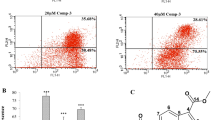
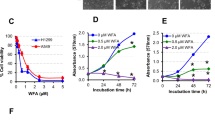
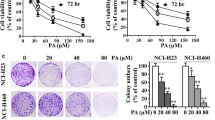
Abstract
Phytocompounds have shown hopeful results in cancer therapy. Piperlongumine (PIP), a naturally derived bioactive alkaloid found in our dietary spice, exhibits promising pharmacological relevance including anticancer activity. This study reconnoitred the anti-lung cancer effect of PIP and the allied mechanisms, in vitro and ex vivo. The cytotoxic, anti-proliferative, and apoptotic effects of PIP on lung cancer cells (LCC) were checked via cell viability, colony formation, cell migration, invasion, comet assay, and various staining techniques. Further, multicellular spheroids assay explored the anti-lung cancer potential of PIP, ex vivo. Preliminary results explored that PIP exerts selective cytotoxic and anti-proliferative effects on LCC by DNA damage and cell cycle arrest. PIP remarkably escalated the cellular and mitochondrial reactive oxygen species (ROS) generation and promoted dissipation of mitochondrial membrane potential (MMP), which triggers activation of caspase-dependent apoptotic pathway in LCC. Mechanistically, PIP showed F-actin deformation mediated significant anti-migratory and anti-invasive activity against LCC. Herein, we also found that F-actin dis-organization modulates the expression of epithelial to mesenchymal transition (EMT) markers and inhibits the expression of stemness marker proteins, like SOX9, CD-133, and CD-44. Moreover, PIP effectively reduced the size of spheroids with strong apoptotic and cytotoxic effects, ex vivo. This has been the first study to discover the high expression of SOX9 supporting the survival of LCC, whereas its inhibition induces higher sensitivity to PIP treatment. This study concludes a newer therapeutic agent (PIP) with promising anticancer activity against LCC by escalating ROS and attenuating MMP, stemness, and EMT.
Graphical Abstract










Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Alagheband Y, Jafari-gharabaghlou D, Imani M, Mousazadeh H, Dadashpour M, Firouzi-Amandi A, Zarghami N (2022) Design and fabrication of a dual-drug loaded nano-platform for synergistic anticancer and cytotoxicity effects on the expression of leptin in lung cancer treatment. J Drug Deliv Sci Technol 73:103389
Belo J, Krishnamurthy M, Oakie A, Wang R (2013) The role of SOX9 transcription factor in pancreatic and duodenal development. Stem Cells Dev 22(22):2935–2943. https://doi.org/10.1089/scd.2013.0106
Bezerra DP, Pessoa C, de Moraes MO, Saker-Neto N, Silveira ER, Costa-Lotufo LV (2013) Overview of the therapeutic potential of piplartine (piperlongumine). Eur J Pharm Sci 48(3):453–463. https://doi.org/10.1016/j.ejps.2012.12.003
Carlos JAEG, Lima K, Coelho-Silva JL, de Melo Alves-Paiva R, Moreno NC, Vicari HP, de Souza Santos FP, Hamerschlak N, Costa-Lotufo LV, Traina F (2020) Reversine exerts cytotoxic effects through multiple cell death mechanisms in acute lymphoblastic leukemia. Cell Oncol 43(6):1191–1201. https://doi.org/10.1007/s13402-020-00551-3
Chen Y, Liu JM, Xiong XX, Qiu XY, Pan F, Liu D, Lan SJ, Jin S, Yu SB, Chen XQ (2015) Piperlongumine selectively kills hepatocellular carcinoma cells and preferentially inhibits their invasion via ROS-ER-MAPKs-CHOP. Oncotarget 6(8):6406. https://doi.org/10.18632/oncotarget.3444
Chen YJ, Kuo CC, Ting LL, Lu LS, Lu YC, Cheng AJ, Lin YT, Chen CH, Tsai JT, Chiou JF (2018) Piperlongumine inhibits cancer stem cell properties and regulates multiple malignant phenotypes in oral cancer. Oncol Lett 15(2):1789–1798. https://doi.org/10.3892/ol.2017.7486
Chen SY, Huang HY, Lin HP, Fang CY (2019) Piperlongumine induces autophagy in biliary cancer cells via reactive oxygen species-activated Erk signaling pathway. Int J Mol Med 44(5):1687–1696. https://doi.org/10.3892/ijmm.2019.4324
Collins A, Møller P, Gajski G, Vodenková S, Abdulwahed A, Anderson D, Bankoglu EE, Bonassi S, Boutet-Robinet E, Brunborg G (2023) Measuring DNA modifications with the comet assay: a compendium of protocols. Nat Protoc 18(3):929–989
Domenici G, Aurrekoetxea-Rodríguez I, Simões BM, Rábano M, Lee SY, San Millán J, Comaills V, Oliemuller E, López-Ruiz JA, Zabalza I (2019) A Sox2–Sox9 signalling axis maintains human breast luminal progenitor and breast cancer stem cells. Oncogene 38(17):3151–3169. https://doi.org/10.1038/s41388-018-0656-7
Dudás J, Ladányi A, Ingruber J, Steinbichler TB, Riechelmann H (2020) Epithelial to mesenchymal transition: a mechanism that fuels cancer radio/chemoresistance. Cells 9(2):428. https://doi.org/10.3390/cells9020428
Han SJ, Kwon S, Kim KS (2021) Challenges of applying multicellular tumor spheroids in preclinical phase. Cancer Cell Int 21:1–19
Haynes J, Srivastava J, Madson N, Wittmann T, Barber DL (2011) Dynamic actin remodeling during epithelial–mesenchymal transition depends on increased moesin expression. Mol Biol Cell 22(24):4750–4764. https://doi.org/10.1091/mbc.E11-02-0119
Javan ES, Lotfi F, Jafari-Gharabaghlou D, Mousazadeh H, Dadashpour M, Zarghami N (2022) Development of a magnetic nanostructure for co-delivery of metformin and silibinin on growth of lung cancer cells: possible action through leptin gene and its receptor regulation. Asian Pac J Cancer Prev: APJCP 23(2):519
Kim BN, Ahn DH, Kang N, Yeo CD, Kim YK, Lee KY, Kim T-J, Lee SH, Park MS, Yim HW (2020) TGF-β induced EMT and stemness characteristics are associated with epigenetic regulation in lung cancer. Sci Rep 10(1):10597
Lamouille S, Xu J, Derynck R (2014) Molecular mechanisms of epithelial–mesenchymal transition. Nat Rev Mol Cell Biol 15(3):178–196. https://doi.org/10.1038/nrm3758
Li S, Yang K, Chen X, Zhu X, Zhou H, Li P, Chen Y, Jiang Y, Li T, Qin X (2021) Simultaneous 2D and 3D cell culture array for multicellular geometry, drug discovery and tumor microenvironment reconstruction. Biofabrication 13(4):045013. https://doi.org/10.1088/1758-5090/ac1ea8
Liu D, Qiu XY, Wu X, Hu DX, Li CY, Yu SB, Pan F, Chen XQ (2017) Piperlongumine suppresses bladder cancer invasion via inhibiting epithelial mesenchymal transition and F-actin reorganization. Biochem Biophys Res Commun 494(1–2):165–172. https://doi.org/10.1016/j.bbrc.2017.10.061
Mittal V (2016) Epithelial mesenchymal transition in aggressive lung cancers. Adv Exp Med Biol 890:37–56. https://doi.org/10.1007/978-3-319-24932-2_3
Nayak N, Basha SA, Tripathi SK, Biswal BK, Mishra M, Sarkar D (2021) Non-cytotoxic and non-genotoxic wear debris of strontium oxide doped (Zirconia Toughened Alumina)(SrO-ZTA) implant for hip prosthesis. Mater Chem Phys 274:125187
Nejati K, Rastegar M, Fathi F, Dadashpour M, Arabzadeh A (2022) Nanoparticle-based drug delivery systems to overcome gastric cancer drug resistance. J Drug Deliv Sci Technol 70:103231
Otsuki Y, Saya H, Arima Y (2018) Prospects for new lung cancer treatments that target EMT signaling. Dev Dyn 247(3):462–472. https://doi.org/10.1002/dvdy.24596
Panda M, Tripathi SK, Biswal BK (2021) SOX9: an emerging driving factor from cancer progression to drug resistance. Biochim Biophys Acta Rev Cancer 1875(2):188517. https://doi.org/10.1016/j.bbcan.2021.188517
Pandey K, Tripathi SK, Panda M, Biswal BK (2020) Prooxidative activity of plumbagin induces apoptosis in human pancreatic ductal adenocarcinoma cells via intrinsic apoptotic pathway. Toxicol in Vitro 65:104788. https://doi.org/10.1016/j.tiv.2020.104788
Phi LTH, Sari IN, Yang Y-G, Lee S-H, Jun N, Kim KS, Lee YK, Kwon HY (2018) Cancer stem cells (CSCs) in drug resistance and their therapeutic implications in cancer treatment. Stem Cells Int 2018:5416923. https://doi.org/10.1155/2018/5416923
Pouremamali F, Pouremamali A, Dadashpour M, Soozangar N, Jeddi F (2022) An update of Nrf2 activators and inhibitors in cancer prevention/promotion. Cell Commun Signal 20(1):100
Rajabi S, Maresca M, Yumashev AV, Choopani R, Hajimehdipoor H (2021) The most competent plant-derived natural products for targeting apoptosis in cancer therapy. Biomolecules 11(4):534. https://doi.org/10.3390/Biom11040534
Ranjan A, Ramachandran S, Gupta N, Kaushik I, Wright S, Srivastava S, Das H, Srivastava S, Prasad S, Srivastava SK (2019) Role of phytochemicals in cancer prevention. Int J Mol Sci 20(20):4981. https://doi.org/10.3390/ijms20204981
Shrivastava S, Kulkarni P, Thummuri D, Jeengar MK, Naidu V, Alvala M, Redddy GB, Ramakrishna S (2014) Piperlongumine, an alkaloid causes inhibition of PI3 K/Akt/mTOR signaling axis to induce caspase-dependent apoptosis in human triple-negative breast cancer cells. Apoptosis 19(7):1148–1164. https://doi.org/10.1007/s10495-014-0991-2
Song B, Zhan H, Bian Q, Gu J (2016) Piperlongumine inhibits gastric cancer cells via suppression of the JAK1, 2/STAT3 signaling pathway. Mol Med Rep 13(5):4475–4480. https://doi.org/10.3892/mmr.2016.5091
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: a cancer Journal for Clinicians 71(3):209–249. https://doi.org/10.3322/caac.21660
Tripathi SK, Biswal BK (2018) Pterospermum acerifolium (L.) wild bark extract induces anticarcinogenic effect in human cancer cells through mitochondrial-mediated ROS generation. Mol Biol Rep 45:2283–2294
Tripathi SK, Biswal BK (2020) Piperlongumine, a potent anticancer phytotherapeutic: Perspectives on contemporary status and future possibilities as an anticancer agent. Pharmacol Res 156:104772. https://doi.org/10.1016/J.Phrs.2020.104772
Tripathi SK, Biswal BK (2021) SOX9 promotes epidermal growth factor receptor-tyrosine kinase inhibitor resistance via targeting β-catenin and epithelial to mesenchymal transition in lung cancer. Life Sci 277:119608
Tripathi SK, Panda M, Biswal BK (2019) Emerging role of plumbagin: cytotoxic potential and pharmaceutical relevance towards cancer therapy. Food Chem Toxicol 125:566–582. https://doi.org/10.1016/j.fct.2019.01.018
Tripathi SK, Rengasamy KR, Biswal BK (2020) Plumbagin engenders apoptosis in lung cancer cells via caspase-9 activation and targeting mitochondrial-mediated ROS induction. Arch Pharmacal Res 43(2):242–256. https://doi.org/10.1007/s12272-020-01221-6
Tripathi SK, Sahoo RK, Biswal BK (2022) SOX9 as an emerging target for anticancer drugs and a prognostic biomarker for cancer drug resistance. Drug Discovery Today 27(9):2541–2550. https://doi.org/10.1016/j.drudis.2022.05.022
Yasamineh S, Gholizadeh O, Kalajahi HG, Yasamineh P, Firouzi-Amandi A, Dadashpour M (2023) Future prospects of natural polymer-based drug delivery systems in combating lung diseases. In: Dureja H, Adams J, Löbenberg R, Andreoli Pinto TdJ, Dua K (eds) Natural polymeric materials based drug delivery systems in lung diseases. Springer, Singapore. https://doi.org/10.1007/978-981-19-7656-8_25
Ye X, Weinberg RA (2015) Epithelial–mesenchymal plasticity: a central regulator of cancer progression. Trends Cell Biol 25(11):675–686. https://doi.org/10.1016/j.tcb.2015.07.012
Funding
This research work is supported by the project (Grant Number: ECR/2016/000792) under the Department of Science and Technology, Science and Engineering Research Board (DST, SERB), New Delhi, India, and Department of Science and Technology, Odisha, India, (Grant no-1201).
Author information
Authors and Affiliations
Contributions
SKT and BKB designed the research. SKT conducted all the experiments and analyzed data. SKT and RKS prepared the manuscript. All authors read and approved the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval
As the in vitro experiments were performed on cell lines, approval from the ethics committee was not required for this study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tripathi, S.K., Sahoo, R.K. & Biswal, B.K. Exposure of piperlongumine attenuates stemness and epithelial to mesenchymal transition phenotype with more potent anti-metastatic activity in SOX9 deficient human lung cancer cells. Naunyn-Schmiedeberg's Arch Pharmacol (2024). https://doi.org/10.1007/s00210-024-02965-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-024-02965-4