Abstract
Introduction
Patients treated with immune checkpoint inhibitors (ICIs) may not response to treatment and are at risk for immune-related adverse events (irAEs). Platelet function has been linked to both oncogenesis and immune evasion. We studied the association between the change in mean platelet volume (MPV), platelet count, survival, and the risk of developing irAEs in patients with metastatic non-small cell lung cancer (NSCLC) who have received first-line ICI.
Methods
In this retrospective study, delta (∆) MPV was defined as the difference between cycle 2 and baseline MPV. Patient data were collected via chart review, and Cox proportional hazard and Kaplan–Meier method were used to assess the risk and estimate median overall survival.
Results
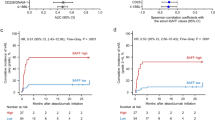
We identified 188 patients treated with first-line pembrolizumab, with or without concurrent chemotherapy. There were 80 (42.6%) patients received pembrolizumab monotherapy, and 108 (57.4%) received pembrolizumab in combination with platinum-based chemotherapy. Patients whose MPV (∆MPV ≤ 0) decreased had hazard ratio (HR) = 0.64 (95% CI 0.43–0.94) for death with p = 0.023. Patients with ∆MPV ≤ − 0.2 fL (median), there was a 58% increase in the risk of developing irAE (HR = 1.58, 95% CI 1.04–2.40, p = 0.031). Thrombocytosis at baseline and cycle 2 was associated with shorter OS with p = 0.014 and 0.039, respectively.
Conclusion
Change in MPV after 1 cycle of pembrolizumab-based treatment was significantly associated with overall survival as well as the occurrence of irAEs in patients with metastatic NSCLC in the first-line setting. In addition, thrombocytosis was associated with poor survival.




Similar content being viewed by others

Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- ∆MPV:
-
Delta mean platelet volume
- ECOG:
-
Eastern Cooperative Oncology Group performance status
- HR:
-
Hazard ratio
- IQR:
-
Interquartile range
- irAEs:
-
Immune-related adverse events
- MPV:
-
Mean platelet volume
- NSCLC:
-
Non-small cell lung cancer
- PD-L1:
-
Programmed death-ligand 1
- OS:
-
Overall survival
References
Gandhi L et al (2018) Pembrolizumab plus chemotherapy in metastatic non–small-cell lung cancer. N Engl J Med 378(22):2078–2092
Mok TSK et al (2019) Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet 393(10183):1819–1830
Reck M et al (2016) Pembrolizumab versus chemotherapy for PD-L1–positive non–small-cell lung cancer. N Engl J Med 375(19):1823–1833
Antonia SJ et al (2017) Durvalumab after chemoradiotherapy in stage III non–small-cell lung cancer. N Engl J Med 377(20):1919–1929
Qiu Z et al (2019) Achievements and futures of immune checkpoint inhibitors in non-small cell lung cancer. Exp Hematol Oncol 8(1):19
Postow MA, Sidlow R, Hellmann MD (2018) Immune-related adverse events associated with immune checkpoint blockade. New Engl J Med 378(2):158–168
Couey MA et al (2019) Delayed immune-related events (DIRE) after discontinuation of immunotherapy: diagnostic hazard of autoimmunity at a distance. J Immunother Cancer 7(1):165
Havel JJ, Chowell D, Chan TA (2019) The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat Rev Cancer 19(3):133–150
Webb NJ et al (1998) Vascular endothelial growth factor (VEGF) is released from platelets during blood clotting: implications for measurement of circulating VEGF levels in clinical disease. Clin Sci 94(4):395–404
Selheim F, Holmsen H, Vassbotn FS (2002) Identification of functional VEGF receptors on human platelets. FEBS Lett 512(1):107–110
Lindemann S et al (2001) Activated platelets mediate inflammatory signaling by regulated interleukin 1beta synthesis. J Cell Biol 154(3):485–490
Rachidi S et al (2017) Platelets subvert T cell immunity against cancer via GARP-TGFβ axis. Sci Immunol 2(11):eaai7911
Labelle M, Begum S, Hynes RO (2011) Direct signaling between platelets and cancer cells induces an epithelial-mesenchymal-like transition and promotes metastasis. Cancer Cell 20(5):576–590
Riesenberg BP et al (2019) Cutting edge: targeting thrombocytes to rewire anticancer immunity in the tumor microenvironment and potentiate efficacy of PD-1 blockade. J Immunol 203(5):1105–1110
Metelli A et al (2020) Thrombin contributes to cancer immune evasion via proteolysis of platelet-bound GARP to activate LTGF-β. Sci Transl Med 12(525):eaay4860
Handtke S et al (2019) Role of platelet size revisited—function and protein composition of large and small platelets. Thromb Haemost 119(03):407–420
Li N et al (2017) Elevated mean platelet volume predicts poor prognosis in colorectal cancer. Sci Rep 7(1):10261
Matowicka-Karna J et al (2013) Platelets and inflammatory markers in patients with gastric cancer. Clin Dev Immunol 2013:401623
Korniluk A et al (2019) Mean platelet volume (MPV): new perspectives for an old marker in the course and prognosis of inflammatory conditions. Mediat Inflamm 2019:9213074
Hsiehchen D et al (2022) Association between immune-related adverse event timing and treatment outcomes. Oncoimmunology 11(1):2017162
Martini C et al (2020) Platelets disrupt vasculogenic mimicry by cancer cells. Sci Rep 10(1):5869
Kanikarla-Marie P et al (2018) Platelet metabolism and other targeted drugs potential impact on immunotherapy. Front Oncol 8:107
Coussens LM, Werb Z (2002) Inflammation and cancer. Nature 420(6917):860–867
Wood LS (2019) Immune-related adverse events from immunotherapy: incorporating care step pathways to improve management across tumor types. J Adv Pract Oncol 10(Suppl 1):47–62
Davies M, Duffield EA (2017) Safety of checkpoint inhibitors for cancer treatment: strategies for patient monitoring and management of immune-mediated adverse events. Immunotargets Ther 6:51–71
Assi HA et al (2020) Development of thrombocytopenia is associated with improved survival in patients treated with immunotherapy. Future Sci OA 6(7):FSO581
Haddad TC et al (2022) Immune checkpoint inhibitor-related thrombocytopenia: incidence, risk factors and effect on survival. Cancer Immunol Immunother 71(5):1157–1165
Ma Y et al (2021) Prognostic significance of thrombocytosis in lung cancer: a systematic review and meta-analysis. Platelets 32(7):919–927
Zhuo Y, Lin L, Zhang M (2017) Pretreatment thrombocytosis as a significant prognostic factor in malignant mesothelioma: a meta-analysis. Platelets 28(6):560–566
Ye Q et al (2019) Association of pretreatment thrombocytosis with prognosis in ovarian cancer: a systematic review and meta-analysis. J Gynecol Oncol 30(1):e5
Rachidi S et al (2019) Platelet count correlates with stage and predicts survival in melanoma. Platelets 30(8):1042–1046
Foster CC et al (2021) Immune-related adverse events are associated with improved response, progression-free survival, and overall survival for patients with head and neck cancer receiving immune checkpoint inhibitors. Cancer 127(24):4565–4573
Haratani K et al (2018) Association of immune-related adverse events with nivolumab efficacy in non–small-cell lung cancer. JAMA Oncol 4(3):374–378
Eggermont AMM et al (2020) Association between immune-related adverse events and recurrence-free survival among patients with stage III melanoma randomized to receive pembrolizumab or placebo: a secondary analysis of a randomized clinical trial. JAMA Oncol 6(4):519–527
Freeman-Keller M et al (2016) Nivolumab in resected and unresectable metastatic melanoma: characteristics of immune-related adverse events and association with outcomes. Clin Cancer Res 22(4):886–894
Li M et al (2019) Change in neutrophil to lymphocyte ratio during immunotherapy treatment is a non-linear predictor of patient outcomes in advanced cancers. J Cancer Res Clin Oncol 145(10):2541–2546
Bagley SJ et al (2017) Pretreatment neutrophil-to-lymphocyte ratio as a marker of outcomes in nivolumab-treated patients with advanced non-small-cell lung cancer. Lung Cancer 106:1–7
Bai R et al (2020) Predictive biomarkers for cancer immunotherapy with immune checkpoint inhibitors. Biomark Res 8(1):34
Acknowledgements
The authors would like to thank the Biostatistics Shared Resource at The Ohio State University Comprehensive Cancer Center, Columbus, OH for statistical support. Dr. Owen is supported by the LUNGevity Foundation Career Development Award.
Funding
This study was supported by the National Institutes of Health (P30CA016058). Research support provided by the REDCap project and The Ohio State University Center for Clinical and Translational Science grant support (National Center for Advancing Translational Sciences, Grant UL1TR002733).
Author information
Authors and Affiliations
Contributions
ML and DO contributed to the study conception and design. Material preparation, data collection, and analysis were performed by all authors. The first draft of the manuscript was written by all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was reviewed and approved by the institutional review board (IRB 2018C0177) at the Ohio State University.
Consent to participate
This was a retrospective study and a waiver for consent from each individual patient was approved by the institutional review board.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, M., Zhao, S., Lopez, G. et al. Mean platelet volume, thrombocytosis, and survival in non-small cell lung cancer patients treated with first-line pembrolizumab alone or with chemotherapy. Cancer Immunol Immunother 72, 2067–2074 (2023). https://doi.org/10.1007/s00262-023-03392-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-023-03392-9