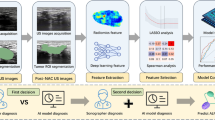
Abstract
Objectives
To compare computed tomography (CT)–based radiomics for preoperatively differentiating type I and II epithelial ovarian cancers (EOCs) using different machine learning classifiers and to construct and validate the best diagnostic model.
Methods
A total of 470 patients with EOCs were included retrospectively. Patients were divided into a training dataset (N = 329) and a test dataset (N = 141). A total of 1316 radiomics features were extracted from the portal venous phase of contrast-enhanced CT images for each patient, followed by dimension reduction of the features. The support vector machine (SVM), k-nearest neighbor (KNN), random forest (RF), naïve Bayes (NB), logistic regression (LR), and eXtreme Gradient Boosting (XGBoost) classifiers were trained to obtain the radiomics signatures. The performance of each radiomics signature was evaluated and compared by the area under the receiver operating characteristic curve (AUC) and relative standard deviation (RSD). The best radiomics signature was selected and combined with clinical and radiological features to establish a combined model. The diagnostic value of the combined model was assessed.
Results
The LR-based radiomics signature performed well in the test dataset, with an AUC of 0.879 and an accuracy of 0.773. The combined model performed best in both the training and test datasets, with AUCs of 0.900 and 0.934 and accuracies of 0.848 and 0.823, respectively.
Conclusion
The combined model showed the best diagnostic performance for distinguishing between type I and II EOCs preoperatively. Therefore, it can be a useful tool for clinical individualized EOC classification.
Key Points
• Radiomics features extracted from computed tomography (CT) could be used to differentiate type I and II epithelial ovarian cancers (EOCs).
• Machine learning can improve the performance of differentiating type I and II EOCs.
• The combined model exhibited the best diagnostic capability over the other models in both the training and test datasets.






Similar content being viewed by others
Abbreviations
- EOCs:
-
Epithelial ovarian cancers
- GLCM:
-
Gray-level co-occurrence matrix
- GLDM:
-
Gray-level dependence matrix
- GLRLM:
-
Gray-level run-length matrix
- GLSZM:
-
Gray-level size zone matrix
- ICC:
-
Intraclass correlation coefficient
- KNN:
-
K-nearest neighbor
- LR:
-
Logistic regression
- NB:
-
Naïve Bayes
- NGTDM:
-
Neighboring gray-tone difference matrix
- OC:
-
Ovarian cancer
- RF:
-
Random forest
- RSD:
-
Relative standard deviation
- SVM:
-
Support vector machines
- VOI:
-
Volume of interest
- XGBoost:
-
eXtreme Gradient Boosting
References
Torre LA, Trabert B, DeSantis CE et al (2018) Ovarian cancer statistics, 2018. CA Cancer J Clin 68:284–296
Siegel RL, Miller KD, Fuchs HE, Jemal A (2021) Cancer statistics, 2021. CA Cancer J Clin 71:7–33
Kurman RJ, Shih Ie M (2008) Pathogenesis of ovarian cancer: lessons from morphology and molecular biology and their clinical implications. Int J Gynecol Pathol 27:151–160
Kurman RJ, Shih Ie M (2016) The dualistic model of ovarian carcinogenesis: revisited, revised, and expanded. Am J Pathol 186:733–747
Cree IA, White VA, Indave BI, Lokuhetty D (2020) Revising the WHO classification: female genital tract tumours. Histopathology 76:151–156
Lambin P, Leijenaar RTH, Deist TM et al (2017) Radiomics: the bridge between medical imaging and personalized medicine. Nat Rev Clin Oncol 14:749–762
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: images are more than pictures, they are data. Radiology 278:563–577
Chiappa V, Bogani G, Interlenghi M et al (2021) The Adoption of Radiomics and machine learning improves the diagnostic processes of women with Ovarian MAsses (the AROMA pilot study). J Ultrasound 24:429–437
Gu D, Hu Y, Ding H et al (2019) CT radiomics may predict the grade of pancreatic neuroendocrine tumors: a multicenter study. Eur Radiol 29:6880–6890
Cui X, Wang N, Zhao Y et al (2019) Preoperative prediction of axillary lymph node metastasis in breast cancer using radiomics features of DCE-MRI. Sci Rep 9:2240
Zhou Y, He L, Huang Y et al (2017) CT-based radiomics signature: a potential biomarker for preoperative prediction of early recurrence in hepatocellular carcinoma. Abdom Radiol (NY) 42:1695–1704
Qian L, Ren J, Liu A et al (2020) MR imaging of epithelial ovarian cancer: a combined model to predict histologic subtypes. Eur Radiol 30:5815–5825
Jian J, Li Y, Pickhardt PJ et al (2021) MR image-based radiomics to differentiate type Ι and type ΙΙ epithelial ovarian cancers. Eur Radiol 31:403–410
Zhang H, Mao Y, Chen X et al (2019) Magnetic resonance imaging radiomics in categorizing ovarian masses and predicting clinical outcome: a preliminary study. Eur Radiol 29:3358–3371
Forstner R, Sala E, Kinkel K, Spencer JA (2010) ESUR guidelines: ovarian cancer staging and follow-up. Eur Radiol 20:2773–2780
An H, Wang Y, Wong EMF et al (2021) CT texture analysis in histological classification of epithelial ovarian carcinoma. Eur Radiol 31:5050–5058
van Griethuysen JJM, Fedorov A, Parmar C et al (2017) Computational radiomics system to decode the radiographic phenotype. Cancer Res 77:e104–e107
Zhang Y, Zhang B, Liang F et al (2019) Radiomics features on non-contrast-enhanced CT scan can precisely classify AVM-related hematomas from other spontaneous intraparenchymal hematoma types. Eur Radiol 29:2157–2165
McCluggage WG (2011) Morphological subtypes of ovarian carcinoma: a review with emphasis on new developments and pathogenesis. Pathology 43:420–432
Masoodi T, Siraj S, Siraj AK et al (2020) Genetic heterogeneity and evolutionary history of high-grade ovarian carcinoma and matched distant metastases. Br J Cancer 122:1219–1230
Liu D, Zhang L, Indima N et al (2017) CT and MRI findings of type I and type II epithelial ovarian cancer. Eur J Radiol 90:225–233
Papanikolaou N, Matos C, Koh DM (2020) How to develop a meaningful radiomic signature for clinical use in oncologic patients. Cancer Imaging 20:33
Xie XJ, Liu SY, Chen JY et al (2021) Development of unenhanced CT-based imaging signature for BAP1 mutation status prediction in malignant pleural mesothelioma: Consideration of 2D and 3D segmentation. Lung Cancer 157:30–39
Ortiz-Ramon R, Larroza A, Ruiz-Espana S, Arana E, Moratal D (2018) Classifying brain metastases by their primary site of origin using a radiomics approach based on texture analysis: a feasibility study. Eur Radiol 28:4514–4523
Yang L, Yang J, Zhou X et al (2019) Development of a radiomics nomogram based on the 2D and 3D CT features to predict the survival of non-small cell lung cancer patients. Eur Radiol 29:2196–2206
Fu GS, Levin-Schwartz Y, Lin QH, Zhang D (2019) Machine learning for medical imaging. J Healthc Eng 2019:9874591
Qian Z, Li Y, Wang Y et al (2019) Differentiation of glioblastoma from solitary brain metastases using radiomic machine-learning classifiers. Cancer Lett 451:128–135
Liu J, Zeng P, Guo W et al (2021) Prediction of high-risk cytogenetic status in multiple myeloma based on magnetic resonance imaging: utility of radiomics and comparison of machine learning methods. J Magn Reson Imaging 54:1303–1311
Chen H, Zhang X, Wang X et al (2021) MRI-based radiomics signature for pretreatment prediction of pathological response to neoadjuvant chemotherapy in osteosarcoma: a multicenter study. Eur Radiol 31:7913–7924
Fang J, Zhang B, Wang S et al (2020) Association of MRI-derived radiomic biomarker with disease-free survival in patients with early-stage cervical cancer. Theranostics 10:2284–2292
Shu Z, Fang S, Ding Z et al (2019) MRI-based radiomics nomogram to detect primary rectal cancer with synchronous liver metastases. Sci Rep 9:3374
Gui J, Li H (2005) Penalized Cox regression analysis in the high-dimensional and low-sample size settings, with applications to microarray gene expression data. Bioinformatics 21:3001–3008
Yin P, Mao N, Zhao C et al (2019) Comparison of radiomics machine-learning classifiers and feature selection for differentiation of sacral chordoma and sacral giant cell tumour based on 3D computed tomography features. Eur Radiol 29:1841–1847
Lee SM, Park CM, Goo JM, Lee HJ, Wi JY, Kang CH (2013) Invasive pulmonary adenocarcinomas versus preinvasive lesions appearing as ground-glass nodules: differentiation by using CT features. Radiology 268:265–273
Aslan K, Onan MA, Yilmaz C, Bukan N, Erdem M (2020) Comparison of HE 4, CA 125, ROMA score and ultrasound score in the differential diagnosis of ovarian masses. J Gynecol Obstet Hum Reprod 49:101713
Gąsiorowska E, Michalak M, Warchoł W et al (2015) Clinical application of HE4 and CA125 in ovarian cancer type I and type II detection and differential diagnosis. Ginekol Pol 86:88–93
Kurman RJ, Carcangiu ML, Herrington CS (2014) World Health Organisation classification of tumours of the female reproductive organs. International Agency for Research on Cancer
Yu XP, Wang L, Yu HY et al (2021) MDCT-based radiomics features for the differentiation of serous borderline ovarian tumors and serous malignant ovarian tumors. Cancer Manag Res 13:329–336
Funding
This work was supported by the Chinese National Key Research and Development Project (Grant No. 2021YFC2500400 and Grant No. 2021YFC2500402), the Breeding Project of National Natural Science Foundation of China, Tianjin Medical University Cancer Institute and Hospital (Grant No. 210207), and the Tianjin Key Medical Discipline (Specialty) Construction Project (TJYXZDXK-009A).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr. Zhaoxiang Ye.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
• diagnostic study
• performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 615 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, J., Li, X., Ma, J. et al. Computed tomography–based radiomics machine learning classifiers to differentiate type I and type II epithelial ovarian cancers. Eur Radiol 33, 5193–5204 (2023). https://doi.org/10.1007/s00330-022-09318-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-022-09318-w