Abstract
Background
Hepatoblastoma (HB) is a common primary malignant liver tumour in children, mainly treated by means of traditional chemotherapy using platinum and doxorubicin (ADM). There has been limited progress in the research and development of new drugs for treating HB.
Methods
A tumour biopsy from a child with HB was implanted into immunodeficient mice. The primary tumour and patient-derived xenograft (PDX) tumour were extensively characterised by histology, immunohistochemistry (IHC), and humanisation identification. We used the PDX model to evaluate the anti-tumour effects of anlotinib oxaliplatin (L-OHP) and sorafenib on childhood HB.
Results
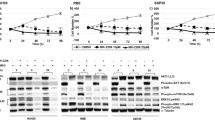
The established PDX model maintained the histological characteristics of the primary tumour. Anlotinib, L-OHP, and sorafenib can significantly inhibit the tumour growth in the PDX model. There was no obvious damage of the drugs to the heart, liver and kidney of the mice, and the side effects observed were light.
Conclusion
We have successfully established a PDX model of childhood HB. The model retains important molecular characteristics of human primary tumours. Using the model, it was found that anlotinib, L-OHP, and sorafenib have a good inhibitory effect on the growth of childhood HB. This provides a preliminary research basis for the clinical application of the drugs.








Similar content being viewed by others
References
Hiyama E et al (2015) Mortality and morbidity in primarily resected hepatoblastomas in Japan: experience of the JPLT (Japanese Study Group for Pediatric Liver Tumor) trials. J Pediatr Surg 50(12):2098–2101. https://doi.org/10.1016/j.jpedsurg.2015.08.035
Czauderna P et al (2016) The children’s hepatic tumors international collaboration (CHIC): novel global rare tumor database yields new prognostic factors in hepatoblastoma and becomes a research model. Eur J Cancer 52:92–101. https://doi.org/10.1016/j.ejca.2015.09.023
DeBord LC et al (2018) The chick chorioallantoic membrane (CAM) as a versatile patient-derived xenograft (PDX) platform for precision medicine and preclinical research. Am J Cancer Res 8(8):1642–1660
Fujii E, Kato A, Suzuki M (2020) Patient-derived xenograft (PDX) models: characteristics and points to consider for the process of establishment. J Toxicol Pathol 33(3):153–160. https://doi.org/10.1293/tox.2020-0007
Xin Y et al (2020) Establishment of a jaw fibrosarcoma patient-derived xenograft and evaluation of the tumor suppression efficacy of plumbagin against jaw fibrosarcoma. Front Oncol 10:1479. https://doi.org/10.3389/fonc.2020.01479
Rivankar S (2014) An overview of doxorubicin formulations in cancer therapy. J Cancer Res Ther 10:853–858. https://doi.org/10.4103/0973-1482.139267
Carvalho FS et al (2014) Doxorubicin-induced cardiotoxicity: from bioenergetic failure and cell death to cardiomyopathy. Med Res Rev 34(1):106–135. https://doi.org/10.1002/med.21280
Limsuwan T, Castells MC (2010) Outcomes and safety of rapid desensitization for chemotherapy hypersensitivity. Expert Opin Drug Saf 9(1):39–53. https://doi.org/10.1517/14740330903446936
Han B et al (2018) Anlotinib as a third-line therapy in patients with refractory advanced non-small-cell lung cancer: a multicentre, randomised phase II trial (ALTER0302). Br J Cancer 118(5):654–661. https://doi.org/10.1038/bjc.2017.478
Ruan X et al (2019) Antitumor effects of anlotinib in thyroid cancer. Endocr Relat Cancer 26(1):153–164. https://doi.org/10.1530/ERC-17-0558
Zhou A-P et al (2019) Anlotinib versus sunitinib as first-line treatment for metastatic renal cell carcinoma: a randomized phase II clinical trial. Oncologist 24(8):e702–e708. https://doi.org/10.1634/theoncologist.2018-0839
Song F et al (2020) Anlotinib suppresses tumor progression via blocking the VEGFR2/PI3K/AKT cascade in intrahepatic cholangiocarcinoma. Cell Death Dis 11(7):573. https://doi.org/10.1038/s41419-020-02749-7
Noda S et al (2020) Sorafenib exposure and its correlation with response and safety in advanced hepatocellular carcinoma: results from an observational retrospective study. Cancer Chemother Pharmacol 86(1):129–139. https://doi.org/10.1007/s00280-020-04105-0
Chollet P et al (1996) Single agent activity of oxaliplatin in heavily pretreated advanced epithelial ovarian cancer. Ann Oncol 7(10):1065–1070. https://doi.org/10.1093/oxfordjournals.annonc.a010500
Esim O et al (2020) Development, optimization and in vitro evaluation of oxaliplatin loaded nanoparticles in non-small cell lung cancer. DARU J Pharm Sci 28(2):673–684. https://doi.org/10.1007/s40199-020-00374-5
Tanaka R et al (2020) Reactive fibrosis precedes doxorubicin-induced heart failure through sterile inflammation. ESC Heart Fail 7(2):588–603. https://doi.org/10.1002/ehf2.12616
Kimura G et al (2017) Sorafenib as a potential strategy for refractory pulmonary arterial hypertension. Pulm Pharmacol Ther 44:46–49. https://doi.org/10.1016/j.pupt.2017.03.009
Cividalli A et al (2002) Radiosensitization by oxaliplatin in a mouse adenocarcinoma: influence of treatment schedule. Int J Radiat Oncol*Biol*Phys 52(4):1092–1098. https://doi.org/10.1016/S0360-3016(01)02792-4
Bondoc A et al (2021) Identification of distinct tumor cell populations and key genetic mechanisms through single cell sequencing in hepatoblastoma. Commun Biol 4(1):1049–1049. https://doi.org/10.1038/s42003-021-02562-8
Johnston Ii ME et al (2021) Olaparib inhibits tumor growth of hepatoblastoma in patient-derived xenograft models. Hepatology 74(4):2201–2215. https://doi.org/10.1002/hep.31919
Xu R et al (2019) Clinical value of spectral CT imaging combined with AFP in identifying liver cancer and hepatic focal nodular hyperplasia. J B.U.ON: Off J Balkan Union Oncol 24(4):1429–1434
Marcu L, Bezak E (2010) Modelling of tumour repopulation after chemotherapy. Australas Phys Eng Sci Med 33(3):265–270. https://doi.org/10.1007/s13246-010-0026-4
Senan S, Smit EF (2007) Design of clinical trials of radiation combined with antiangiogenic therapy. Oncologist 12(4):465–477. https://doi.org/10.1634/theoncologist.12-4-465
Jain RK et al (2006) Lessons from phase III clinical trials on anti-VEGF therapy for cancer. Nat Clin Pract Oncol 3(1):24–40. https://doi.org/10.1038/ncponc0403
Lin B et al (2018) Anlotinib inhibits angiogenesis via suppressing the activation of VEGFR2, PDGFRβ and FGFR1. Gene 654:77–86. https://doi.org/10.1016/j.gene.2018.02.026
Rini BI et al (2020) Tivozanib versus sorafenib in patients with advanced renal cell carcinoma (TIVO-3): a phase 3, multicentre, randomised, controlled, open-label study. Lancet Oncol 21(1):95–104. https://doi.org/10.1016/S1470-2045(19)30735-1
Falcon BL et al (2011) Reduced VEGF production, angiogenesis, and vascular regrowth contribute to the antitumor properties of dual mTORC1/mTORC2 inhibitors. Can Res 71(5):1573. https://doi.org/10.1158/0008-5472.CAN-10-3126
Yang C et al (2018) Ki67 targeted strategies for cancer therapy. Clin Transl Oncol 20(5):570–575. https://doi.org/10.1007/s12094-017-1774-3
Li LT et al (2015) Ki67 is a promising molecular target in the diagnosis of cancer (review). Mol Med Rep 11(3):1566–1572. https://doi.org/10.3892/mmr.2014.2914
Hsieh MJ et al (2020) Inactivation of APC induces CD34 upregulation to promote epithelial-mesenchymal transition and cancer stem cell traits in pancreatic cancer. Int J Mol Sci. https://doi.org/10.3390/ijms21124473
Acknowledgements
This study was funded by the Futang Children Health Development Planning Foundation (FTCSF-2018-03), the National Natural Science Foundation of China (No: 81960101; 81960443), Science Technology Foundation of Jiangxi Province (20212BAG70003), and Jiangxi Province Health Committee Research Project (20203689; 202110098).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare there is no interest conflict.
Ethical approval
All experiments using immunodeficient mice were performed in accordance with guidelines approved by the Institutional Animal Care and Use Committee of Nanchang University. The study was approved by the Ethics Committee of Affiliated Children’s Hospital of Nanchang University. Informed consent was obtained from the patient’s guardian.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, D., Chen, F., Gong, D. et al. Establishment of childhood hepatoblastoma xenografts and evaluation of the anti-tumour effects of anlotinib, oxaliplatin and sorafenib. Pediatr Surg Int 38, 465–472 (2022). https://doi.org/10.1007/s00383-021-05043-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-021-05043-5