Zhang S, Li M, Chen C, Yao Q (2010) Trop2 expression contributes to tumor pathogenesis by activating the ERK MAPK pathway. Mol Cancer 9:253. https://doi.org/10.1186/1476-4598-9-253
Fornaro M, Dell’Arciprete R, Stella M, Bucci C, Nutini M, Capri MG, Alberti S (1995) Cloning of the gene encoding Trop-2, a cell-surface glycoprotein expressed by human carcinomas. Int J Cancer 62:610–618. https://doi.org/10.1002/ijc.2910620520
Gray JE et al (2017) Therapy of small cell lung cancer (SCLC) with a Topoisomerase-I-inhibiting antibody-drug conjugate (ADC) targeting Trop-2, Sacituzumab Govitecan. Clin Cancer Res 23:5711–5719. https://doi.org/10.1158/1078-0432.CCR-17-0933
Gu QZ, Nijiati A, Gao X, Tao KL, Li CD, Fan XP, Tian Z (2018) TROP2 promotes cell proliferation and migration in osteosarcoma through PI3K/AKT signaling. Mol Med Rep 18:1782–1788. https://doi.org/10.3892/mmr.2018.9083
Heist RS et al (2017) Therapy of advanced non-small-cell lung cancer with an SN-38-Anti-Trop-2 drug conjugate, Sacituzumab Govitecan. J Clin Oncol 35:2790–2797. https://doi.org/10.1200/JCO.2016.72.1894
Inamura K et al (2017) Association of tumor Trop2 expression with prognosis varies among lung cancer subtypes. Oncotarget 8:28725–28735. https://doi.org/10.18632/oncotarget.15647
Jiang A, Gao X, Zhang D, Zhang L, Lu H (2013) Expression and clinical significance of the Trop-2 gene in advanced non-small cell lung carcinoma. Oncol Lett 6:375–380. https://doi.org/10.3892/ol.2013.1368
Kobayashi H et al (2010) Expression of the GA733 gene family and its relationship to prognosis in pulmonary adenocarcinoma. Virchows Arch 457:69–76. https://doi.org/10.1007/s00428-010-0930-8
Li X et al (2019) Correction: Trop2 promotes proliferation, migration and metastasis of gallbladder cancer cells by regulating PI3K/AKT pathway and inducing EMT. Oncotarget 10:6540. https://doi.org/10.18632/oncotarget.27299
Lipinski M, Parks DR, Rouse RV, Herzenberg LA (1981) Human trophoblast cell-surface antigens defined by monoclonal antibodies. Proc Natl Acad Sci USA 78:5147–5150. https://doi.org/10.1073/pnas.78.8.5147
Lisberg AE et al (2020) Dose escalation and expansion from the phase I study of DS-1062, a trophoblast cell-surface antigen 2 (TROP2) antibody drug conjugate (ADC), in patients (pts) with advanced non-small cell lung cancer (NSCLC). J Clin Oncol 38:9619–9619. https://doi.org/10.1200/JCO.2020.38.15_suppl.9619
Ogitani Y, Hagihara K, Oitate M, Naito H, Agatsuma T (2016) Bystander killing effect of DS-8201a, a novel anti-human epidermal growth factor receptor 2 antibody-drug conjugate, in tumors with human epidermal growth factor receptor 2 heterogeneity. Cancer Sci 107:1039–1046. https://doi.org/10.1111/cas.12966
Ohmachi T, Tanaka F, Mimori K, Inoue H, Yanaga K, Mori M (2006) Clinical significance of TROP2 expression in colorectal cancer. Clin Cancer Res 12:3057–3063. https://doi.org/10.1158/1078-0432.CCR-05-1961
Pak MG, Shin DH, Lee CH, Lee MK (2012) Significance of EpCAM and TROP2 expression in non-small cell lung cancer. World J Surg Oncol 10:53. https://doi.org/10.1186/1477-7819-10-53
Ripani E, Sacchetti A, Corda D, Alberti S (1998) Human Trop-2 is a tumor-associated calcium signal transducer. Int J Cancer 76:671–676. https://doi.org/10.1002/(sici)1097-0215(19980529)76:5%3c671::aid-ijc10%3e3.0.co;2-7
Sands JM et al (2019) First-in-human phase 1 study of DS-1062a in patients with advanced solid tumors. J Clin Oncol 37:9051–9051. https://doi.org/10.1200/JCO.2019.37.15_suppl.9051
Shvartsur A, Bonavida B (2015) Trop2 and its overexpression in cancers: regulation and clinical/therapeutic implications. Genes Cancer 6:84–105. https://doi.org/10.18632/genesandcancer.40
Stepan LP, Trueblood ES, Hale K, Babcook J, Borges L, Sutherland CL (2011) Expression of Trop2 cell surface glycoprotein in normal and tumor tissues: potential implications as a cancer therapeutic target. J Histochem Cytochem 59:701–710. https://doi.org/10.1369/0022155411410430
Wu H et al (2013) Potential therapeutic target and independent prognostic marker of TROP2 in laryngeal squamous cell carcinoma. Head Neck 35:1373–1378. https://doi.org/10.1002/hed.23138
Xu P et al (2017) Prognostic role and clinical significance of trophoblast cell surface antigen 2 in various carcinomas. Cancer Manag Res 9:821–837. https://doi.org/10.2147/CMAR.S147033
Zeng P, Chen MB, Zhou LN, Tang M, Liu CY, Lu PH (2016) Impact of TROP2 expression on prognosis in solid tumors: a systematic review and meta-analysis. Sci Rep 6:33658. https://doi.org/10.1038/srep33658
Zhao P, Zhang Z (2018) TNF-alpha promotes colon cancer cell migration and invasion by upregulating TROP-2. Oncol Lett 15:3820–3827. https://doi.org/10.3892/ol.2018.7735
Zhao W et al (2018) Trop2 is a potential biomarker for the promotion of EMT in human breast cancer. Oncol Rep 40:759–766. https://doi.org/10.3892/or.2018.6496
Acknowledgments
We thank all the patients who participated in this study and their families. We also thank all the clinical pathology technicians who supported this study.
Funding
This study was supported by a contract research fund from Daiichi Sankyo Co., Ltd., Tokyo, Japan.
Author information
Authors and Affiliations
Contributions
SO was involved in study concept and design; acquisition, analysis, or interpretation of data; drafting of the manuscript; full access to all data in the study and responsible for the integrity of the data and accuracy of the data analysis. KM, TK, and TS were involved in pathological support. EM, TM, NM, TK, HK, KN, KW, AO, TN, HM were involved in material support. HK was involved in administrative, technical or material support. TT was involved in study supervision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest directly relevant or directly related to the content of this article.
Ethics approval
This study was approved by the Institutional Review Board of the Shizuoka Cancer Center, Japan (Approval No. T29-17-29-1; July 27, 2017).
Consent to participate
An opt-out method was used on the basis of the Ethical Guidelines for Epidemiological Research in Japan.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Omori, S., Muramatsu, K., Kawata, T. et al. Trophoblast cell-surface antigen 2 expression in lung cancer patients and the effects of anti-cancer treatments. J Cancer Res Clin Oncol 148, 2455–2463 (2022). https://doi.org/10.1007/s00432-021-03784-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03784-3