Abstract
Purpose
Glutathione peroxidase 1 (GPx-1) is a selenium-dependent detoxifying enzyme involved in the protection of cells against oxidative damage. Some genetic association studies reported significant associations between GPx-1 Pro198Leu variant and carcinogenesis across different populations; however, the impact of this variant on nasopharyngeal carcinoma (NPC) has not been explored. Therefore, the present study was planned to evaluate the potential involvement of the GPx-1 Pro198Leu variant and plasma GPx activity in the risk of developing NPC in a Tunisian population.
Methods
The GPx-1 Pro198Leu genotype was determined in 327 NPC patients and 150 healthy controls by the RFLP-PCR analysis. The correlation between the GPx-1 variant and the clinicopathological parameters was examined. GPx activity was assessed in the plasma of 119 NPC patients and 58 healthy control subjects and according to GPx-1 genotypes and clinicopathological characteristics of NPC patients.
Results
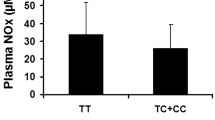
A significant association was found between GPx-1 Pro198Leu variant and NPC risk in a Tunisian population. The allelic frequencies of Pro and Leu alleles were 32% versus 68% and 41% versus 59% in NPC cases and controls, respectively. Thus, the minor 198 Leu allele increased significantly in NPC patients and appeared as a potential risk factor for NPC occurrence (OR = 1.48, CI 95% = 1.14–1.91, p = 0.002). The plasma GPx activity was significantly higher in NPC patients than in controls (p = 0.03). According to the clinicopathological characteristics of NPC patients, GPx activity decreased significantly in patients with lymph node metastasis (p = 0.004).
Conclusion
This is the first study showing a strong association between GPx-1 Pro198Leu genetic variant and NPC risk. GPx-1 Pro198Leu variant increased the development of regional lymph node metastasis. Plasma GPx activity was higher in NPC patients. Thus, GPx-1 gene could be considered as a determinant factor influencing NPC risk and progression.

Similar content being viewed by others
References
Khaali W, Moumad K, Ben Driss EK et al (2016) No association between TGF-β1 polymorphisms and risk of nasopharyngeal carcinoma in a large North African case control study. BMC Med Genet 17(1):72
Loong HH, Ma BB, Chan AT (2008) Update on the management and therapeutic monitoring of advanced nasopharyngeal cancer. HematolOncolClin North Am 22(6):1267–1278
Afqir S, Ismaili N, Errihani H (2009) Concurrent chemoradiotherapy in the management of advanced nasopharyngeal carcinoma: current status. J Cancer Res Ther 5(1):3–7
Yun M, Bai HY, Zhang JX et al (2015) ULK1: a promising biomarker in predicting poor prognosis and therapeutic response in human nasopharygeal carcinoma. PLoS ONE 10:e0117375
Boussen H, Ghorbal L, Naouel L et al (2012) Nasopharyngeal cancer around the Mediterranean area: standard of care. Crit Rev OncolHematol 84(Suppl 1):e106–e109
Chang ET, Adami HO (2006) The enigmatic epidemiology of nasopharyngeal carcinoma. Cancer EpidemiolBiomarkPrev 15(10):1765–1777
Hildesheim A, Wang CP (2012) Genetic predisposition factors and nasopharyngeal carcinoma risk: a review of epidemiological association studies, 2000–2011: Rosetta Stone for NPC: genetics, viral infection, and other environmental factors. Semin Cancer Biol 22:107–116
Feng BJ, Khyatti M, Ben-Ayoub W et al (2009) Cannabis, tobacco and domestic fumes intake are associated with nasopharyngeal carcinoma in North Africa. Br J Cancer 101(7):1207–1212
Simons MJ (2011) Nasopharyngeal carcinoma as a paradigm of cancer genetics. Chin J Cancer 30(2):79–84
Aouf S, Laaribi E, Gabbouj S (2019) Contribution of Nitric oxide synthase 3 genetic variants to nasopharyngeal carcinoma risk and progression in a Tunisian population. Eur Arch Otorhinolaryngol 276(4):1231–1239
Huang YJ, Zhang BB, Ma N, Murata M, Tang AZ, Huang GW (2011) Nitrativeand oxidative DNA damage as potential survival biomarkers for nasopharyngeal carcinoma. Med Oncol 28:377–384
Sharma A, Yuen D, Huet O et al (2016) Lack of glutathione peroxidase-1 facilitates a pro-inflammatory and activated vascular endothelium. VasculPharmacol 79:32–42
Flohé L (1988) Glutathione peroxidase. Basic Life Sci 49:663–668
Ichimura Y, Habuchi T, Tsuchiya N et al (2004) Increased risk of bladder cancer associated with a glutathione peroxidase 1 codon 198 variant. J Urol 172(2):728–732
Raaschou-Nielsen O, Sørensen M, Hansen RD et al (2007) GPX1 Pro198Leu polymorphism, interactions with smoking and alcohol consumption, and risk for lung cancer. CancerLett 247:293–300
Hu J, Zhou GW, Wang N et al (2010) GPX1 Pro198Leu polymorphism and breast cancer risk: a meta-analysis. Breast Cancer Res Treat 124(2):425–431
Hansen RD, Krath BN, Frederiksen K et al (2009) GPX1 Pro(198)Leu polymorphism, erythrocyte GPX activity, interaction with alcohol consumption and smoking, and risk of colorectal cancer. Mutat Res 664(1–2):13–19
Arsova-Sarafinovska Z, Matevska N, Eken A et al (2009) Glutathione peroxidase 1 (GPX1) genetic polymorphism, erythrocyte GPX activity, and prostate cancer risk. IntUrolNephrol 41(1):63–70
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16(3):1215
Aydin A, Hilmi O, Sayal A (2001) Oxidative stress and nitric oxide related parameters in type II diabetes mellitus: effects of glycemic control. ClinBiochem 34:65–70
Whang-Peng J, Knutsen T, Gazdar A et al (1991) structural and numerical chromosome changes in non-small cell lung cancer. Genes Chromosomes Cancer 3:168–188
Yokoyama S, Yamakawa K, Tsuchiya E (1992) Deletion mapping on the short arm of chromosome 3 in squamous cell carcinoma and adenocarcinoma of the lung. Cancer Res 52:873–877
Zhang JX, Wang ZM, Zhang JJ (2014) Association of glutathione peroxidase-1 (GPx-1) rs1050450 Pro198Leu and Pro197Leu polymorphisms with cardiovascular risk: a meta-analysis of observational studies. J GeriatrCardiol 11(2):141–150
Diwadkar-Navsariwala V, Diamond AM (2004) The link between selenium and chemoprevention: a case for selenoproteins. J Nutr 134:2899–2902
Zhuo P, Diamond AM (2009) Molecular mechanisms by which selenoproteins affect cancer risk and progression. BiochimBiophysActa 1790:1546–1554
Gouaze V, Andrieu-Abadie N, Cuvillier O et al (2002) Glutathione peroxidase-1 protects from CD95-induced apoptosis. J BiolChem 277:42867–42874
Baliga MS, Wang H, Zhuo P (2007) Selenium and GPx-1 overexpression protect mammalian cells against UV-induced DNA damage. Biol Trace Elem Res 115:227–242
Diamond AM, Hu YJ, Mansur DB (2001) Glutathione peroxidase and viral replication: implications for viral evolution and chemoprevention. BioFactors 14:205–210
Jiao Y, Wang Y, Guo S, Wang G (2017) Glutathione peroxidases as oncotargets. Oncotarget 8(45):80093–80102
Brigelius-Flohé R, Kipp AP (2012) Physiological functions of GPx2 and its role in inflammation-triggered carcinogenesis. Ann N Y AcadSci 1259:19–25
Zhuo P, Goldberg M, Herman L (2009) Molecular consequences of genetic variations in the glutathione peroxidase 1 selenoenzyme. Can Res 69(20):8183–8190
Lee C-H, Lee KY, Choe K-H (2006) Effects of oxidative DNA damage and genetic polymorphism of the glutathione peroxidase 1 (GPX1) and 8-oxoguanine glycosylase1 (hOGG1) on lung cancer. J Prev Med Public Health 39(2):130–134
Liwei L, Wei Z, Ruifa H et al (2012) Association between genetic variants in glutathione peroxidase 1 gene and risk of prostate cancer: a meta-analysis. MolBiol Rep 39:8615–8619
Parlaktas BS, Atilgan D, Gencten Y (2015) A pilot study of the association of manganese superoxide dismutase and glutathione peroxidase 1 single gene polymorphisms with prostate cancer and serum prostate specific antigen levels. Arch Med Sci 11(5):994–1000
Men T, Zhang X, Yang J et al (2014) The rs1050450 C > T polymorphism of GPX1 is associated with the risk of bladder but not prostate cancer: evidence from a meta-analysis. TumorBiol 35(1):269–275
Paz-y-Mino C, Munoz MJ, Lopez-Cortes A et al (2010) Frequency of polymorphisms pro198leu in GPX-1 gene and ile58thr in MnSOD gene in the altitude Ecuadorian population with bladder cancer. Oncol Res 18:395–400
Erdem O, Eken A, Akay C (2012) Association of GPX1 polymorphism, GPX activity and prostate cancer risk. Hum ExpToxicol 31(1):24–31
Choi JY, Neuhouser ML, Barnett M et al (2007) Polymorphisms in oxidative stress-related genes are not associated with prostate cancer risk in heavy smokers. Cancer Epidemiol Biomarkers Prev 16:1115–1120
Ravn-Haren G, Olsen A, Tjonneland A et al (2006) Associations between GPX1 Pro198Leu polymorphism, erythrocyte GPX activity, alcohol consumption and breast cancer risk in a prospective cohort study. Carcinogenesis 27:820–825
Hu Y, Benya RV, Carroll RE et al (2005) Allelic loss of the gene for the GPX1 selenium-containing protein is a common event in cancer. J Nutr 135:3021S-3024S
Knight JA, Onay UV, Wells S (2004) Genetic variants of GPX1 and SOD2 and breast cancer risk at the Ontario site of breast cancer family registry. Cancer EpidemiolBiomarkPrev 13:146–149
Cox DG, Hankinson SE, Kraft P et al (2004) No association between GPX1Pro198Leu and breast cancer risk. Cancer EpidemiolBiomarkPrev 13:1821–1822
Ratnasinghe D, Tangrea JA, Andersen MR et al (2000) Glutathione peroxidase codon 198 polymorphism variant increases lung cancer risk. Cancer Res 60(22):6381–6383
Yang P, Bamlet WR, Ebbert JO (2004) Glutathione pathway genes and lung cancer risk in young and old populations. Carcinogenesis 25:1935–1944
Fadhlaoui-Zid K, Garcia-Bertrand R, Alfonso-Sanchez MA (2015) Sousse: extreme genetic heterogeneity in North Africa. J Hum Genet 60(1):41–49
Ishida K, Morino T, Takagi K, Sukenaga Y (1987) Nucleotide sequence of a human gene for glutathione peroxidase. Nucleic Acids Res 15(23):10051
Chen J, Cao Q, Qin C et al (2011) GPx-1 polymorphism (rs1050450) contributes to tumor susceptibility: evidence from meta-analysis. J Cancer Res ClinOncol 137:1553–1561
Dragsted LO, Pedersen A, Hermetter A et al (2004) The 6-a-day study: effects of fruit and vegetables on markers of oxidative stress and antioxidativedefense in healthy nonsmokers. Am J ClinNutr 79:1060–1072
Zergoun AA, Zebboudg A, Sellam SL et al (2016) IL-6/NOS2 inflammatory signals regulate MMP-9 and MMP-2 activity and disease outcome in nasopharyngeal carcinoma patients. TumorBiol 37(3):3505–3514
Zhang Y, Handy DE, Loscalzo J (2005) Adenosine-dependant induction of glutathione peroxidase 1 in human primary endothelial cells and protection against oxidative stress. Circ Res 96:831–837
Gourzones C, Barjon C, Busson P (2012) Host-tumor interactions in nasopharyngeal carcinomas. Semin Cancer Biol 22:127–136
Acknowledgments
This study was supported by the Ministry of Higher Education and Scientific Research and by the Ministry of Health of the Republic of Tunisia. The authors would like to thank Dr. Samir Bougattaya for proof-reading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Laribi, A., Aouf, S., Gabbouj, S. et al. Human glutathione peroxidase codon 198 variant increases nasopharyngeal carcinoma risk and progression. Eur Arch Otorhinolaryngol 278, 4027–4034 (2021). https://doi.org/10.1007/s00405-021-06628-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-021-06628-5