Abstract
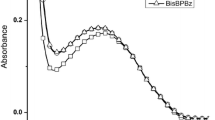
The photolysis kinetics of 4,4’-diazidostilbene stereoisomers in a polymeric matrix (polystyrene films) were studied by the techniques of IR and UV spectroscopy, thin-layer chromatography, and spot test analysis, and primary and secondary amines were determined as the photolysis products, their yield being dependent on the type of stereoisomer. It was shown that, in addition to degradation of the azido group and reactions of the produced nitrenes, stereoisomerization (trans-cis) takes place in the photolysis. As a polystyrene film is formed, the cis-stereoisomers converts into the trans-form
Similar content being viewed by others
References
Karyakina, L.N. and Oleinik, A.V.,Khim. Vys. Energ., 1998, vol. 32, no. 4, p. 277 [High Energy Chem. (Engl. transl.), 1998, vol. 32, no. 4, p. 243].
Karyakina, L.N. and Oleinik, A.V.,Khim. Vys. Energ., 1997, vol. 31, no. 1, p. 37[High Energy Chem. (Engl. transi), 1997, vol. 31, no. 1, p. 34].
Karyakina, L.N., Gubinov, A.V., and Oleinik, A.V.,Khim. Vys. Energ., 1999, vol. 33, no. 6, p. 462 [High Energy Chenu (Engl. transl.), 1999, vol 33, no. 6, p. 401].
Calvin, M. and Alter, H.W.,J. Chem. Phys., 1951, vol. 19, p. 768.
Schulte-Frohlinde, D., Blume, H., and Gusten, H.,J. Phys. Chem., 1962, vol. 66, no. 12, p. 2486.
Calvert, J.G. and Pitts, J.N. Jr.,Photochemistry, New York: Wiley, 1966. Translated under the title Fotokhimiya, Moscow: Mir, 1968.
Treushnikov, V.M., Rubbakh, Ts.N., Zelentsova, N.V., and Oleinik, A.V.,Khim. Vys. Energ., 1983, vol. 17, no. 5, p. 441.
Treushnikov, V.M., Zelentsova, N.V., and Oleinik, A.V.,Zh. Nauchn. Prikl. Fotogr. Kinematogr., 1984, vol. 29, no. 3, p. 226.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Karyakina, L.N., Gubinov, A.V. Photolysis of 4,4’-diazidostilbene in a polymeric matrix. High Energy Chem 34, 256–259 (2000). https://doi.org/10.1007/BF02770894
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02770894