Abstract
Objective
Uptake of the imaging tracers [18F]AlF-NOTA-FAPI-04 and [18F]FDG varies in some inflammatory lesions, which may result in false-positive findings for malignancy on PET/CT. Our aim was to compare the [18F]AlF-NOTA-FAPI-04 and [18F]FDG PET/CT imaging features of malignant and various inflammatory lung lesions and to analyze their value for differential diagnosis.
Methods
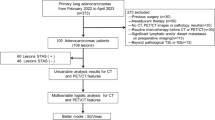
We retrospectively analyzed [18F]AlF-NOTA-FAPI-04 PET/CT scans from 67 cancer patients taken between December 2020 and January 2022, as well as the scans of 32 patients who also underwent [18F]FDG PET/CT imaging. The maximum and mean standardized uptake values (SUVmax and SUVmean, respectively) and lesion-to-background ratio (LBR) were calculated. The predictive capabilities of semiquantitative PET/CT parameters were analyzed by receiver operating characteristic curve analysis.
Results
A total of 70 inflammatory and 37 malignant lung lesions were evaluated by [18F]AlF‑NOTA‑FAPI‑04 PET/CT, and 33 inflammatory and 26 malignant lung lesions also were evaluated by [18F]FDG PET/CT. Inflammatory lesions exhibited lower [18F]AlF-NOTA-FAPI-04 and [18F]FDG uptake compared to malignant lesions, with statistically significant differences in SUVmax, SUVmean, and LBR (all p < 0.001). [18F]AlF-NOTA-FAPI-04 uptake also varied among different types of inflammatory lesions (SUVmax, p = 0.005; SUVmean, p = 0.008; LBR, p < 0.001), with the highest uptake observed in bronchiectasis with infection, followed by postobstructive pneumonia, and the lowest in pneumonia. [18F]FDG uptake was higher in postobstructive pneumonia than in pneumonia (SUVmax, p = 0.009; SUVmean, p = 0.016; LBR, p = 0.004).
Conclusion
[18F]AlF-NOTA-FAPI-04/[18F]FDG PET/CT showed significantly lower uptake in inflammatory lesions than malignancies as well as variation in different types of inflammatory lesions, and thus, may be valuable for distinguishing malignant and various inflammatory findings.
Clinical relevance statement
Our study confirmed that the uptake of [18F]AlF-NOTA-FAPI-04/[18F]FDG PET/CT in inflammatory and malignant lung lesions is different, which is beneficial to distinguish inflammatory and malignant lung lesions in clinic.
Key Points
• Malignant and different inflammatory lung lesions showed varying degrees of uptake of [18F]AlF-NOTA-FAPI-04 and [18F]FDG.
• Inflammatory lung lesions showed significantly less uptake than malignancies, and uptake varied among different types of inflammatory lesions.
• Both types of PET/CT could differentiate malignant and various inflammatory lung findings.





Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the ROC curve
- CAF:
-
Cancer-associated fibroblast
- CT:
-
Computed tomography
- FAP:
-
Fibroblast activation protein
- FAPI:
-
Fibroblast activation protein-specific inhibitor
- FDG:
-
Fluorodeoxyglucose
- fILD:
-
Fibrotic interstitial lung disease
- IQR:
-
Interquartile range
- LBR:
-
Lesion-to-background ratio
- PET:
-
Positron emission tomography
- PMF:
-
Progressive massive fibrosis
- ROC:
-
Receiver operating characteristic
- ROI:
-
Region of interest
- SD:
-
Standard deviation
- SUVmax:
-
Maximum standardized uptake value
- SUVmean:
-
Mean standardized uptake value
References
Davis SL, Nuermberger EL, Um PK et al (2009) Noninvasive pulmonary [18F]-2-fluoro-deoxy-D-glucose positron emission tomography correlates with bactericidal activity of tuberculosis drug treatment. Antimicrob Agents Chemother 53:4879–4884
Goo JM, Im JG, Do KH et al (2000) Pulmonary tuberculoma evaluated by means of FDG PET: findings in 10 cases. Radiology 216:117–121
Demura Y, Tsuchida T, Ishizaki T et al (2003) 18F-FDG accumulation with PET for differentiation between benign and malignant lesions in the thorax. J Nucl Med 44:540–548
Alavi A, Gupta N, Alberini JL et al (2002) Positron emission tomography imaging in nonmalignant thoracic disorders. Semin Nucl Med 32:293–321
de Sostoa J, Fajardo CA, Moreno R, Ramos MD, Farrera-Sal M, Alemany R (2019) Targeting the tumor stroma with an oncolytic adenovirus secreting a fibroblast activation protein-targeted bispecific T-cell engager. J Immunother Cancer 7:19
Giesel FL, Kratochwil C, Lindner T et al (2019) (68)Ga-FAPI PET/CT: biodistribution and preliminary dosimetry estimate of 2 DOTA-containing FAP-targeting agents in patients with various cancers. J Nucl Med 60:386–392
Puré E, Blomberg R (2018) Pro-tumorigenic roles of fibroblast activation protein in cancer: back to the basics. Oncogene 37:4343–4357
Altmann A, Haberkorn U, Siveke J (2021) The latest developments in imaging of fibroblast activation protein. J Nucl Med 62:160–167
Loktev A, Lindner T, Mier W et al (2018) A tumor-imaging method targeting cancer-associated fibroblasts. J Nucl Med 59:1423–1429
Kratochwil C, Flechsig P, Lindner T et al (2019) (68)Ga-FAPI PET/CT: tracer uptake in 28 different kinds of cancer. J Nucl Med 60:801–805
Chen H, Pang Y, Wu J et al (2020) Comparison of [(68)Ga]Ga-DOTA-FAPI-04 and [(18)F] FDG PET/CT for the diagnosis of primary and metastatic lesions in patients with various types of cancer. Eur J Nucl Med Mol Imaging 47:1820–1832
Egger C, Cannet C, Gerard C et al (2017) Effects of the fibroblast activation protein inhibitor, PT100, in a murine model of pulmonary fibrosis. Eur J Pharmacol 809:64–72
Li M, Younis MH, Zhang Y, Cai W, Lan X (2022) Clinical summary of fibroblast activation protein inhibitor-based radiopharmaceuticals: cancer and beyond. Eur J Nucl Med Mol Imaging 49:2844–2868
Milner JM, Kevorkian L, Young DA et al (2006) Fibroblast activation protein alpha is expressed by chondrocytes following a pro-inflammatory stimulus and is elevated in osteoarthritis. Arthritis Res Ther 8:R23
Bauer S, Jendro MC, Wadle A et al (2006) Fibroblast activation protein is expressed by rheumatoid myofibroblast-like synoviocytes. Arthritis Res Ther 8:R171
Tillmanns J, Hoffmann D, Habbaba Y et al (2015) Fibroblast activation protein alpha expression identifies activated fibroblasts after myocardial infarction. J Mol Cell Cardiol 87:194–203
Heckmann MB, Reinhardt F, Finke D et al (2020) Relationship between cardiac fibroblast activation protein activity by positron emission tomography and cardiovascular disease. Circ Cardiovasc Imaging 13:e010628
Nagaraju CK, Dries E, Popovic N et al (2017) Global fibroblast activation throughout the left ventricle but localized fibrosis after myocardial infarction. Sci Rep 7:10801
Brokopp CE, Schoenauer R, Richards P et al (2011) Fibroblast activation protein is induced by inflammation and degrades type I collagen in thin-cap fibroatheromata. Eur Heart J 32:2713–2722
Zhao L, Chen S, Lin L et al (2020) [(68)Ga]Ga-DOTA-FAPI-04 improves tumor staging and monitors early response to chemoradiotherapy in a patient with esophageal cancer. Eur J Nucl Med Mol Imaging 47:3188–3189
Luo Y, Pan Q, Zhang W (2019) IgG4-related disease revealed by (68)Ga-FAPI and (18)F-FDG PET/CT. Eur J Nucl Med Mol Imaging 46:2625–2626
Chen H, Zhao L, Ruan D et al (2021) Usefulness of [(68)Ga]Ga-DOTA-FAPI-04 PET/CT in patients presenting with inconclusive [(18)F]FDG PET/CT findings. Eur J Nucl Med Mol Imaging 48:73–86
Jain S, Self WH, Wunderink RG et al (2015) Community-acquired pneumonia requiring hospitalization among U.S. adults. N Engl J Med 373:415–427
Abers MS, Sandvall BP, Sampath R et al (2016) Postobstructive pneumonia: an underdescribed syndrome. Clin Infect Dis 62:957–961
Burke M, Fraser R (1988) Obstructive pneumonitis: a pathologic and pathogenetic reappraisal. Radiology 166:699–704
Rolston KV (2016) Postobstructive pneumonia in cancer patients. Clin Infect Dis 63:707–708
Valvani A, Martin A, Devarajan A, Chandy D (2019) Postobstructive pneumonia in lung cancer. Ann Transl Med 7:357
Liu H, Yang X, Liu L, Lei L, Wang L, Chen Y (2021) Clinical significance of diffusely increased uptake of (68)Ga-FAPI in thyroid gland. Front Med (Lausanne) 8:782231
Conen P, Pennetta F, Dendl K et al (2022) [(68) Ga]Ga-FAPI uptake correlates with the state of chronic kidney disease. Eur J Nucl Med Mol Imaging 49:3365–3372
Glatting FM, Hoppner J, Liew DP et al (2022) Repetitive early (68)Ga-FAPI PET acquisition comparing (68)Ga-FAPI-02, (68)Ga-FAPI-46, and (68)Ga-FAPI-74: methodologic and diagnostic implications for malignant, inflammatory/reactive, and degenerative lesions. J Nucl Med 63:1844–1851
Dilsizian V, Budde RPJ, Chen W, Mankad SV, Lindner JR, Nieman K (2022) Best practices for imaging cardiac device-related infections and endocarditis: a JACC: Cardiovascular Imaging expert panel statement. JACC Cardiovasc Imaging 15:891–911
Rogers S, Macheda ML, Docherty SE et al (2002) Identification of a novel glucose transporter-like protein-GLUT-12. Am J Physiol Endocrinol Metab 282:E733-738
Shin L, Katz DS, Yung E (2004) Hypermetabolism on F-18 FDG PET of multiple pulmonary nodules resulting from bronchiolitis obliterans organizing pneumonia. Clin Nucl Med 29:654–656
Rohrich M, Leitz D, Glatting FM et al (2022) Fibroblast activation protein-specific PET/CT imaging in fibrotic interstitial lung diseases and lung cancer: a translational exploratory study. J Nucl Med 63:127–133
Choi EK, Park HL, Yoo IR, Kim SJ, Kim YK (2020) The clinical value of F-18 FDG PET/CT in differentiating malignant from benign lesions in pneumoconiosis patients. Eur Radiol 30:442–451
Ohno Y, Kishida Y, Seki S et al (2018) Amide proton transfer-weighted imaging to differentiate malignant from benign pulmonary lesions: comparison with diffusion-weighted imaging and FDG-PET/CT. J Magn Reson Imaging 47:1013–1021
Giesel FL, Kratochwil C, Schlittenhardt J et al (2021) Head-to-head intra-individual comparison of biodistribution and tumor uptake of (68)Ga-FAPI and (18)F-FDG PET/CT in cancer patients. Eur J Nucl Med Mol Imaging 48:4377–4385
Parghane RV, Basu S (2017) Dual–time point 18 F-FDG-PET and PET/CT for differentiating benign from malignant musculoskeletal lesions: opportunities and limitations. Semin Nucl Med 47:373–391
Nakajo M, Jinguji M, Aoki M, Tani A, Sato M, Yoshiura T (2020) The clinical value of texture analysis of dual-time-point (18)F-FDG-PET/CT imaging to differentiate between (18)F-FDG-avid benign and malignant pulmonary lesions. Eur Radiol 30:1759–1769
Funding
This work was funded by the Natural Science Foundation of China (NSFC82203218), the Natural Science Foundation of Shandong Province (ZR2021QH008), and Bethune Charitable Foundation (flzh202116).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Yuchun Wei.
Conflict of interest
The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the institutional review board due to the retrospective and observational nature of the study.
Ethical approval
Institutional review board approval was obtained.
Study subjects or cohorts overlap
No study subjects or cohorts have been previously reported.
Methodology
• retrospective
• diagnostic or prognostic study
• performed at one institution
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiao, K., Qin, X., Fu, S. et al. Value of [18F]AlF-NOTA-FAPI-04 PET/CT for differential diagnosis of malignant and various inflammatory lung lesions: comparison with [18F]FDG PET/CT. Eur Radiol 34, 1948–1959 (2024). https://doi.org/10.1007/s00330-023-10208-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-023-10208-y