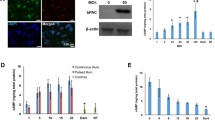
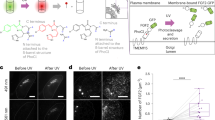
Abstract
Physical cues such as light, heat, or an electrical field can be utilized for traceless, on-demand activation of the expression of a desired therapeutic gene in appropriately engineered cells with excellent spatiotemporal resolution, good inducibility, and simple reversibility. A similar approach can be applied to build a depolarization-based protein secretion system that enables rapid release of a therapeutic protein pre-stored in intracellular vesicles in mammalian cells. Here, we present a protocol to create designer β-cells that exhibit light-controllable rapid release (within 15 min) of a pre-synthesized proinsulin–nanoluciferase construct from vesicular stores. The construct is cleaved extracellularly to afford secreted insulin as a therapeutic protein and nanoluciferase as a reporter molecule. Such posttranslational remote control offers a much faster response than expression-based systems.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Xie M, Fussenegger M (2018) Designing cell function: assembly of synthetic gene circuits for cell biology applications. Nat Rev Mol Cell Biol 19:507–525
Mansouri M, Fussenegger M (2021) Therapeutic cell engineering: designing programmable synthetic genetic circuits in mammalian cells. Protein Cell 13:476. https://doi.org/10.1007/S13238-021-00876-1
Rippe RA, Stefanovic B (2005) Methods for assessing the molecular mechanisms controlling gene regulation. Methods Mol Med 117:141–160. https://doi.org/10.1385/1-59259-940-0:141
Mansouri M, Ray PG, Franko N, Xue S, Fussenegger M (2013) Design of programmable post-translational switch control platform for on-demand protein secretion in mammalian cells. Nucleic Acids Res 1:13–14. https://doi.org/10.1093/NAR/GKAC916
Mansouri M, Fussenegger M (2021) Remote control of mammalian therapeutic designer cells. 53–67. https://doi.org/10.1007/978-3-030-79871-0_2
Mansouri M, Fussenegger M (2021) Synthetic biology-based optogenetic approaches to control therapeutic designer cells. Curr Opin Syst Biol 28:100396. https://doi.org/10.1016/J.COISB.2021.100396
Mansouri M, Fussenegger M (2022) Electrogenetics: bridging synthetic biology and electronics to remotely control the behavior of mammalian designer cells. Curr Opin Chem Biol 68:102151. https://doi.org/10.1016/J.CBPA.2022.102151
Krawczyk K, Xue S, Buchmann P, Charpin-El-Hamri G, Saxena P, Hussherr MD, Shao J, Ye H, Xie M, Fussenegger M (2020) Electrogenetic cellular insulin release for real-time glycemic control in type 1 diabetic mice. Science 368:993–1001. https://doi.org/10.1126/science.aau7187
McCluskey JT, Hamid M, Guo-Parke H, McClenaghan NH, Gomis R, Flatt PR (2011) Development and functional characterization of insulin-releasing human pancreatic beta cell lines produced by electrofusion. J Biol Chem 286:21982–21992. https://doi.org/10.1074/jbc.M111.226795
Burns SM, Vetere A, Walpita D, Dančík V, Khodier C, Perez J, Clemons PA, Wagner BK, Altshuler D (2015) High-throughput luminescent reporter of insulin secretion for discovering regulators of pancreatic beta-cell function. Cell Metab 21:126–137. https://doi.org/10.1016/j.cmet.2014.12.010
Hou JC, Min L, Pessin JE (2009) Insulin granule biogenesis, trafficking and exocytosis. Vitam Horm 80:473. https://doi.org/10.1016/S0083-6729(08)00616-X
Mansouri M, Xue S, Hussherr M-D, Strittmatter T, Camenisch G, Fussenegger M (2021) Smartphone-flashlight-mediated remote control of rapid insulin secretion restores glucose homeostasis in experimental Type-1 diabetes. Small 2101939. https://doi.org/10.1002/SMLL.202101939
Stefanov B-A, Mansouri M, Charpin-El Hamri G, Fussenegger M, Stefanov B, Mansouri M, Fussenegger M, Charpin-El Hamri G (2022) Sunlight-controllable biopharmaceutical production for remote emergency supply of directly injectable therapeutic proteins. Small 18:2202566. https://doi.org/10.1002/SMLL.202202566
Ye H, El BMD, Peng RW, Fussenegger M (2011) A synthetic optogenetic transcription device enhances blood-glucose homeostasis in mice. Science 332:1565–1568. https://doi.org/10.1126/science.1203535
Ausländer S, Fuchs D, Hürlemann S, Ausländer D, Fussenegger M (2016) Engineering a ribozyme cleavage-induced split fluorescent aptamer complementation assay. Nucleic Acids Res 44:94. https://doi.org/10.1093/nar/gkw117
Mansouri M, Lichtenstein S, Strittmatter T, Buchmann P, Fussenegger M (2020) Construction of a multiwell light-induction platform for traceless control of gene expression in mammalian cells. In: Methods in molecular biology. Humana Press Inc., pp 189–199
Acknowledgments
Work in the laboratory of M.F. is financially supported in part through a European Research Council advanced grant (ElectroGene, no. 785800) and in part by the National Centre of Competence in Research (NCCR) for Molecular Systems Engineering as well as the EC Horizon 2020 Framework Programme ENLIGHT.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Mansouri, M., Fussenegger, M. (2024). Posttranslational Remote Control Mediated by Physical Inducers for Rapid Protein Release in Engineered Mammalian Cells. In: Ceroni, F., Polizzi, K. (eds) Mammalian Synthetic Systems. Methods in Molecular Biology, vol 2774. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-3718-0_15
Download citation
DOI: https://doi.org/10.1007/978-1-0716-3718-0_15
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-3717-3
Online ISBN: 978-1-0716-3718-0
eBook Packages: Springer Protocols