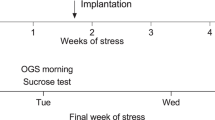
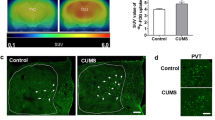
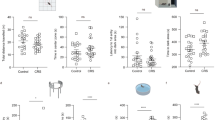
Abstract
A number of different receptors are distributed in glutamatergic neurons of the lateral habenula (LHb). These glutamatergic neurons are involved in different neural pathways, which may identify how the LHb regulates various physiological functions. However, the role of dopamine D1 receptor (D1R)-expressing habenular neurons projecting to the ventral tegmental area (VTA) (LHbD1R–VTA) remains not well understood. In the current study, to determine the activity of D1R-expressing neurons in LHb, D1R-Cre mice were used to establish the chronic restraint stress (CRS) depression model. Adeno-associated virus was injected into bilateral LHb in D1R-Cre mice to examine whether optogenetic activation of the LHb D1R-expressing neurons and their projections could induce depression-like behavior. Optical fibers were implanted in the LHb and VTA, respectively. To investigate whether optogenetic inhibition of the LHbD1R–VTA circuit could produce antidepressant-like effects, the adeno-associated virus was injected into the bilateral LHb in the D1R-Cre CRS model, and optical fibers were implanted in the bilateral VTA. The D1R-expressing neuronal activity in the LHb was increased in the CRS depression model. Optogenetic activation of the D1R-expressing neurons in LHb induced behavioral despair and anhedonia, which could also be induced by activation of the LHbD1R–VTA axons. Conversely, optogenetic inhibition of the LHbD1R–VTA circuit improved behavioral despair and anhedonia in the CRS depression model. D1R-expressing glutamatergic neurons in the LHb and their projections to the VTA are involved in the occurrence and regulation of depressive-like behavior.






Similar content being viewed by others
Data availability
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
References
Abi-Dargham A, Mawlawi O, Lombardo I, Gil R, Martinez D, Huang Y, Hwang DR, Keilp J, Kochan L, Van Heertum R, Gorman JM, Laruelle M (2002) Prefrontal dopamine d1 receptors and working memory in schizophrenia. J Neurosci 22:3708–3719
Aizawa H, Kobayashi M, Tanaka S, Fukai T, Okamoto H (2012) Molecular characterization of the subnuclei in rat habenula. J Comp Neurol 520:4051–4066
Baño-Otálora B, Piggins HD (2017) Contributions of the lateral habenula to circadian timekeeping. Pharmacol Biochem Behav 162:46–54
Bao W, Ding J, Jiang S, Yao Z, Qu W, Li W, Huang Z, Han Y (2023) Selective activation of nac d1r-vp/lh circuits promotes reanimation from sevoflurane anesthesia in mice. Anesth Analg 137:87–97
Bastide MF, de la Crompe B, Doudnikoff E, Fernagut PO, Gross CE, Mallet N, Boraud T, Bézard E (2016) Inhibiting lateral habenula improves l-dopa-induced dyskinesia. Biol Psychiatry 79:345–353
Brinschwitz K, Dittgen A, Madai VI, Lommel R, Geisler S, Veh RW (2010) Glutamatergic axons from the lateral habenula mainly terminate on gabaergic neurons of the ventral midbrain. Neuroscience 168:463–476
Brown PL, Palacorolla H, Brady D, Riegger K, Elmer GI, Shepard PD (2017) Habenula-induced inhibition of midbrain dopamine neurons is diminished by lesions of the rostromedial tegmental nucleus. J Neurosci 37:217–225
Cerniauskas I, Winterer J, de Jong JW, Lukacsovich D, Yang H, Khan F, Peck JR, Obayashi SK, Lilascharoen V, Lim BK, Földy C, Lammel S (2019) Chronic stress induces activity, synaptic, and transcriptional remodeling of the lateral habenula associated with deficits in motivated behaviors. Neuron 104:899-915.e898
Chan J, Guan X, Ni Y, Luo L, Yang L, Zhang P, Zhang J, Chen Y (2017) Dopamine d1-like receptor in lateral habenula nucleus affects contextual fear memory and long-term potentiation in hippocampal ca1 in rats. Behav Brain Res 321:61–68
Chan J, Ni Y, Zhang P, Zhang J, Chen Y (2017) D1-like dopamine receptor dysfunction in the lateral habenula nucleus increased anxiety-like behavior in rat. Neuroscience 340:542–550
Channabasavanna SM, Khanna S (1997) Therapy with amineptine, a dopamine reuptake inhibitor, in patients with major depression. Indian J Psychiatry 39:147–153
Christoph GR, Leonzio RJ, Wilcox KS (1986) Stimulation of the lateral habenula inhibits dopamine-containing neurons in the substantia nigra and ventral tegmental area of the rat. J Neurosci 6:613–619
Dai D, Li W, Chen A, Gao XF, Xiong L (2022) Lateral habenula and its potential roles in pain and related behaviors. ACS Chem Neurosci 13:1108–1118
Durieux L, Mathis V, Herbeaux K, Muller MA, Barbelivien A, Mathis C, Schlichter R, Hugel S, Majchrzak M, Lecourtier L (2020) Involvement of the lateral habenula in fear memory. Brain Struct Funct 225:2029–2044
Friedman A, Friedman Y, Dremencov E, Yadid G (2008) Vta dopamine neuron bursting is altered in an animal model of depression and corrected by desipramine. J Mol Neurosci 34:201–209
Gruber C, Kahl A, Lebenheim L, Kowski A, Dittgen A, Veh RW (2007) Dopaminergic projections from the VTA substantially contribute to the mesohabenular pathway in the rat. Neurosci Lett 427:165–170
Hare BD, Shinohara R, Liu RJ, Pothula S, DiLeone RJ, Duman RS (2019) Optogenetic stimulation of medial prefrontal cortex Drd1 neurons produces rapid and long-lasting antidepressant effects. Nat Commun 10:223
Hashikawa Y, Hashikawa K, Rossi MA, Basiri ML, Liu Y, Johnston NL, Ahmad OR, Stuber GD (2020) Transcriptional and spatial resolution of cell types in the mammalian habenula. Neuron 106:743-758.e745
Hong S, Jhou TC, Smith M, Saleem KS, Hikosaka O (2011) Negative reward signals from the lateral habenula to dopamine neurons are mediated by rostromedial tegmental nucleus in primates. J Neurosci 31:11457–11471
Ji H, Shepard PD (2007) Lateral habenula stimulation inhibits rat midbrain dopamine neurons through a gaba(a) receptor-mediated mechanism. J Neurosci 27:6923–6930
Kowski AB, Veh RW, Weiss T (2009) Dopaminergic activation excites rat lateral habenular neurons in vivo. Neuroscience 161:1154–1165
Lecca S, Trusel M, Mameli M (2017) Footshock-induced plasticity of gaba(b) signalling in the lateral habenula requires dopamine and glucocorticoid receptors. Synapse 71:e21984
Lemon N, Manahan-Vaughan D (2006) Dopamine d1/d5 receptors gate the acquisition of novel information through hippocampal long-term potentiation and long-term depression. J Neurosci 26:7723–7729
Li J, Fan R, Liu X, Shen X, Liu X, Zhao H (2021) The convergence of aversion and reward signals in individual neurons of the mice lateral habenula. Exp Neurol 339:113637
Li J, Li Y, Zhang B, Shen X, Zhao H (2016) Why depression and pain often coexist and mutually reinforce: Role of the lateral habenula. Exp Neurol 284:106–113
Li K, Zhou T, Liao L, Yang Z, Wong C, Henn F, Malinow R, Yates JR 3rd, Hu H (2013) Βcamkii in lateral habenula mediates core symptoms of depression. Science 341:1016–1020
Liu H, Rastogi A, Narain P, Xu Q, Sabanovic M, Alhammadi AD, Guo L, Cao JL, Zhang H, Aqel H, Mlambo V, Rezgui R, Radwan B, Chaudhury D (2021) Blunted diurnal firing in lateral habenula projections to dorsal raphe nucleus and delayed photoentrainment in stress-susceptible mice. PLoS Biol 19:e3000709
Ma Z, Zhong Y, Hines CS, Wu Y, Li Y, Pang M, Li J, Wang C, Fox PT, Zhang N, Wang C (2020) Identifying generalized anxiety disorder using resting state habenular circuitry. Brain Imaging Behav 14:1406–1418
McNab F, Varrone A, Farde L, Jucaite A, Bystritsky P, Forssberg H, Klingberg T (2009) Changes in cortical dopamine d1 receptor binding associated with cognitive training. Science 323:800–802
Morales M, Root DH (2014) Glutamate neurons within the midbrain dopamine regions. Neuroscience 282:60–68
Park H, Rhee J, Park K, Han JS, Malinow R, Chung C (2017) Exposure to stressors facilitates long-term synaptic potentiation in the lateral habenula. J Neurosci 37:6021–6030
Petzel A, Bernard R, Poller WC, Veh RW (2017) Anterior and posterior parts of the rat ventral tegmental area and the rostromedial tegmental nucleus receive topographically distinct afferents from the lateral habenular complex. J Comp Neurol 525:2310–2327
Shen X, Ruan X, Zhao H (2012) Stimulation of midbrain dopaminergic structures modifies firing rates of rat lateral habenula neurons. PLoS ONE 7:e34323
Shumake J, Edwards E, Gonzalez-Lima F (2003) Opposite metabolic changes in the habenula and ventral tegmental area of a genetic model of helpless behavior. Brain Res 963:274–281
Stamatakis AM, Jennings JH, Ung RL, Blair GA, Weinberg RJ, Neve RL, Boyce F, Mattis J, Ramakrishnan C, Deisseroth K, Stuber GD (2013) A unique population of ventral tegmental area neurons inhibits the lateral habenula to promote reward. Neuron 80:1039–1053
Stujenske JM, Spellman T, Gordon JA (2015) Modeling the spatiotemporal dynamics of light and heat propagation for in vivo optogenetics. Cell Rep 12:525–534
Stukalin Y, Lan A, Einat H (2020) Revisiting the validity of the mouse tail suspension test: systematic review and meta-analysis of the effects of prototypic antidepressants. Neurosci Biobehav Rev 112:39–47
Takahashi A, Durand-de Cuttoli R, Flanigan ME, Hasegawa E, Tsunematsu T, Aleyasin H, Cherasse Y, Miya K, Okada T, Keino-Masu K, Mitsui K, Li L, Patel V, Blitzer RD, Lazarus M, Tanaka KF, Yamanaka A, Sakurai T, Ogawa S, Russo SJ (2022) Lateral habenula glutamatergic neurons projecting to the dorsal raphe nucleus promote aggressive arousal in mice. Nat Commun 13:4039
Tye KM, Mirzabekov JJ, Warden MR, Ferenczi EA, Tsai HC, Finkelstein J, Kim SY, Adhikari A, Thompson KR, Andalman AS, Gunaydin LA, Witten IB, Deisseroth K (2013) Dopamine neurons modulate neural encoding and expression of depression-related behaviour. Nature 493:537–541
Wallace ML, Huang KW, Hochbaum D, Hyun M, Radeljic G, Sabatini BL (2020) Anatomical and single-cell transcriptional profiling of the murine habenular complex. Elife 9:e51271
Wang D, Li Y, Feng Q, Guo Q, Zhou J, Luo M (2017) Learning shapes the aversion and reward responses of lateral habenula neurons. Elife 6:e23045
Winter C, von Rumohr A, Mundt A, Petrus D, Klein J, Lee T, Morgenstern R, Kupsch A, Juckel G (2007) Lesions of dopaminergic neurons in the substantia nigra pars compacta and in the ventral tegmental area enhance depressive-like behavior in rats. Behav Brain Res 184:133–141
Wirtshafter D, Krebs JC (1997) Interactive effects of stimulation of d1 and d2 dopamine receptors on Fos expression in the lateral habenula. Brain Res 750:245–250
Yang Y, Wang H, Hu J, Hu H (2018) Lateral habenula in the pathophysiology of depression. Curr Opin Neurobiol 48:90–96
Zhao H, Zhang BL, Yang SJ, Rusak B (2015) The role of lateral habenula-dorsal raphe nucleus circuits in higher brain functions and psychiatric illness. Behav Brain Res 277:89–98
Zhou L, Liu MZ, Li Q, Deng J, Mu D, Sun YG (2017) Organization of functional long-range circuits controlling the activity of serotonergic neurons in the dorsal raphe nucleus. Cell Rep 18:3018–3032
Acknowledgements
We thank members of the Neuroscience Research Center, First Hospital of Jilin University for the technical support.
Funding
This study was supported by the National Natural Science Foundation of China (32171145), the Program for JLU Science and Technology Innovative Research Team (JLUSTIRT:2017TD-30), Natural Science Foundation of Jilin Province (Grant no. 20200201330JC). The funding bodies had no role in study design, sample collection, data analysis, interpretation, and manuscript preparation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to report.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, X., Liu, X., Luan, S. et al. Optogenetic activation of the lateral habenulaD1R–ventral tegmental area circuit induces depression-like behavior in mice. Eur Arch Psychiatry Clin Neurosci (2024). https://doi.org/10.1007/s00406-023-01743-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00406-023-01743-2