Abstract
Purpose
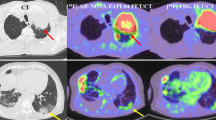
Retrospective analysis revealed increased [18F]AlF-NOTA-FAPI-04 uptake in the myocardium of patients with esophageal squamous cell cancer (ESCC) treated with concurrent chemoradiotherapy (CCRT). This study investigated and verified the feasibility of [18F]AlF-NOTA-FAPI-04 PET/CT for detecting radiation-induced myocardial damage (RIMD).
Methods
Myocardial FAPI uptake was analyzed before and during radiotherapy in thirteen ESCC patients treated with CCRT. In the animal study, a single dose of 50 Gy was delivered to the cardiac apex of Wistar rats (24 rats, including 16 RIMD model rats and 8 control model rats). RIMD model rats were scanned with [18F]AlF-NOTA-FAPI-04 PET/CT weekly for 12 weeks, and left ventricular ejection fraction (LVEF) was measured by magnetic resonance imaging. Dynamic, blocking, and [18F]FDG PET/CT studies (4 rats/group) were performed on RIMD rats at 5 weeks post-radiation, and histopathological analyses were conducted.
Results
Increased FAPI uptake in the myocardium was found after CCRT (1.53 ± 0.53 vs 1.88 ± 0.70, P = 0.015). In RIMD rats, significantly increased FAPI uptake in the damaged myocardium was observed from the 2nd week post-radiation exposure and peaked in the 5th week. Significantly more intense tracer accumulation was observed in the damaged myocardium than in the remote myocardium, as identified by decreased [18F]FDG uptake and confirmed by autoradiography, hematoxylin–eosin, Masson’s trichrome, and immunohistochemical staining. The LVEF remained unchanged at the 3rd week post-radiation exposure but was remarkably decreased compared with that in the control group at the 8th week.
Conclusion
Through clinical phenomena and animal experimental studies, this study indicated that [18F]AlF-NOTA-FAPI-04 PET/CT imaging can detect RIMD noninvasively and before a decrease in LVEF, indicating the clinical potential of [18F]AlF-NOTA-FAPI-04 as a PET/CT tracer for early monitoring of RIMD.







Similar content being viewed by others
Availability of data and material
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
All software applications or custom code are available in the public repository.
References
Yu AF, Ho AY, Braunstein LZ, Thor ME, Lee CK, Eaton A, et al. Assessment of early radiation-induced changes in left ventricular function by myocardial strain imaging after breast radiation therapy. J Am Soc Echocardiogr. 2019;32:521–8.
Beukema JC, van Luijk P, Widder J, Langendijk JA, Muijs CT. Is cardiac toxicity a relevant issue in the radiation treatment of esophageal cancer. Radiother Oncol. 2015;114:85–90.
Beukema JC, de Groot C, Plukker JTM, Vliegenthart R, Langendijk JA, van Luijk P, et al. Late cardiac toxicity of neo-adjuvant chemoradiation in esophageal cancer survivors: a prospective cross-sectional pilot study. Radiother Oncol. 2022;167:72–7.
Dess RT, Sun Y, Matuszak MM, Sun G, Soni PD, Bazzi L, et al. Cardiac events after radiation therapy: combined analysis of prospective multicenter trials for locally advanced non-small-cell lung cancer. J Clin Oncol. 2017;35:1395–402.
Speirs CK, DeWees TA, Rehman S, Molotievschi A, Velez MA, Mullen D, et al. Heart dose is an independent dosimetric predictor of overall survival in locally advanced non-small cell lung cancer. J Thorac Oncol. 2017;12:293–301.
Tohidinezhad F, Pennetta F, van Loon J, Dekker A, de Ruysscher D, Traverso A. Prediction models for treatment-induced cardiac toxicity in patients with non-small-cell lung cancer: a systematic review and meta-analysis. Clin Transl Radiat Oncol. 2022;33:134–44.
Burnhope E, Polcaro A, Runge JH, Granlund I, Bosio F, Troelstra MA, et al. Assessment of myocardial stiffness in patients with left ventricular hypertrophy: CMR elastography using intrinsic actuation. JACC Cardiovasc Imaging. 2022;15:1163–5.
Dreyfuss AD, Goia D, Shoniyozov K, Shewale SV, Velalopoulou A, Mazzoni S, et al. A novel mouse model of radiation-induced cardiac injury reveals biological and radiological biomarkers of cardiac dysfunction with potential clinical relevance. Clin Cancer Res. 2021;27:2266–76.
Mayr A, Klug G, Reindl M, Lechner I, Tiller C, Holzknecht M, et al. Evolution of myocardial tissue injury: a CMR study over a decade after STEMI. JACC Cardiovasc Imaging. 2022;15:1030–42.
Humeres C, Frangogiannis NG. Fibroblasts in the infarcted, remodeling, and failing heart. JACC Basic Transl Sci. 2019;4:449–67.
Hamson EJ, Keane FM, Tholen S, Schilling O, Gorrell MD. Understanding fibroblast activation protein (FAP): substrates, activities, expression and targeting for cancer therapy. Proteomics Clin Appl. 2014;8:454–63.
Brilla CG. Renin-angiotensin-aldosterone system and myocardial fibrosis. Cardiovasc Res. 2000;47:1–3.
Tillmanns J, Hoffmann D, Habbaba Y, Schmitto JD, Sedding D, Fraccarollo D, et al. Fibroblast activation protein alpha expression identifies activated fibroblasts after myocardial infarction. J Mol Cell Cardiol. 2015;87:194–203.
Henry LR, Lee HO, Lee JS, Klein-Szanto A, Watts P, Ross EA, et al. Clinical implications of fibroblast activation protein in patients with colon cancer. Clin Cancer Res. 2007;13:1736–41.
Chen L, Qiu X, Wang X, He J. FAP positive fibroblasts induce immune checkpoint blockade resistance in colorectal cancer via promoting immunosuppression. Biochem Biophys Res Commun. 2017;487:8–14.
Milner JM, Kevorkian L, Young DA, Jones D, Wait R, Donell ST, et al. Fibroblast activation protein alpha is expressed by chondrocytes following a pro-inflammatory stimulus and is elevated in osteoarthritis. Arthritis Res Ther. 2006;8:R23.
Acharya PS, Zukas A, Chandan V, Katzenstein AL, Pure E. Fibroblast activation protein: a serine protease expressed at the remodeling interface in idiopathic pulmonary fibrosis. Hum Pathol. 2006;37:352–60.
Rosenkrans ZT, Massey CF, Bernau K, Ferreira CA, Jeffery JJ, Schulte JJ, et al. [(68) Ga]Ga FAPI-46 PET for non-invasive detection of pulmonary fibrosis disease activity. Eur J Nucl Med Mol Imaging. 2022;49(11):3705–16.
Kaur H, Takefuji M, Ngai CY, Carvalho J, Bayer J, Wietelmann A, et al. Targeted ablation of periostin-expressing activated fibroblasts prevents adverse cardiac remodeling in mice. Circ Res. 2016;118:1906–17.
Varasteh Z, Mohanta S, Robu S, Braeuer M, Li Y, Omidvari N, et al. Molecular imaging of fibroblast activity after myocardial infarction using a (68)Ga-labeled fibroblast activation protein inhibitor, FAPI-04. J Nucl Med. 2019;60:1743–9.
Kratochwil C, Flechsig P, Lindner T, Abderrahim L, Altmann A, Mier W, et al. (68)Ga-FAPI PET/CT: tracer uptake in 28 different kinds of cancer. J Nucl Med. 2019;60:801–5.
Komek H, AUID- Oho, Can C, Kaplan I, Gundogan C, Kepenek F, et al. Comparison of [(68) Ga]Ga-DOTAFAPI-04 PET/CT and [(18)F]FDG PET/CT in colorectal cancer. Eur J Nucl Med Mol Imaging 2022;49:3898–909.
Gu Y, Han K, Zhang Z, Zhao Z, Yan C, Wang L, et al. (68)Ga-FAPI PET/CT for molecular assessment of fibroblast activation in right heart in pulmonary arterial hypertension: a single-center, pilot study. J Nucl Cardiol. 2022. https://doi.org/10.1007/s12350-022-02952-3.
Diekmann J, AUID- Oho, Koenig T, Thackeray JT, Derlin T, Czerner C, et al. Cardiac Fibroblast Activation in Patients Early After Acute Myocardial Infarction: Integration with Magnetic Resonance Tissue Characterization and Subsequent Functional Outcome. J Nucl Med. 2022. https://doi.org/10.2967/jnumed.121.263555.
Wu M, Ning J, Li J, Lai Z, Shi X, Xing H, et al. Feasibility of in vivo imaging of fibroblast activation protein in human arterial walls. J Nucl Med. 2022;63:948–51.
Siebermair J, Kohler MI, Kupusovic J, Nekolla SG, Kessler L, Ferdinandus J, et al. Cardiac fibroblast activation detected by Ga-68 FAPI PET imaging as a potential novel biomarker of cardiac injury/remodeling. J Nucl Cardiol. 2021;28:812–21.
Wei Y, Cheng K, Fu Z, Zheng J, Mu Z, Zhao C, et al. [(18)F]AlF-NOTA-FAPI-04 PET/CT uptake in metastatic lesions on PET/CT imaging might distinguish different pathological types of lung cancer. Eur J Nucl Med Mol Imaging. 2022;49:1671–81.
Anvari A, Modiri A, Pandita R, Mahmood J, Sawant A. Online dose delivery verification in small animal image-guided radiotherapy. Med Phys. 2020;47:1871–9.
Ruan JL, Browning RJ, Yildiz YO, Gray M, Bau L, Kamila S, et al. Ultrasound-mediated gemcitabine delivery reduces the normal-tissue toxicity of chemoradiation therapy in a muscle-invasive bladder cancer model. Int J Radiat Oncol Biol Phys. 2021;109:1472–82.
Watzinger N, Saeed M, Wendland MF, Akbari H, Lund G, Higgins CB. Myocardial viability: magnetic resonance assessment of functional reserve and tissue characterization. J Cardiovasc Magn Reson. 2001;3:195–208.
Yushkevich PA, Gerig G. ITK-SNAP: an intractive medical image segmentation tool to meet the need for expert-guided segmentation of complex medical images. IEEE Pulse. 2017;8:54–7.
Rushton M, Kappel C, Lima I, Tuna M, Pritchard K, Hawken S, et al. Cardiac monitoring and heart failure in advanced breast cancer patients treated with trastuzumab in Ontario. Canada Front Cardiovasc Med. 2022;9: 850674.
Loap P, Orlandi E, De Marzi L, Vitolo V, Barcellini A, Iannalfi A, et al. Cardiotoxicity modelbased patient selection for Hodgkin lymphoma proton therapy. Acta Oncol. 2022;61:979–86.
Ong DS, Aertker RA, Clark AN, Kiefer T, Hughes GC, Harrison JK, et al. Radiation-associated valvular heart disease. J Heart Valve Dis. 2013;22:883–92.
Hershman DL, Till C, Shen S, Wright JD, Ramsey SD, Barlow WE, et al. Association of cardiovascular risk factors with cardiac events and survival outcomes among patients with breast cancer enrolled in SWOG clinical trials. J Clin Oncol. 2018;36:2710–7.
Melendez GC, Jordan JH, D’Agostino RB Jr, Vasu S, Hamilton CA, Hundley WG. Progressive 3-month increase in LV myocardial ECV after anthracycline-based chemotherapy. JACC Cardiovasc Imaging. 2017;10:708–9.
van den Borne SW, Diez J, Blankesteijn WM, Verjans J, Hofstra L, Narula J. Myocardial remodeling after infarction: the role of myofibroblasts. Nat Rev Cardiol. 2010;7:30–7.
Sollini M, Kirienko M, Gelardi F, et al. State-of-the-art of FAPI-PET imaging: a systematic review and meta-analysis. Eur J Nucl Med Mol Imaging. 2021;48:4396–414.
Conti M, Eriksson L. Physics of pure and non-pure positron emitters for PET: a review and a discussion. EJNMMI Phys. 2016;3:8.
Sharma UC, Sonkawade SD, Spernyak JA, Sexton S, Nguyen J, Dahal S, et al. A small peptide Ac-SDKP inhibits radiation-induced cardiomyopathy. Circ Heart Fail. 2018;11: e004867.
Tripp P, Malhotra HK, Javle M, Shaukat A, Russo R, De Boer S, et al. Cardiac function after chemoradiation for esophageal cancer: comparison of heart dose-volume histogram parameters to multiple gated acquisition scan changes. Dis Esophagus. 2005;18:400–5.
Sioka C, Exarchopoulos T, Tasiou I, Tzima E, Fotou N, Capizzello A, et al. Myocardial perfusion imaging with (99 m)Tc-tetrofosmin SPECT in breast cancer patients that received postoperative radiotherapy: a case-control study. Radiat Oncol. 2011;6:151.
Funding
Jinming Yu has received grants from the Academic Promotion Program of Shandong First Medical University (2019ZL002); the Research Unit of Radiation Oncology, Chinese Academy of Medical Sciences (2019RU071); the National Natural Science Foundation of China (81627901, 81972863, and 82030082); and the Natural Science Foundation of Shandong (ZR201911040452). Yuchun Wei has received grants from the Natural Science Foundation of Shandong Province (ZR2021QH008).
Author information
Authors and Affiliations
Contributions
Jinming Yu and Jinli Pei conceived of the study and participated in its design. Yuchun Wei participated in the experiments and drafted the manuscript. Xueting Qin and Yuanyuan Tao are responsible for collecting PET/CT images. Gongsen Zhang carried out the nuclear medicine. Yuhong Sun and Junyan Liu carried out the pathology. Shengnan Xu conducted the statistical analysis. Shijie Wang prepared the [18F]AlF-NOTA-FAPI-04 and [18F]FDG. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was approved by the local ethics committee of Shandong Cancer Hospital and Institute, and the patients gave written, informed consent before the study.
Consent for publication
All authors of the current manuscript meet the specified criteria for authorship and agreed to publish this manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Cardiology.
Supplementary Information
Supplementary Table 1
(DOCX 17 kb)
Supplementary Fig. 1
Dose distributions in the transverse, sagittal, and coronal sections and dose–volume histogram (DVH). (PDF 113 kb)
Supplementary Fig. 2
(A) Long-axis and short-axis of a RIMD rat heart. (B) Short-axis end-systolic and end-diastolic time frames from a cine of the control and RIMD rat (3rd week after radiation) using IG-FLASH scans. (C) Short-axis end-systolic and end-diastolic time frames from a cine of the control and RIMD rat (8th week after radiation) using IG-FLASH scans. (PDF 118 kb)

Supplementary Fig. 3
Change in rat weight throughout the study. (PNG 22 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wei, Y., Sun, Y., Liu, J. et al. Early detection of radiation-induced myocardial damage by [18F]AlF-NOTA-FAPI-04 PET/CT imaging. Eur J Nucl Med Mol Imaging 50, 453–464 (2023). https://doi.org/10.1007/s00259-022-05962-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-05962-y