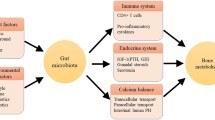
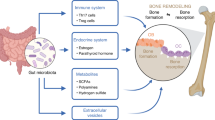
Abstract
Osteoporosis is the prevalent metabolic bone disease characterized by a decrease in bone quantity and/or quality and an increase in skeletal fragility, which increases susceptibility to fractures. Osteoporotic fractures severely affect the patients’ quality of life and mortality. A plethora of evidences have suggested that the alterations in gut microbiome are associated with the changes in bone mass and microstructure. We summarized pre-clinical and clinical studies to elucidate the underlying mechanism of gut microbiota in osteoporosis. Probiotics, prebiotics, and traditional Chinese medicine may reverse the gut microbiota dysbiosis and consequently improve bone metabolism. However, the causality of gut microbiota on bone metabolism need to be investigated more in depth. In the present review, we focused on the potential mechanism of the microbiota-gut-bone axis and the positive therapeutic effect of probiotics, prebiotics, and traditional Chinese medicine on osteoporosis. Overall, the current scientific literatures support that the gut microbiota may be a novel therapeutic target in treatment of osteoporosis and fracture prevention.


Similar content being viewed by others
References
Zhu B, Wang X, Li L (2010) Human gut microbiome: the second genome of human body. Protein Cell 1:718–725. https://doi.org/10.1007/s13238-010-0093-z
Yadav D, Ghosh TS, Mande SS (2016) Global investigation of composition and interaction networks in gut microbiomes of individuals belonging to diverse geographies and age-groups. Gut Pathog 8:17. https://doi.org/10.1186/s13099-016-0099-z
Lozupone CA, Stombaugh JI, Gordon JI, Jansson JK, Knight R (2012) Diversity, stability and resilience of the human gut microbiota. Nature 489:220–230. https://doi.org/10.1038/nature11550
Nishida A, Inoue R, Inatomi O, Bamba S, Naito Y, Andoh A (2018) Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin J Gastroenterol 11:1–10. https://doi.org/10.1007/s12328-017-0813-5
Torres-Fuentes C, Schellekens H, Dinan TG, Cryan JF (2017) The microbiota-gut-brain axis in obesity. Lancet Gastroenterol Hepatol 2:747–756. https://doi.org/10.1016/s2468-1253(17)30147-4
Chen Y, Wang M (2021) New insights of anti-hyperglycemic agents and traditional Chinese medicine on gut microbiota in type 2 diabetes. Drug Des Devel Ther 15:4849–4863. https://doi.org/10.2147/dddt.S334325
Zhou W, Cheng Y, Zhu P, Nasser MI, Zhang X, Zhao M (2020) Implication of gut microbiota in cardiovascular diseases. Oxid Med Cell Longev 2020:5394096. https://doi.org/10.1155/2020/5394096
Pai MV (2017) Osteoporosis Prevention and Management. J Obstet Gynaecol India 67:237–242. https://doi.org/10.1007/s13224-017-0994-3
Hadjidakis DJ, Androulakis II (2006) Bone remodeling. Ann N Y Acad Sci 1092:385–396. https://doi.org/10.1196/annals.1365.035
Karlamangla AS, Burnett-Bowie SM, Crandall CJ (2018) Bone health during the menopause transition and beyond. Obstet Gynecol Clin North Am 45:695–708. https://doi.org/10.1016/j.ogc.2018.07.012
He J, Xu S, Zhang B, Xiao C, Chen Z, Si F, Fu J, Lin X, Zheng G, Yu G, Chen J (2020) Gut microbiota and metabolite alterations associated with reduced bone mineral density or bone metabolic indexes in postmenopausal osteoporosis. Aging (Albany NY) 12:8583–8604. https://doi.org/10.18632/aging.103168
Li C, Huang Q, Yang R, Dai Y, Zeng Y, Tao L, Li X, Zeng J, Wang Q (2019) Gut microbiota composition and bone mineral loss-epidemiologic evidence from individuals in Wuhan, China. Osteoporos Int 30:1003–1013. https://doi.org/10.1007/s00198-019-04855-5
Rettedal EA, Ilesanmi-Oyelere BL, Roy NC, Coad J, Kruger MC (2021) The gut microbiome is altered in postmenopausal women with osteoporosis and osteopenia. JBMR Plus 5:e10452. https://doi.org/10.1002/jbm4.10452
Schoultz I, Keita ÅV (2020) The intestinal barrier and current techniques for the assessment of gut permeability. Cells 9:1909. https://doi.org/10.3390/cells9081909
Otani T, Furuse M (2020) Tight junction structure and function revisited. Trends Cell Biol 30:805–817. https://doi.org/10.1016/j.tcb.2020.08.004
Wang N, Ma S, Fu L (2022) Gut microbiota dysbiosis as one cause of osteoporosis by impairing intestinal barrier function. Calcif Tissue Int 110:225–235. https://doi.org/10.1007/s00223-021-00911-7
Ma S, Wang N, Zhang P, Wu W, Fu L (2021) Fecal microbiota transplantation mitigates bone loss by improving gut microbiome composition and gut barrier function in aged rats. PeerJ 9:e12293. https://doi.org/10.7717/peerj.12293
Schepper JD, Collins F, Rios-Arce ND, Kang HJ, Schaefer L, Gardinier JD, Raghuvanshi R, Quinn RA, Britton R, Parameswaran N, McCabe LR (2020) Involvement of the gut microbiota and barrier function in glucocorticoid-induced osteoporosis. J Bone Miner Res 35:801–820. https://doi.org/10.1002/jbmr.3947
Corrêa-Oliveira R, Fachi JL, Vieira A, Sato FT, Vinolo MA (2016) Regulation of immune cell function by short-chain fatty acids. Clin Transl Immunology 5:e73. https://doi.org/10.1038/cti.2016.17
Wallimann A, Magrath W, Thompson K, Moriarty T, Richards RG, Akdis CA, O’Mahony L, Hernandez CJ (2021) Gut microbial-derived short-chain fatty acids and bone: a potential role in fracture healing. Eur Cell Mater 41:454–470. https://doi.org/10.22203/eCM.v041a29
Coburn JM, Wo L, Bernstein N, Bhattacharya R, Aich U, Bingham CO 3rd, Yarema KJ, Elisseeff JH (2013) Short-chain fatty acid-modified hexosamine for tissue-engineering osteoarthritic cartilage. Tissue Eng Part A 19:2035–2044. https://doi.org/10.1089/ten.TEA.2012.0317
Wu Y, He F, Zhang C, Zhang Q, Su X, Zhu X, Liu A, Shi W, Lin W, Jin Z, Yang H, Lin J (2021) Melatonin alleviates titanium nanoparticles induced osteolysis via activation of butyrate/GPR109A signaling pathway. J Nanobiotechnology 19:170. https://doi.org/10.1186/s12951-021-00915-3
Lv S, Wang Y, Zhang W, Shang H (2022) Trimethylamine oxide: a potential target for heart failure therapy. Heart 108:917–922. https://doi.org/10.1136/heartjnl-2021-320054
Lin H, Liu T, Li X, Gao X, Wu T, Li P (2020) The role of gut microbiota metabolite trimethylamine N-oxide in functional impairment of bone marrow mesenchymal stem cells in osteoporosis disease. Ann Transl Med 8:1009. https://doi.org/10.21037/atm-20-5307
Liu Y, Guo YL, Meng S, Gao H, Sui LJ, Jin S, Li Y, Fan SG (2020) Gut microbiota-dependent Trimethylamine N-Oxide are related with hip fracture in postmenopausal women: a matched case-control study. Aging (Albany NY) 12:10633–10641. https://doi.org/10.18632/aging.103283
Arron JR, Choi Y (2000) Bone versus immune system. Nature 408:535–536. https://doi.org/10.1038/35046196
Zhu L, Hua F, Ding W, Ding K, Zhang Y, Xu C (2020) The correlation between the Th17/Treg cell balance and bone health. Immun Ageing 17:30. https://doi.org/10.1186/s12979-020-00202-z
Zaiss MM, Axmann R, Zwerina J, Polzer K, Gückel E, Skapenko A, Schulze-Koops H, Horwood N, Cope A, Schett G (2007) Treg cells suppress osteoclast formation: a new link between the immune system and bone. Arthritis Rheum 56:4104–4112. https://doi.org/10.1002/art.23138
Yuan FL, Li X, Lu WG, Xu RS, Zhao YQ, Li CW, Li JP, Chen FH (2010) Regulatory T cells as a potent target for controlling bone loss. Biochem Biophys Res Commun 402:173–176. https://doi.org/10.1016/j.bbrc.2010.09.120
Sato K, Suematsu A, Okamoto K, Yamaguchi A, Morishita Y, Kadono Y, Tanaka S, Kodama T, Akira S, Iwakura Y, Cua DJ, Takayanagi H (2006) Th17 functions as an osteoclastogenic helper T cell subset that links T cell activation and bone destruction. J Exp Med 203:2673–2682. https://doi.org/10.1084/jem.20061775
Hsu E, Pacifici R (2018) From osteoimmunology to osteomicrobiology: how the microbiota and the immune system regulate bone. Calcif Tissue Int 102:512–521. https://doi.org/10.1007/s00223-017-0321-0
Talaat RM, Sidek A, Mosalem A, Kholief A (2015) Effect of bisphosphonates treatment on cytokine imbalance between TH17 and Treg in osteoporosis. Inflammopharmacology 23:119–125. https://doi.org/10.1007/s10787-015-0233-4
Furusawa Y, Obata Y, Fukuda S, Endo TA, Nakato G, Takahashi D, Nakanishi Y, Uetake C, Kato K, Kato T, Takahashi M, Fukuda NN, Murakami S, Miyauchi E, Hino S, Atarashi K, Onawa S, Fujimura Y, Lockett T, Clarke JM, Topping DL, Tomita M, Hori S, Ohara O, Morita T, Koseki H, Kikuchi J, Honda K, Hase K, Ohno H (2013) Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells. Nature 504:446–450. https://doi.org/10.1038/nature12721
Luu M, Pautz S, Kohl V, Singh R, Romero R, Lucas S, Hofmann J, Raifer H, Vachharajani N, Carrascosa LC, Lamp B, Nist A, Stiewe T, Shaul Y, Adhikary T, Zaiss MM, Lauth M, Steinhoff U, Visekruna A (2019) The short-chain fatty acid pentanoate suppresses autoimmunity by modulating the metabolic-epigenetic crosstalk in lymphocytes. Nat Commun 10:760. https://doi.org/10.1038/s41467-019-08711-2
Sun P, Zhang C, Huang Y, Yang J, Zhou F, Zeng J, Lin Y (2022) Jiangu granule ameliorated OVX rats bone loss by modulating gut microbiota-SCFAs-Treg/Th17 axis. Biomed Pharmacother 150:112975. https://doi.org/10.1016/j.biopha.2022.112975
Dar HY, Pal S, Shukla P, Mishra PK, Tomar GB, Chattopadhyay N, Srivastava RK (2018) Bacillus clausii inhibits bone loss by skewing Treg-Th17 cell equilibrium in postmenopausal osteoporotic mice model. Nutrition 54:118–128. https://doi.org/10.1016/j.nut.2018.02.013
Dar HY, Shukla P, Mishra PK, Anupam R, Mondal RK, Tomar GB, Sharma V, Srivastava RK (2018) Lactobacillus acidophilus inhibits bone loss and increases bone heterogeneity in osteoporotic mice via modulating Treg-Th17 cell balance. Bone Rep 8:46–56. https://doi.org/10.1016/j.bonr.2018.02.001
Sapra L, Dar HY, Bhardwaj A, Pandey A, Kumari S, Azam Z, Upmanyu V, Anwar A, Shukla P, Mishra PK, Saini C, Verma B, Srivastava RK (2021) Lactobacillus rhamnosus attenuates bone loss and maintains bone health by skewing Treg-Th17 cell balance in Ovx mice. Sci Rep 11:1807. https://doi.org/10.1038/s41598-020-80536-2
Wein MN, Kronenberg HM (2018) Regulation of bone remodeling by parathyroid hormone. Cold Spring Harb Perspect Med 8:a031237. https://doi.org/10.1101/cshperspect.a031237
Iida-Klein A, Lu SS, Kapadia R, Burkhart M, Moreno A, Dempster DW, Lindsay R (2005) Short-term continuous infusion of human parathyroid hormone 1–34 fragment is catabolic with decreased trabecular connectivity density accompanied by hypercalcemia in C57BL/J6 mice. J Endocrinol 186:549–557. https://doi.org/10.1677/joe.1.06270
Walker MD, Silverberg SJ (2018) Primary hyperparathyroidism. Nat Rev Endocrinol 14:115–125. https://doi.org/10.1038/nrendo.2017.104
Jilka RL (2007) Molecular and cellular mechanisms of the anabolic effect of intermittent PTH. Bone 40:1434–1446. https://doi.org/10.1016/j.bone.2007.03.017
Li JY, Yu M, Pal S, Tyagi AM, Dar H, Adams J, Weitzmann MN, Jones RM, Pacifici R (2020) Parathyroid hormone-dependent bone formation requires butyrate production by intestinal microbiota. J Clin Invest 130:1767–1781. https://doi.org/10.1172/jci133473
Khosla S (2020) The microbiome adds to the complexity of parathyroid hormone action on bone. J Clin Invest 130:1615–1617. https://doi.org/10.1172/jci135712
Bennett CN, Ouyang H, Ma YL, Zeng Q, Gerin I, Sousa KM, Lane TF, Krishnan V, Hankenson KD, MacDougald OA (2007) Wnt10b increases postnatal bone formation by enhancing osteoblast differentiation. J Bone Miner Res 22:1924–1932. https://doi.org/10.1359/jbmr.070810
Bennett CN, Longo KA, Wright WS, Suva LJ, Lane TF, Hankenson KD, MacDougald OA (2005) Regulation of osteoblastogenesis and bone mass by Wnt10b. Proc Natl Acad Sci U S A 102:3324–3329. https://doi.org/10.1073/pnas.0408742102
Dixit M, Poudel SB, Yakar S (2021) Effects of GH/IGF axis on bone and cartilage. Mol Cell Endocrinol 519:111052. https://doi.org/10.1016/j.mce.2020.111052
Schwarzer M, Makki K, Storelli G, Machuca-Gayet I, Srutkova D, Hermanova P, Martino ME, Balmand S, Hudcovic T, Heddi A, Rieusset J, Kozakova H, Vidal H, Leulier F (2016) Lactobacillus plantarum strain maintains growth of infant mice during chronic undernutrition. Science 351:854–857. https://doi.org/10.1126/science.aad8588
Yan J, Herzog JW, Tsang K, Brennan CA, Bower MA, Garrett WS, Sartor BR, Aliprantis AO, Charles JF (2016) Gut microbiota induce IGF-1 and promote bone formation and growth. Proc Natl Acad Sci USA 113:E7554-e7563. https://doi.org/10.1073/pnas.1607235113
Balakrishna P, George S, Hatoum H, Mukherjee S (2021) Serotonin pathway in cancer. Int J Mol Sci 22:1268. https://doi.org/10.3390/ijms22031268
Bliziotes M (2010) Update in serotonin and bone. J Clin Endocrinol Metab 95:4124–4132. https://doi.org/10.1210/jc.2010-0861
Lavoie B, Roberts JA, Haag MM, Spohn SN, Margolis KG, Sharkey KA, Lian JB, Mawe GM (2019) Gut-derived serotonin contributes to bone deficits in colitis. Pharmacol Res 140:75–84. https://doi.org/10.1016/j.phrs.2018.07.018
Carsote M, Radoi V, Geleriu A, Mihai A, Ferechide D, Opris D, Paun D, Poiana C (2014) Serotonin and the bone assessment. J Med Life 7:49–53
Wei QS, Chen ZQ, Tan X, Kang LC, Jiang XB, Liang J, He W, Deng WM (2017) Serum serotonin concentration associated with bone mineral density in Chinese postmenopausal women. Scand J Clin Lab Invest 77:40–44. https://doi.org/10.1080/00365513.2016.1247983
Ducy P, Karsenty G (2010) The two faces of serotonin in bone biology. J Cell Biol 191:7–13. https://doi.org/10.1083/jcb.201006123
Yano JM, Yu K, Donaldson GP, Shastri GG, Ann P, Ma L, Nagler CR, Ismagilov RF, Mazmanian SK, Hsiao EY (2015) Indigenous bacteria from the gut microbiota regulate host serotonin biosynthesis. Cell 161:264–276. https://doi.org/10.1016/j.cell.2015.02.047
Reigstad CS, Salmonson CE, Rainey JF 3rd, Szurszewski JH, Linden DR, Sonnenburg JL, Farrugia G, Kashyap PC (2015) Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. Faseb j 29:1395–1403. https://doi.org/10.1096/fj.14-259598
Yan J, Takakura A, Zandi-Nejad K, Charles JF (2018) Mechanisms of gut microbiota-mediated bone remodeling. Gut Microbes 9:84–92. https://doi.org/10.1080/19490976.2017.1371893
Mödder UI, Achenbach SJ, Amin S, Riggs BL, Melton LJ 3rd, Khosla S (2010) Relation of serum serotonin levels to bone density and structural parameters in women. J Bone Miner Res 25:415–422. https://doi.org/10.1359/jbmr.090721
Morris HA, O’Loughlin PD, Anderson PH (2010) Experimental evidence for the effects of calcium and vitamin D on bone: a review. Nutrients 2:1026–1035. https://doi.org/10.3390/nu2091026
Weaver CM (2015) Diet, gut microbiome, and bone health. Curr Osteoporos Rep 13:125–130. https://doi.org/10.1007/s11914-015-0257-0
Wallace TC, Marzorati M, Spence L, Weaver CM, Williamson PS (2017) New frontiers in fibers: innovative and emerging research on the gut microbiome and bone health. J Am Coll Nutr 36:218–222. https://doi.org/10.1080/07315724.2016.1257961
Mineo H, Hara H, Tomita F (2001) Short-chain fatty acids enhance diffusional ca transport in the epithelium of the rat cecum and colon. Life Sci 69:517–526. https://doi.org/10.1016/s0024-3205(01)01146-8
Jones ML, Martoni CJ, Prakash S (2013) Oral supplementation with probiotic L. reuteri NCIMB 30242 increases mean circulating 25-hydroxyvitamin D: a post hoc analysis of a randomized controlled trial. J Clin Endocrinol Metab 98:2944–2951. https://doi.org/10.1210/jc.2012-4262
Wu S, Yoon S, Zhang YG, Lu R, Xia Y, Wan J, Petrof EO, Claud EC, Chen D, Sun J (2015) Vitamin D receptor pathway is required for probiotic protection in colitis. Am J Physiol Gastrointest Liver Physiol 309:G341-349. https://doi.org/10.1152/ajpgi.00105.2015
Black DM, Rosen CJ (2016) Clinical practice. Postmenopausal osteoporosis N Engl J Med 374:254–262. https://doi.org/10.1056/NEJMcp1513724
Ma S, Qin J, Hao Y, Shi Y, Fu L (2020) Structural and functional changes of gut microbiota in ovariectomized rats and their correlations with altered bone mass. Aging (Albany NY) 12:10736–10753. https://doi.org/10.18632/aging.103290
Ma S, Qin J, Hao Y, Fu L (2020) Association of gut microbiota composition and function with an aged rat model of senile osteoporosis using 16S rRNA and metagenomic sequencing analysis. Aging (Albany NY) 12:10795–10808. https://doi.org/10.18632/aging.103293
Das M, Cronin O, Keohane DM, Cormac EM, Nugent H, Nugent M, Molloy C, O’Toole PW, Shanahan F, Molloy MG, Jeffery IB (2019) Gut microbiota alterations associated with reduced bone mineral density in older adults. Rheumatology (Oxford) 58:2295–2304. https://doi.org/10.1093/rheumatology/kez302
Wen K, Tao L, Tao Z, Meng Y, Zhou S, Chen J, Yang K, Da W, Zhu Y (2020) Fecal and serum metabolomic signatures and microbial community profiling of postmenopausal osteoporosis mice model. Front Cell Infect Microbiol 10:535310. https://doi.org/10.3389/fcimb.2020.535310
Wang J, Wang Y, Gao W, Wang B, Zhao H, Zeng Y, Ji Y, Hao D (2017) Diversity analysis of gut microbiota in osteoporosis and osteopenia patients. PeerJ 5:e3450. https://doi.org/10.7717/peerj.3450
Wei M, Li C, Dai Y, Zhou H, Cui Y, Zeng Y, Huang Q, Wang Q (2021) High-throughput absolute quantification sequencing revealed osteoporosis-related gut microbiota alterations in Han Chinese elderly. Front Cell Infect Microbiol 11:630372. https://doi.org/10.3389/fcimb.2021.630372
Eastell R, Szulc P (2017) Use of bone turnover markers in postmenopausal osteoporosis. Lancet Diabetes Endocrinol 5:908–923. https://doi.org/10.1016/s2213-8587(17)30184-5
Stock M, Schett G (2021) Vitamin K-dependent proteins in skeletal development and disease. Int J Mol Sci 22:9328. https://doi.org/10.3390/ijms22179328
Guss JD, Taylor E, Rouse Z, Roubert S, Higgins CH, Thomas CJ, Baker SP, Vashishth D, Donnelly E, Shea MK, Booth SL, Bicalho RC, Hernandez CJ (2019) The microbial metagenome and bone tissue composition in mice with microbiome-induced reductions in bone strength. Bone 127:146–154. https://doi.org/10.1016/j.bone.2019.06.010
Ozaki D, Kubota R, Maeno T, Abdelhakim M, Hitosugi N (2021) Association between gut microbiota, bone metabolism, and fracture risk in postmenopausal Japanese women. Osteoporos Int 32:145–156. https://doi.org/10.1007/s00198-020-05728-y
Halleen JM, Tiitinen SL, Ylipahkala H, Fagerlund KM, Väänänen HK (2006) Tartrate-resistant acid phosphatase 5b (TRACP 5b) as a marker of bone resorption. Clin Lab 52:499–509
Ling CW, Miao Z, Xiao ML, Zhou H, Jiang Z, Fu Y, Xiong F, Zuo LS, Liu YP, Wu YY, Jing LP, Dong HL, Chen GD, Ding D, Wang C, Zeng FF, Zhu HL, He Y, Zheng JS, Chen YM (2021) The association of gut microbiota with osteoporosis is mediated by amino acid metabolism: multiomics in a large cohort. J Clin Endocrinol Metab 106:e3852–e3864. https://doi.org/10.1210/clinem/dgab492
Antoine JM (2010) Probiotics: beneficial factors of the defence system. Proc Nutr Soc 69:429–433. https://doi.org/10.1017/s0029665110001692
Van Loo JA (2004) Prebiotics promote good health: the basis, the potential, and the emerging evidence. J Clin Gastroenterol 38:S70-75. https://doi.org/10.1097/01.mcg.0000128928.99037.e6
Schepper JD, Irwin R, Kang J, Dagenais K, Lemon T, Shinouskis A, Parameswaran N, McCabe LR (2017) Probiotics in gut-bone signaling. Adv Exp Med Biol 1033:225–247. https://doi.org/10.1007/978-3-319-66653-2_11
Whisner CM, Castillo LF (2018) Prebiotics, bone and mineral metabolism. Calcif Tissue Int 102:443–479. https://doi.org/10.1007/s00223-017-0339-3
Kim DE, Kim JK, Han SK, Jang SE, Han MJ, Kim DH (2019) Lactobacillus plantarum NK3 and Bifidobacterium longum NK49 alleviate bacterial vaginosis and osteoporosis in mice by suppressing NF-κB-linked TNF-α expression. J Med Food 22:1022–1031. https://doi.org/10.1089/jmf.2019.4419
Yuan S, Shen J (2021) Bacteroides vulgatus diminishes colonic microbiota dysbiosis ameliorating lumbar bone loss in ovariectomized mice. Bone 142:115710. https://doi.org/10.1016/j.bone.2020.115710
Parvaneh K, Ebrahimi M, Sabran MR, Karimi G, Hwei AN, Abdul-Majeed S, Ahmad Z, Ibrahim Z, Jamaluddin R (2015) Probiotics (Bifidobacterium longum) increase bone mass density and upregulate Sparc and Bmp-2 genes in rats with bone loss resulting from ovariectomy. Biomed Res Int 2015:897639. https://doi.org/10.1155/2015/897639
Britton RA, Irwin R, Quach D, Schaefer L, Zhang J, Lee T, Parameswaran N, McCabe LR (2014) Probiotic L. reuteri treatment prevents bone loss in a menopausal ovariectomized mouse model. J Cell Physiol 229:1822–1830. https://doi.org/10.1002/jcp.24636
Liu H, Gu R, Li W, Zhou W, Cong Z, Xue J, Liu Y, Wei Q, Zhou Y (2019) Lactobacillus rhamnosus GG attenuates tenofovir disoproxil fumarate-induced bone loss in male mice via gut-microbiota-dependent anti-inflammation. Ther Adv Chronic Dis 10:2040622319860653. https://doi.org/10.1177/2040622319860653
Li P, Sundh D, Ji B, Lappa D, Ye L, Nielsen J, Lorentzon M (2021) Metabolic alterations in older women with low bone mineral density supplemented with Lactobacillus reuteri. JBMR Plus 5:e10478. https://doi.org/10.1002/jbm4.10478
Chen C, Dong B, Wang Y, Zhang Q, Wang B, Feng S, Zhu Y (2020) The role of Bacillus acidophilus in osteoporosis and its roles in proliferation and differentiation. J Clin Lab Anal 34:e23471. https://doi.org/10.1002/jcla.23471
Takimoto T, Hatanaka M, Hoshino T, Takara T, Tanaka K, Shimizu A, Morita H, Nakamura T (2018) Effect of Bacillus subtilis C-3102 on bone mineral density in healthy postmenopausal Japanese women: a randomized, placebo-controlled, double-blind clinical trial. Biosci Microbiota Food Health 37:87–96. https://doi.org/10.12938/bmfh.18-006
Porwal K, Pal S, Kulkarni C, Singh P, Sharma S, Singh P, Prajapati G, Gayen JR, Ampapathi RS, Mullick A, Chattopadhyay N (2020) A prebiotic, short-chain fructo-oligosaccharides promotes peak bone mass and maintains bone mass in ovariectomized rats by an osteogenic mechanism. Biomed Pharmacother 129:110448. https://doi.org/10.1016/j.biopha.2020.110448
Tousen Y, Matsumoto Y, Nagahata Y, Kobayashi I, Inoue M, Ishimi Y (2019) Resistant starch attenuates bone loss in ovariectomised mice by regulating the intestinal microbiota and bone-marrow inflammation. Nutrients 11:297. https://doi.org/10.3390/nu11020297
Tanabe K, Nakamura S, Moriyama-Hashiguchi M, Kitajima M, Ejima H, Imori C, Oku T (2019) Dietary fructooligosaccharide and glucomannan alter gut microbiota and improve bone metabolism in senescence-accelerated mouse. J Agric Food Chem 67:867–874. https://doi.org/10.1021/acs.jafc.8b05164
Chen X, Zhang Z, Hu Y, Cui J, Zhi X, Li X, Jiang H, Wang Y, Gu Z, Qiu Z, Dong X, Li Y, Su J (2020) Lactulose suppresses osteoclastogenesis and ameliorates estrogen deficiency-induced bone loss in mice. Aging Dis 11:629–641. https://doi.org/10.14336/ad.2019.0613
(2021) Management of osteoporosis in postmenopausal women: the 2021 position statement of The North American Menopause Society. Menopause 28:973-997 https://doi.org/10.1097/gme.0000000000001831
Greenway F, Liu Z, Yu Y, Gupta A (2011) A clinical trial testing the safety and efficacy of a standardized Eucommia ulmoides Oliver bark extract to treat hypertension. Altern Med Rev 16:338–347
He X, Wang J, Li M, Hao D, Yang Y, Zhang C, He R, Tao R (2014) Eucommia ulmoides Oliv.: ethnopharmacology, phytochemistry and pharmacology of an important traditional Chinese medicine. J Ethnopharmacol 151:78–92. https://doi.org/10.1016/j.jep.2013.11.023
Zhang R, Pan YL, Hu SJ, Kong XH, Juan W, Mei QB (2014) Effects of total lignans from Eucommia ulmoides barks prevent bone loss in vivo and in vitro. J Ethnopharmacol 155:104–112. https://doi.org/10.1016/j.jep.2014.04.031
Zhao X, Wang Y, Nie Z, Han L, Zhong X, Yan X, Gao X (2020) Eucommia ulmoides leaf extract alters gut microbiota composition, enhances short-chain fatty acids production, and ameliorates osteoporosis in the senescence-accelerated mouse P6 (SAMP6) model. Food Sci Nutr 8:4897–4906. https://doi.org/10.1002/fsn3.1779
Mei F, Meng K, Gu Z, Yun Y, Zhang W, Zhang C, Zhong Q, Pan F, Shen X, Xia G, Chen H (2021) Arecanut (Areca catechu L.) Seed polyphenol-ameliorated osteoporosis by altering gut microbiome via LYZ and the immune system in estrogen-deficient rats. J Agric Food Chem 69:246–258. https://doi.org/10.1021/acs.jafc.0c06671
Geng JL, Dai Y, Yao ZH, Qin ZF, Wang XL, Qin L, Yao XS (2014) Metabolites profile of Xian-Ling-Gu-Bao capsule, a traditional Chinese medicine prescription, in rats by ultra performance liquid chromatography coupled with quadrupole time-of-flight tandem mass spectrometry analysis. J Pharm Biomed Anal 96:90–103. https://doi.org/10.1016/j.jpba.2014.03.024
Tang XY, Gao MX, Xiao HH, Dai ZQ, Yao ZH, Dai Y, Yao XS (2021) Effects of Xian-Ling-Gu-Bao capsule on the gut microbiota in ovariectomized rats: metabolism and modulation. J Chromatogr B Analyt Technol Biomed Life Sci 1176:122771. https://doi.org/10.1016/j.jchromb.2021.122771
Ding Z, Hani A, Li W, Gao L, Ke W, Guo X (2020) Influence of a cholesterol-lowering strain Lactobacillus plantarum LP3 isolated from traditional fermented yak milk on gut bacterial microbiota and metabolome of rats fed with a high-fat diet. Food Funct 11:8342–8353. https://doi.org/10.1039/d0fo01939a
Kumari S, Raines JM, Martin JM, Rodriguez JM (2015) Thermal stability of kudzu root (Pueraria Radix) isoflavones as additives to beef patties. J Food Sci Technol 52:1578–1585. https://doi.org/10.1007/s13197-013-1112-x
Suthon S, Jaroenporn S, Charoenphandhu N, Suntornsaratoon P, Malaivijitnond S (2016) Anti-osteoporotic effects of Pueraria candollei var. mirifica on bone mineral density and histomorphometry in estrogen-deficient rats. J Nat Med 70:225–233. https://doi.org/10.1007/s11418-016-0965-5
Urasopon N, Hamada Y, Cherdshewasart W, Malaivijitnond S (2008) Preventive effects of Pueraria mirifica on bone loss in ovariectomized rats. Maturitas 59:137–148. https://doi.org/10.1016/j.maturitas.2008.01.001
Li B, Liu M, Wang Y, Gong S, Yao W, Li W, Gao H, Wei M (2020) Puerarin improves the bone micro-environment to inhibit OVX-induced osteoporosis via modulating SCFAs released by the gut microbiota and repairing intestinal mucosal integrity. Biomed Pharmacother 132:110923. https://doi.org/10.1016/j.biopha.2020.110923
Chen B, Wang L, Li L, Zhu R, Liu H, Liu C, Ma R, Jia Q, Zhao D, Niu J, Fu M, Gao S, Zhang D (2017) Fructus Ligustri Lucidi in osteoporosis: a review of its pharmacology, phytochemistry, pharmacokinetics and safety. Molecules 22:1469. https://doi.org/10.3390/molecules22091469
Zhang Y, Dong XL, Leung PC, Che CT, Wong MS (2008) Fructus ligustri lucidi extract improves calcium balance and modulates the calciotropic hormone level and vitamin D-dependent gene expression in aged ovariectomized rats. Menopause 15:558–565. https://doi.org/10.1097/gme.0b013e31814fad27
Zhang Y, Leung PC, Che CT, Chow HK, Wu CF, Wong MS (2008) Improvement of bone properties and enhancement of mineralization by ethanol extract of Fructus Ligustri Lucidi. Br J Nutr 99:494–502. https://doi.org/10.1017/s0007114507801589
Li L, Chen B, Zhu R, Li R, Tian Y, Liu C, Jia Q, Wang L, Tang J, Zhao D, Mo F, Liu Y, Li Y, Orekhov AN, Brömme D, Zhang D, Gao S (2019) Fructus Ligustri Lucidi preserves bone quality through the regulation of gut microbiota diversity, oxidative stress, TMAO and Sirt6 levels in aging mice. Aging (Albany NY) 11:9348–9368. https://doi.org/10.18632/aging.102376
Xue C, Pan W, Lu X, Guo J, Xu G, Sheng Y, Yuan G, Zhao N, Sun J, Guo X, Wang M, Li H, Du P, An L, Han X (2021) Effects of compound deer bone extract on osteoporosis model mice and intestinal microflora. J Food Biochem 45:e13740. https://doi.org/10.1111/jfbc.13740
Zeng X, Feng Q, Zhao F, Sun C, Zhou T, Yang J, Zhan X (2018) Puerarin inhibits TRPM3/miR-204 to promote MC3T3-E1 cells proliferation, differentiation and mineralization. Phytother Res 32:996–1003. https://doi.org/10.1002/ptr.6034
Hong S, Cha KH, Kwon DY, Son YJ, Kim SM, Choi JH, Yoo G, Nho CW (2021) Agastache rugosa ethanol extract suppresses bone loss via induction of osteoblast differentiation with alteration of gut microbiota. Phytomedicine 84:153517. https://doi.org/10.1016/j.phymed.2021.153517
Hu K, Adachi JD (2019) Glucocorticoid induced osteoporosis. Expert Rev Endocrinol Metab 14:259–266. https://doi.org/10.1080/17446651.2019.1617131
Nemati M, Kamilah H, Huda N, Ariffin F (2015) In vitro calcium availability in bakery products fortified with tuna bone powder as a natural calcium source. Int J Food Sci Nutr 67:535–540. https://doi.org/10.1080/09637486.2016.1179269
Jung WK, Lee BJ, Kim SK (2006) Fish-bone peptide increases calcium solubility and bioavailability in ovariectomised rats. Br J Nutr 95:124–128. https://doi.org/10.1079/bjn20051615
Li J, Yang M, Lu C, Han J, Tang S, Zhou J, Li Y, Ming T, Wang ZJ, Su X (2020) Tuna bone powder alleviates glucocorticoid-induced osteoporosis via coregulation of the NF-κB and Wnt/β-catenin signaling pathways and modulation of gut microbiota composition and metabolism. Mol Nutr Food Res 64:e1900861. https://doi.org/10.1002/mnfr.201900861
Zheng Y, Ren W, Zhang L, Zhang Y, Liu D, Liu Y (2020) A review of the pharmacological action of astragalus polysaccharide. Front Pharmacol 11:349. https://doi.org/10.3389/fphar.2020.00349
Huo J and Sun X (2016) Effect of Astragalus polysaccharides on ovariectomy-induced osteoporosis in mice. Genet Mol Res 15. https://doi.org/10.4238/gmr15049169
Ou L, Wei P, Li M, Gao F (2019) Inhibitory effect of Astragalus polysaccharide on osteoporosis in ovariectomized rats by regulating FoxO3a/Wnt signaling pathway. Acta Cir Bras 34:e201900502. https://doi.org/10.1590/s0102-865020190050000002
Liu J, Liu J, Liu L, Zhang G, Zhou A, Peng X (2020) The gut microbiota alteration and the key bacteria in Astragalus polysaccharides (APS)-improved osteoporosis. Food Res Int 138:109811. https://doi.org/10.1016/j.foodres.2020.109811
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, Y., Chen, Y. The potential mechanism of the microbiota-gut-bone axis in osteoporosis: a review. Osteoporos Int 33, 2495–2506 (2022). https://doi.org/10.1007/s00198-022-06557-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-022-06557-x