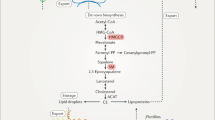
Abstract
The side-chain hydroxylation of cholesterol by specific enzymes produces 24(S)-hydroxycholesterol, 25-hydroxycholesterol, 27-hydroxycholesterol, and other products. These enzymatically formed side-chain oxysterols act as intermediates in the biosynthesis of bile acids and serve as signaling molecules that regulate cholesterol homeostasis. Besides these intracellular functions, an imbalance in oxysterol homeostasis is implicated in pathophysiology. Furthermore, growing evidence reveals that oxysterols affect cell proliferation and cause cell death. This chapter provides an overview of the pathophysiological role of side-chain oxysterols in developing human diseases. We also summarize our understanding of the molecular mechanisms underlying the induction of various forms of cell death by side-chain oxysterols.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Adams CM et al (2004) Cholesterol and 25-hydroxycholesterol inhibit activation of SREBPs by different mechanisms, both involving SCAP and Insigs. J Biol Chem 279:52772–52780. https://doi.org/10.1074/jbc.m410302200
Appukuttan A et al (2013) Oxysterol-induced apoptosis of smooth muscle cells is under the control of a soluble adenylyl cyclase. Cardiovasc Res 99:734–742. https://doi.org/10.1093/cvr/cvt137
Ares MP et al (1997) Ca2+ channel blockers verapamil and nifedipine inhibit apoptosis induced by 25-hydroxycholesterol in human aortic smooth muscle cells. J Lipid Res 38:2049–2061. https://doi.org/10.1016/S0022-2275(20)37135-2
Baek AE et al (2017) The cholesterol metabolite 27 hydroxycholesterol facilitates breast cancer metastasis through its actions on immune cells. Nat Commun 8:864. https://doi.org/10.1038/s41467-017-00910-z
Björkhem I (2009) Are side-chain oxidized oxysterols regulators also in vivo? J Lipid Res 50(Suppl):S213–S218. https://doi.org/10.1194/jlr.r800025-jlr200
Björkhem I, Meaney S (2004) Brain cholesterol: long secret life behind a barrier. Arterioscler Thromb Vasc Biol 24:806–815. https://doi.org/10.1161/01.atv.0000120374.59826.1b
Björkhem I et al (1997) Importance of a novel oxidative mechanism for elimination of brain cholesterol. Turnover of cholesterol and 24(S)-hydroxycholesterol in rat brain as measured with 18O2 techniques in vivo and in vitro. J Biol Chem 272:30178–30184. https://doi.org/10.1074/jbc.272.48.30178
Björkhem I et al (2013) Oxysterols and Parkinson’s disease: evidence that levels of 24S-hydroxycholesterol in cerebrospinal fluid correlates with the duration of the disease. Neurosci Lett 555:102–105. https://doi.org/10.1016/j.neulet.2013.09.003
Björkhem I et al (2019) On the fluxes of side-chain oxidized oxysterols across blood-brain and blood-CSF barriers and origin of these steroids in CSF. J Steroid Biochem Mol Biol 188:86–89. https://doi.org/10.1016/j.jsbmb.2018.12.009
Blanc M et al (2013) The transcription factor STAT-1 couples macrophage synthesis of 25-hydroxycholesterol to the interferon antiviral response. Immunity 38:106–118. https://doi.org/10.1016/j.immuni.2012.11.004
Boussicault L et al (2016) CYP46A1, the rate-limiting enzyme for cholesterol degradation, is neuroprotective in Huntington’s disease. Brain 139:953–970. https://doi.org/10.1093/brain/awv384
Boussicault L et al (2018) CYP46A1 protects against NMDA-mediated excitotoxicity in Huntington’s disease: Analysis of lipid raft content. Biochimie 153:70–79. https://doi.org/10.1016/j.biochi.2018.07.019
Bretillon L et al (2000) Plasma levels of 24S-hydroxycholesterol in patients with neurological diseases. Neurosci Lett 293:87–90. https://doi.org/10.1016/s0304-3940(00)01466-x
Brown J 3rd et al (2004) Differential expression of cholesterol hydroxylases in Alzheimer’s disease. J Biol Chem 279:34674–34681. https://doi.org/10.1074/jbc.M402324200
Brown MS et al (2018) Retrospective on cholesterol homeostasis: the central role of scap. Annu Rev Biochem 87:783–807. https://doi.org/10.1146/annurev-biochem-062917-011852
Cali JJ et al (1991) Mutations in the bile acid biosynthetic enzyme sterol 27-hydroxylase underlie cerebrotendinous xanthomatosis. J Biol Chem 266:7779–7783. https://doi.org/10.1016/S0021-9258(20)89518-0
Cao Q et al (2020) Multiple roles of 25-hydroxycholesterol in lipid metabolism, antivirus process, inflammatory response, and cell survival. Oxid Med Cell Longev 2020:8893305. https://doi.org/10.1155/2020/8893305
Chang TY et al (2009) Acyl-coenzyme A:cholesterol acyltransferases. Am J Physiol Endocrinol Metab 297:E1–E9. https://doi.org/10.1152/ajpendo.90926.2008
Chang TY et al (2017) Cellular cholesterol homeostasis and Alzheimer’s disease. J Lipid Res 58:2239–2254. https://doi.org/10.1194/jlr.r075630
Charman M et al (2014) Oxysterol-binding protein (OSBP)-related protein 4 (ORP4) is essential for cell proliferation and survival. J Biol Chem 289:15705–15717. https://doi.org/10.1074/jbc.M114.571216
Chen W, Chiang JY (2003) Regulation of human sterol 27-hydroxylase gene (CYP27A1) by bile acids and hepatocyte nuclear factor 4alpha (HNF4alpha). Gene 313:71–82. https://doi.org/10.1016/S0378-1119(03)00631-0
Chen S et al (2019) 27-Hydroxycholesterol contributes to lysosomal membrane permeabilization-mediated pyroptosis in co-cultured SH-SY5Y cells and C6 cells. Front Mol Neurosci 12:14. https://doi.org/10.3389/fnmol.2019.00014
Chiba R et al (2023) α-Tocopherol suppresses 24(S)-hydroxycholesterol-induced cell death via inhibition of endoplasmic reticulum membrane disruption. Steroids 189:109136. https://doi.org/10.1016/j.steroids.2022.109136
Cigliano L et al (2019) 24S-hydroxycholesterol affects redox homeostasis in human glial U-87 MG cells. Mol Cell Endocrinol 486:25–33. https://doi.org/10.1016/j.mce.2019.02.013
Costa-Mattioli M, Walter P (2020) The integrated stress response: From mechanism to disease. Science 368:eaat5314. https://doi.org/10.1126/science.aat5314
Cyster JG et al (2014) 25-Hydroxycholesterols in innate and adaptive immunity. Nat Rev Immunol 14:731–743. https://doi.org/10.1038/nri3755
Dai L et al (2021) Cholesterol metabolism in neurodegenerative diseases: molecular mechanisms and therapeutic targets. Mol Neurobiol 58:2183–2201. https://doi.org/10.1007/s12035-020-02232-6
Dias IH et al (2019) Localisation of oxysterols at the sub-cellular level and in biological fluids. J Steroid Biochem Mol Biol 193:105426. https://doi.org/10.1016/j.jsbmb.2019.105426
Diczfalusy U (2013) On the formation and possible biological role of 25-hydroxycholesterol. Biochimie 95:455–460. https://doi.org/10.1016/j.biochi.2012.06.016
Diczfalusy U, Björkhem I (2011) Still another activity by the highly promiscuous enzyme CYP3A4: 25-hydroxylation of cholesterol. J Lipid Res 52:1447–1449. https://doi.org/10.1194/jlr.e017806
Dong Q et al (2020) 25-Hydroxycholesterol promotes vascular calcification via activation of endoplasmic reticulum stress. Eur J Pharmacol 880:173165. https://doi.org/10.1016/j.ejphar.2020.173165
Emanuelsson I, Norlin M (2012) Protective effects of 27- and 24-hydroxycholesterol against staurosporine-induced cell death in undifferentiated neuroblastoma SH-SY5Y cells. Neurosci Lett 525:44–48. https://doi.org/10.1016/j.neulet.2012.07.057
Fellows Maxwell K et al (2019) Oxysterols and apolipoproteins in multiple sclerosis: a 5 year follow-up study. J Lipid Res 60:1190–1198. https://doi.org/10.1194/jlr.m089664
Fourgeux C et al (2009) Primary open-angle glaucoma: association with cholesterol 24S-hydroxylase (CYP46A1) gene polymorphism and plasma 24-hydroxycholesterol levels. Invest Ophthalmol Vis Sci 50:5712–5717. https://doi.org/10.1167/iovs.09-3655
Galluzzi L et al (2018) Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ 25:486–541. https://doi.org/10.1038/s41418-017-0012-4
Gamba P et al (2011) Interaction between 24-hydroxycholesterol, oxidative stress, and amyloid-β in amplifying neuronal damage in Alzheimer’s disease: three partners in crime. Aging Cell 10:403–417. https://doi.org/10.1111/j.1474-9726.2011.00681.x
Gamba P et al (2014) Up-regulation of β-amyloidogenesis in neuron-like human cells by both 24- and 27-hydroxycholesterol: protective effect of N-acetyl-cysteine. Aging Cell 13:561–572. https://doi.org/10.1111/acel.12206
Goldstein JL, Brown MS (1990) Regulation of the mevalonate pathway. Nature 343:425–430. https://doi.org/10.1038/343425a0
Grayaa S et al (2018) Plasma oxysterol profiling in children reveals 24-hydroxycholesterol as a potential marker for Autism Spectrum Disorders. Biochimie 153:80–85. https://doi.org/10.1016/j.biochi.2018.04.026
Griffiths WJ, Wang Y (2022) Cholesterol metabolism: from lipidomics to immunology. J Lipid Res 63:100165. https://doi.org/10.1016/j.jlr.2021.100165
Guidara W et al (2022) Plasma oxysterols in drug-free patients with schizophrenia. J Steroid Biochem Mol Biol 221:106123. https://doi.org/10.1016/j.jsbmb.2022.106123
Han M et al (2020) Therapeutic implications of altered cholesterol homeostasis mediated by loss of CYP46A1 in human glioblastoma. EMBO Mol Med 12:e10924. https://doi.org/10.15252/emmm.201910924
Hartmann H et al (2022) Cholesterol dyshomeostasis in amyotrophic lateral sclerosis: cause, consequence, or epiphenomenon? FEBS J 289:7688–7709. https://doi.org/10.1111/febs.16175
Hetz C (2012) The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat Rev Mol Cell Biol 13:89–102. https://doi.org/10.1038/nrm3270
Hetz C et al (2020) Mechanisms, regulation and functions of the unfolded protein response. Nat Rev Mol Cell Biol 21:421–438. https://doi.org/10.1038/s41580-020-0250-z
Heverin M et al (2004) Changes in the levels of cerebral and extracerebral sterols in the brain of patients with Alzheimer’s disease. J Lipid Res 45:186–193. https://doi.org/10.1194/jlr.m300320-jlr200
Heverin M et al (2005) Crossing the barrier: net flux of 27-hydroxycholesterol into the human brain. J Lipid Res 46:1047–1052. https://doi.org/10.1194/jlr.M500024-JLR200
Hollien J, Weissman JS (2006) Decay of endoplasmic reticulum-localized mRNAs during the unfolded protein response. Science 313:104–107. https://doi.org/10.1126/science.1129631
Honda A et al (2011) Cholesterol 25-hydroxylation activity of CYP3A. J Lipid Res 52:1509–1516. https://doi.org/10.1194/jlr.m014084
Horton JD et al (2003) Combined analysis of oligonucleotide microarray data from transgenic and knockout mice identifies direct SREBP target genes. Proc Natl Acad Sci U S A 100:12027–12032. https://doi.org/10.1073/pnas.1534923100
Hua X et al (1996) Sterol resistance in CHO cells traced to point mutation in SREBP cleavage-activating protein. Cell 87:415–426. https://doi.org/10.1016/s0092-8674(00)81362-8
Janowski BA et al (1996) An oxysterol signalling pathway mediated by the nuclear receptor LXR alpha. Nature 383:728–731. https://doi.org/10.1038/383728a0
Janowski BA et al (1999) Structural requirements of ligands for the oxysterol liver X receptors LXRalpha and LXRbeta. Proc Natl Acad Sci U S A 96:266–271. https://doi.org/10.1073/pnas.96.1.266
Kim I et al (2008) Cell death and endoplasmic reticulum stress: disease relevance and therapeutic opportunities. Nat Rev Drug Discov 7:1013–1030. https://doi.org/10.1038/nrd2755
Kim SM et al (2013) 27-hydroxycholesterol induces production of tumor necrosis factor-alpha from macrophages. Biochem Biophys Res Commun 430:454–459. https://doi.org/10.1016/j.bbrc.2012.12.021
Kim SM et al (2017) 25-Hydroxycholesterol is involved in the pathogenesis of amyotrophic lateral sclerosis. Oncotarget 8:11855–11867. https://doi.org/10.18632/oncotarget.14416
Kim D et al (2022) Pathophysiological role of 27-hydroxycholesterol in human diseases. Adv Biol Regul 83:100837. https://doi.org/10.1016/j.jbior.2021.100837
Kimura Y et al (2018) Tocopherol suppresses 24(S)-hydroxycholesterol-induced cell death via inhibition of CaMKII phosphorylation. Biochimie 153:203–209. https://doi.org/10.1016/j.biochi.2018.07.004
Kölsch H et al (1999) The neurotoxic effect of 24-hydroxycholesterol on SH-SY5Y human neuroblastoma cells. Brain Res 818:171–175. https://doi.org/10.1016/s0006-8993(98)01274-8
Kölsch H et al (2001) Neurotoxicity of 24-hydroxycholesterol, an important cholesterol elimination product of the brain, may be prevented by vitamin E and estradiol-17beta. J Neural Transm 108:475–488. https://doi.org/10.1007/s007020170068
Kölsch H et al (2002) Polymorphism in the cholesterol 24S-hydroxylase gene is associated with Alzheimer’s disease. Mol Psychiatry 7:899–902. https://doi.org/10.1038/sj.mp.4001109
Kölsch H et al (2004) Altered levels of plasma 24S- and 27-hydroxycholesterol in demented patients. Neurosci Lett 368:303–308. https://doi.org/10.1016/j.neulet.2004.07.031
Leoni V, Caccia C (2013) 24S-hydroxycholesterol in plasma: a marker of cholesterol turnover in neurodegenerative diseases. Biochimie 95:595–612. https://doi.org/10.1016/j.biochi.2012.09.025
Leoni V et al (2002) Changes in human plasma levels of the brain specific oxysterol 24S-hydroxycholesterol during progression of multiple sclerosis. Neurosci Lett 331:163–166. https://doi.org/10.1016/s0304-3940(02)00887-x
Leoni V et al (2013) Plasma 24S-hydroxycholesterol correlation with markers of Huntington disease progression. Neurobiol Dis 55:37–43. https://doi.org/10.1016/j.nbd.2013.03.013
Li J et al (2016) Oxysterol binding protein-related protein 8 mediates the cytotoxicity of 25-hydroxycholesterol. J Lipid Res 57:1845–1853. https://doi.org/10.1194/jlr.M069906
Li J et al (2020) Ferroptosis: past, present and future. Cell Death Dis 11:88. https://doi.org/10.1038/s41419-020-2298-2
Li D et al (2022) Brain cell type-specific cholesterol metabolism and implications for learning and memory. Trends Neurosci 45:401–414. https://doi.org/10.1016/j.tins.2022.01.002
Lin JB et al (2018) Oxysterol signatures distinguish age-related macular degeneration from physiologic aging. EBioMedicine 32:9–20. https://doi.org/10.1016/j.ebiom.2018.05.035
Liu J et al (2005) Investigating the allosterism of acyl-CoA:cholesterol acyltransferase (ACAT) by using various sterols: in vitro and intact cell studies. Biochem J 391:389–397. https://doi.org/10.1042/bj20050428
Liu SY et al (2013) Interferon-inducible cholesterol-25-hydroxylase broadly inhibits viral entry by production of 25-hydroxycholesterol. Immunity 38:92–105. https://doi.org/10.1016/j.immuni.2012.11.005
Liu Z et al (2015) Protein kinase R-like ER kinase and its role in endoplasmic reticulum stress-decided cell fate. Cell Death Dis 6:e1822. https://doi.org/10.1038/cddis.2015.183
Liu Y et al (2018) 25-Hydroxycholesterol activates the expression of cholesterol 25-hydroxylase in an LXR-dependent mechanism. J Lipid Res 59:439–451. https://doi.org/10.1194/jlr.m080440
Liu W et al (2021) Dysregulated cholesterol homeostasis results in resistance to ferroptosis increasing tumorigenicity and metastasis in cancer. Nat Commun 12:5103. https://doi.org/10.1038/s41467-021-25354-4
Lund EG et al (2003) Knockout of the cholesterol 24-hydroxylase gene in mice reveals a brain-specific mechanism of cholesterol turnover. J Biol Chem 278:22980–22988. https://doi.org/10.1074/jbc.m303415200
Luo J et al (2020) Mechanisms and regulation of cholesterol homeostasis. Nat Rev Mol Cell Biol 21:225–245. https://doi.org/10.1038/s41580-019-0190-7
Lütjohann D et al (2000) Plasma 24S-hydroxycholesterol (cerebrosterol) is increased in Alzheimer and vascular demented patients. J Lipid Res 41:195–198. https://doi.org/10.1016/S0022-2275(20)32052-6
Marwarha G et al (2010) Leptin reduces the accumulation of Abeta and phosphorylated tau induced by 27-hydroxycholesterol in rabbit organotypic slices. J Alzheimers Dis 19:1007–1019. https://doi.org/10.3233/JAD-2010-1298
Marwarha G et al (2011) The oxysterol 27-hydroxycholesterol regulates α-synuclein and tyrosine hydroxylase expression levels in human neuroblastoma cells through modulation of liver X receptors and estrogen receptors—relevance to Parkinson’s disease. J Neurochem 119:1119–1136. https://doi.org/10.1111/j.1471-4159.2011.07497.x
Marwarha G et al (2013) Gadd153 and NF-κB crosstalk regulates 27-hydroxycholesterol-induced increase in BACE1 and β-amyloid production in human neuroblastoma SH-SY5Y cells. PLoS One 8:e70773. https://doi.org/10.1371/journal.pone.0070773
Maurel M et al (2014) Getting RIDD of RNA: IRE1 in cell fate regulation. Trends Biochem Sci 39:245–254. https://doi.org/10.1016/j.tibs.2014.02.008
Meir K et al (2002) Human sterol 27-hydroxylase (CYP27) overexpressor transgenic mouse model. Evidence against 27-hydroxycholesterol as a critical regulator of cholesterol homeostasis. J Biol Chem 277:34036–34041. https://doi.org/10.1074/jbc.m201122200
Mutemberezi V et al (2016) Oxysterols: From cholesterol metabolites to key mediators. Prog Lipid Res 64:152–169. https://doi.org/10.1016/j.plipres.2016.09.002
Nakazawa T et al (2017) Effect of vitamin E on 24(S)-hydroxycholesterol-induced necroptosis-like cell death and apoptosis. J Steroid Biochem Mol Biol 169:69–76. https://doi.org/10.1016/j.jsbmb.2016.03.003
Nelson ER et al (2013) 27-Hydroxycholesterol links hypercholesterolemia and breast cancer pathophysiology. Science 342:1094–1098. https://doi.org/10.1126/science.1241908
Noguchi N et al (2014) Diverse functions of 24(S)-hydroxycholesterol in the brain. Biochem Biophys Res Commun 446:692–696. https://doi.org/10.1016/j.bbrc.2014.02.010
Noguchi N et al (2015) New aspects of 24(S)-hydroxycholesterol in modulating neuronal cell death. Free Radic Biol Med 87:366–372. https://doi.org/10.1016/j.freeradbiomed.2015.06.036
Nury T et al (2015) Induction of oxiapoptophagy on 158N murine oligodendrocytes treated by 7-ketocholesterol-, 7β-hydroxycholesterol-, or 24(S)-hydroxycholesterol: Protective effects of α-tocopherol and docosahexaenoic acid (DHA; C22:6 n-3). Steroids 99:194–203. https://doi.org/10.1016/j.steroids.2015.02.003
Nury T et al (2021) Oxiapoptophagy: A type of cell death induced by some oxysterols. Br J Pharmacol 178:3115–3123. https://doi.org/10.1111/bph.15173
Oguro H et al (2017) 27-Hydroxycholesterol induces hematopoietic stem cell mobilization and extramedullary hematopoiesis during pregnancy. J Clin Invest 127:3392–3401. https://doi.org/10.1172/jci94027
Ohtsuki S et al (2007) Brain-to-blood elimination of 24S-hydroxycholesterol from rat brain is mediated by organic anion transporting polypeptide 2 (oatp2) at the blood-brain barrier. J Neurochem 103:1430–1438. https://doi.org/10.1111/j.1471-4159.2007.04901.x
Okabe A et al (2013) Adaptive responses induced by 24S-hydroxycholesterol through liver X receptor pathway reduce 7-ketocholesterol-caused neuronal cell death. Redox Biol 2:28–35. https://doi.org/10.1016/j.redox.2013.11.007
Olivier E et al (2017) 25-Hydroxycholesterol induces both P2X7-dependent pyroptosis and caspase-dependent apoptosis in human skin model: New insights into degenerative pathways. Chem Phys Lipids 207:171–178. https://doi.org/10.1016/j.chemphyslip.2017.06.001
Olsen BN et al (2012) Side-chain oxysterols: from cells to membranes to molecules. Biochim Biophys Acta 1818:330–336. https://doi.org/10.1016/j.bbamem.2011.06.014
Pakos-Zebrucka K et al (2016) The integrated stress response. EMBO Rep 17:1374–1395. https://doi.org/10.15252/embr.201642195
Park K, Scott AL (2010) Cholesterol 25-hydroxylase production by dendritic cells and macrophages is regulated by type I interferons. J Leukoc Biol 88:1081–1087. https://doi.org/10.1189/jlb.0610318
Paul SM et al (2013) The major brain cholesterol metabolite 24(S)-hydroxycholesterol is a potent allosteric modulator of N-methyl-D-aspartate receptors. J Neurosci 33:17290–17300. https://doi.org/10.1523/jneurosci.2619-13.2013
Poli G et al (2022) Oxysterols: From redox bench to industry. Redox Biol 49:102220. https://doi.org/10.1016/j.redox.2021.102220
Radhakrishnan A et al (2007) Sterol-regulated transport of SREBPs from endoplasmic reticulum to Golgi: oxysterols block transport by binding to Insig. Proc Natl Acad Sci U S A 104:6511–6518. https://doi.org/10.1073/pnas.0700899104
Reboldi A et al (2014) Inflammation. 25-Hydroxycholesterol suppresses interleukin-1-driven inflammation downstream of type I interferon. Science 345:679–684. https://doi.org/10.1126/science.1254790
Reineke LC, Neilson JR (2019) Differences between acute and chronic stress granules, and how these differences may impact function in human disease. Biochem Pharmacol 162:123–131. https://doi.org/10.1016/j.bcp.2018.10.009
Repa JJ et al (2000) Disruption of the sterol 27-hydroxylase gene in mice results in hepatomegaly and hypertriglyceridemia. Reversal by cholic acid feeding. J Biol Chem 275:39685–39692. https://doi.org/10.1074/jbc.M007653200
Rogers MA et al (2015) Acyl-CoA:cholesterol acyltransferases (ACATs/SOATs): Enzymes with multiple sterols as substrates and as activators. J Steroid Biochem Mol Biol 151:102–107. https://doi.org/10.1016/j.jsbmb.2014.09.008
Rosen H et al (1998) Markedly reduced bile acid synthesis but maintained levels of cholesterol and vitamin D metabolites in mice with disrupted sterol 27-hydroxylase gene. J Biol Chem 273:14805–14812. https://doi.org/10.1074/jbc.273.24.14805
Russell DW (2000) Oxysterol biosynthetic enzymes. Biochim Biophys Acta 1529:126–135. https://doi.org/10.1016/S1388-1981(00)00142-6
Russell DW et al (2009) Cholesterol 24-hydroxylase: an enzyme of cholesterol turnover in the brain. Annu Rev Biochem 78:1017–1040. https://doi.org/10.1146/annurev.biochem.78.072407.103859
Rutkowski DT et al (2006) Adaptation to ER stress is mediated by differential stabilities of pro-survival and pro-apoptotic mRNAs and proteins. PLoS Biol 4:e374. https://doi.org/10.1371/journal.pbio.0040374
Schönknecht P et al (2002) Cerebrospinal fluid 24S-hydroxycholesterol is increased in patients with Alzheimer’s disease compared to healthy controls. Neurosci Lett 324:83–85. https://doi.org/10.1016/s0304-3940(02)00164-7
Schüle R et al (2010) Marked accumulation of 27-hydroxycholesterol in SPG5 patients with hereditary spastic paresis. J Lipid Res 51:819–823. https://doi.org/10.1194/jlr.M002543
Schumacher MM, DeBose-Boyd RA (2021) Posttranslational regulation of HMG CoA reductase, the rate-limiting enzyme in synthesis of cholesterol. Annu Rev Biochem 90:659–679. https://doi.org/10.1146/annurev-biochem-081820-101010
Sekiya M et al (2014) Absence of Nceh1 augments 25-hydroxycholesterol-induced ER stress and apoptosis in macrophages. J Lipid Res 55:2082–2092. https://doi.org/10.1194/jlr.M050864
Sever N et al (2003) Accelerated degradation of HMG CoA reductase mediated by binding of insig-1 to its sterol-sensing domain. Mol Cell 11:25–33. https://doi.org/10.1016/s1097-2765(02)00822-5
Shibata N et al (2013) 25-Hydroxycholesterol activates the integrated stress response to reprogram transcription and translation in macrophages. J Biol Chem 288:35812–35823. https://doi.org/10.1074/jbc.m113.519637
Sodero AO (2021) 24S-hydroxycholesterol: Cellular effects and variations in brain diseases. J Neurochem 157:899–918. https://doi.org/10.1111/jnc.15228
Sun MY et al (2016) Endogenous 24S-hydroxycholesterol modulates NMDAR-mediated function in hippocampal slices. J Neurophysiol 115:1263–1272. https://doi.org/10.1152/jn.00890.2015
Suzuki A et al (2021) Different functions of vitamin E homologues in the various types of cell death induced by oxysterols. Free Radic Biol Med 176:356–365. https://doi.org/10.1016/j.freeradbiomed.2021.10.008
Tabas I, Ron D (2011) Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat Cell Biol 13:184–190. https://doi.org/10.1038/ncb0311-184
Takabe W et al (2016) Esterification of 24S-OHC induces formation of atypical lipid droplet-like structures, leading to neuronal cell death. J Lipid Res 57:2005–2014. https://doi.org/10.1194/jlr.m068775
Tang D et al (2019) The molecular machinery of regulated cell death. Cell Res 29:347–364. https://doi.org/10.1038/s41422-019-0164-5
Umetani M et al (2014) The cholesterol metabolite 27-hydroxycholesterol promotes atherosclerosis via proinflammatory processes mediated by estrogen receptor alpha. Cell Metab 20:172–182. https://doi.org/10.1016/j.cmet.2014.05.013
Urano Y et al (2013) Suppression of amyloid-β production by 24S-hydroxycholesterol via inhibition of intracellular amyloid precursor protein trafficking. FASEB J 27:4305–4315. https://doi.org/10.1096/fj.13-231456
Urano Y et al (2019) 24(S)-Hydroxycholesterol induces ER dysfunction-mediated unconventional cell death. Cell Death Discov 5:113. https://doi.org/10.1038/s41420-019-0192-4
Urano Y et al (2022) Integrated stress response is involved in the 24(S)-hydroxycholesterol-induced unconventional cell death mechanism. Cell Death Discov 8:406. https://doi.org/10.1038/s41420-022-01197-w
Vandenabeele P et al (2022) Pore-forming proteins as drivers of membrane permeabilization in cell death pathways. Nat Rev Mol Cell Biol. https://doi.org/10.1038/s41580-022-00564-w
Vo DK et al (2015) 24(S)-Hydroxycholesterol induces RIPK1-dependent but MLKL-independent cell death in the absence of caspase-8. Steroids 99:230–237. https://doi.org/10.1016/j.steroids.2015.02.007
Wang B, Tontonoz P (2018) Liver X receptors in lipid signalling and membrane homeostasis. Nat Rev Endocrinol 14:452–463. https://doi.org/10.1038/s41574-018-0037-x
Wang H et al (2016a) The cytotoxicity of 27-hydroxycholesterol in co-cultured SH-SY5Y cells and C6 cells. Neurosci Lett 632:209–217. https://doi.org/10.1016/j.neulet.2016.08.056
Wang HL et al (2016b) Cholesterol, 24-hydroxycholesterol, and 27-hydroxycholesterol as surrogate biomarkers in cerebrospinal fluid in mild cognitive impairment and Alzheimer’s disease: a meta-analysis. J Alzheimers Dis 51:45–55. https://doi.org/10.3233/JAD-150734
Wang S et al (2020) Cholesterol 25-Hydroxylase inhibits SARS-CoV-2 and other coronaviruses by depleting membrane cholesterol. EMBO J 39:e106057. https://doi.org/10.15252/embj.2020106057
Wang Y et al (2021) Cholesterol metabolism pathways—are the intermediates more important than the products? FEBS J 288:3727–3745. https://doi.org/10.1111/febs.15727
Warns J et al (2018) 27-hydroxycholesterol decreases cell proliferation in colon cancer cell lines. Biochimie 153:171–180. https://doi.org/10.1016/j.biochi.2018.07.006
Woo SY et al (2022) Role of reactive oxygen species in regulating 27-hydroxycholesterol-induced apoptosis of hematopoietic progenitor cells and myeloid cell lines. Cell Death Dis 13:916. https://doi.org/10.1038/s41419-022-05360-0
Yamanaka K et al (2011) 24(S)-hydroxycholesterol induces neuronal cell death through necroptosis, a form of programmed necrosis. J Biol Chem 286:24666–24673. https://doi.org/10.1074/jbc.M111.236273
Yamanaka K et al (2014) Induction of apoptosis and necroptosis by 24(S)-hydroxycholesterol is dependent on activity of acyl-CoA:cholesterol acyltransferase 1. Cell Death Dis 5:e990. https://doi.org/10.1038/cddis.2013.524
Yan D et al (2008) OSBP-related protein 8 (ORP8) suppresses ABCA1 expression and cholesterol efflux from macrophages. J Biol Chem 283:332–340. https://doi.org/10.1074/jbc.M705313200
Yang WS, Stockwell BR (2016) Ferroptosis: death by lipid peroxidation. Trends Cell Biol 26:165–176. https://doi.org/10.1016/j.tcb.2015.10.014
Yang T et al (2002) Crucial step in cholesterol homeostasis: sterols promote binding of SCAP to INSIG-1, a membrane protein that facilitates retention of SREBPs in ER. Cell 110:489–500. https://doi.org/10.1016/s0092-8674(02)00872-3
Yokoyama S et al (1999) Suppression of rat liver tumorigenesis by 25-hydroxycholesterol and all-trans retinoic acid: differentiation therapy for hepatocellular carcinoma. Int J Oncol 15:565–574. https://doi.org/10.3892/ijo.15.3.565
You JS et al (2020) 25-Hydroxycholesterol induces death receptor-mediated extrinsic and mitochondria-dependent intrinsic apoptosis in head and neck squamous cell carcinoma cells. Anticancer Res 40:779–788. https://doi.org/10.21873/anticanres.14009
Yuan J et al (2016) Roles of caspases in necrotic cell death. Cell 167:1693–1704. https://doi.org/10.1016/j.cell.2016.11.047
Yuan J et al (2019) Necroptosis and RIPK1-mediated neuroinflammation in CNS diseases. Nat Rev Neurosci 20:19–33. https://doi.org/10.1038/s41583-018-0093-1
Zang R et al (2020) Cholesterol 25-hydroxylase suppresses SARS-CoV-2 replication by blocking membrane fusion. Proc Natl Acad Sci U S A 117:32105–32113. https://doi.org/10.1073/pnas.2012197117
Zhang Y et al (2003) Cholesterol is superior to 7-ketocholesterol or 7 alpha-hydroxycholesterol as an allosteric activator for acyl-coenzyme A:cholesterol acyltransferase 1. J Biol Chem 278:11642–11647. https://doi.org/10.1074/jbc.m211559200
Zhang X et al (2019) 27-hydroxycholesterol promotes Aβ accumulation via altering Aβ metabolism in mild cognitive impairment patients and APP/PS1 mice. Brain Pathol 29:558–573. https://doi.org/10.1111/bpa.12698
Zhong W et al (2016) ORP4L facilitates macrophage survival via G-protein-coupled signaling: ORP4L-/- mice display a reduction of atherosclerosis. Circ Res 119:1296–1312. https://doi.org/10.1161/CIRCRESAHA.116.309603
Zuliani G et al (2011) Plasma 24S-hydroxycholesterol levels in elderly subjects with late onset Alzheimer’s disease or vascular dementia: a case-control study. BMC Neurol 11:121. https://doi.org/10.1186/1471-2377-11-121
Acknowledgments
We thank Ren Chiba for his support in making the figures.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Ethics declarations
The authors declare no competing interests.
Funding
This work was supported in part by a JSPS KAKENHI Grant-in-Aid for Scientific Research (C) 16K08254, 19K07093 and 23K10902 to YU.
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Urano, Y., Noguchi, N. (2024). Enzymatically Formed Oxysterols and Cell Death. In: Lizard, G. (eds) Implication of Oxysterols and Phytosterols in Aging and Human Diseases. Advances in Experimental Medicine and Biology, vol 1440. Springer, Cham. https://doi.org/10.1007/978-3-031-43883-7_10
Download citation
DOI: https://doi.org/10.1007/978-3-031-43883-7_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-43882-0
Online ISBN: 978-3-031-43883-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)