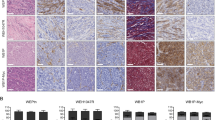
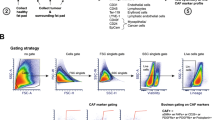
Abstract
Cancer-associated fibroblasts (CAFs) are vital within the tumor ecosystem, regulating tumor growth, dissemination, and response to therapy through crosstalk with tumor cells, infiltrating immune and vascular cells, as well as components of the extracellular matrix (ECM). CAFs have thus emerged as potential therapeutic targets to complement cancer cell–targeted therapies. To study CAF-tumor cell crosstalk ex vivo, robust isolation methods of primary CAFs are required. Here, we present protocols to isolate, expand, and culture two types of fibroblasts: (1) healthy murine mammary gland fibroblasts, a key source of the CAF population in breast tumor models and (2) CAFs derived from syngeneic murine breast tumors. Isolated mammary fibroblasts and CAFs are suitable for use in a variety of downstream cellular and molecular experiments. We expect these methods to be useful to scientists studying the properties of fibroblasts and CAFs and the interaction between CAFs and the various components of the tumor microenvironment (TME).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Hanahan D, Coussens LM (2012) Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 2:309–322
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Kalluri R (2016) The biology and function of fibroblasts in cancer. Nat Rev Cancer 16:582–598
Monteran L, Erez N (2019) The dark side of fibroblasts: cancer-associated fibroblasts as mediators of immunosuppression in the tumor microenvironment. Front Immunol 10:1835
Alkasalias T, Moyano-Galceran L, Aresenian-Henriksson M et al (2018) Fibroblasts in the tumor microenvironment: shield or spear? Int J Mol Sci 19:1532
Sahai E, Astsaturov I, Cukierman E et al (2020) A framework for advancing our understanding of cancer-associated fibroblasts. Nat Rev Cancer 20:174–186
Salimifard S, Masjedi A, Hojjat-Farsangi M et al (2020) Cancer associated fibroblasts as novel promising therapeutic targets in breast cancer. Pathol Res Pract 216:152915
LeBleu VS, Kalluri R (2018) A peek into cancer-associated fibroblasts: origins, functions and translational impact. Dis Model Mech 11:dmm029447
Simon T, Salhia B (2022) Cancer-associated fibroblast subpopulations with diverse and dynamic roles in the tumor microenvironment. Mol Cancer Res 20:183–192
Raz Y, Cohen M, Shani O et al (2018) Bone marrow-derived fibroblasts are a functionally distinct stromal cell population in breast cancer. J Exp Med 215:3075–3093
Agorku DJ, Langhammer A, Heider U et al (2019) CD49b, CD87, and CD95 are markers for activated cancer-associated fibroblasts whereas CD39 Marks quiescent Normal fibroblasts in murine tumor models. Front Oncol 9:716
Seluanov A, Vaidya A, Gorbunova V (2010) Establishing primary adult fibroblast cultures from rodents. J Vis Exp. https://doi.org/10.3791/2033
Sharon Y, Alon L, Glanz S et al (2013) Isolation of normal and cancer-associated fibroblasts from fresh tissues by Fluorescence Activated Cell Sorting (FACS). J Vis Exp. https://doi.org/10.3791/4425
Edelman BL, Redente EF (2018) Isolation and characterization of mouse fibroblasts. Methods Mol Biol 1809:59–67
Saxena M, Kalathur RKR, Neutzner M et al (2018) PyMT-1099, a versatile murine cell model for EMT in breast cancer. Sci Rep 8:12123
Linge C, Green MR, Brooks RF (1989) A method for removal of fibroblasts from human tissue culture systems. Exp Cell Res 185:519–528
Kisselbach L, Merges M, Bossie A et al (2009) CD90 expression on human primary cells and elimination of contaminating fibroblasts from cell cultures. Cytotechnology 59:31–44
Zubeldia-Plazaola A, Ametller E, Mancino M et al (2015) Comparison of methods for the isolation of human breast epithelial and myoepithelial cells. Front Cell Dev Biol 3:32
LeBleu VS, Neilson EG (2020) Origin and functional heterogeneity of fibroblasts. FASEB J 34:3519–3536
Kahounova Z, Kurfurstova D, Bouchal J et al (2018) The fibroblast surface markers FAP, anti-fibroblast, and FSP are expressed by cells of epithelial origin and may be altered during epithelial-to-mesenchymal transition. Cytometry A 93:941–951
Ohlund D, Handly-Santana A, Biffi G et al (2017) Distinct populations of inflammatory fibroblasts and myofibroblasts in pancreatic cancer. J Exp Med 214:579–596
Sebastian A, Hum NR, Martin KA et al (2020) Single-cell transcriptomic analysis of tumor-derived fibroblasts and Normal tissue-resident fibroblasts reveals fibroblast heterogeneity in breast cancer. Cancers (Basel) 12:1307
Bartoschek M, Oskolkov N, Bocci M et al (2018) Spatially and functionally distinct subclasses of breast cancer-associated fibroblasts revealed by single cell RNA sequencing. Nat Commun 9:5150
Friedman G, Levi-Galibov O, David E et al (2020) Cancer-associated fibroblast compositions change with breast cancer progression linking the ratio of S100A4(+) and PDPN(+) CAFs to clinical outcome. Nat Cancer 1:692–708
Guo Q, Bartish M, Goncavles C et al (2021) The MNK1/2-eIF4E Axis supports immune suppression and metastasis in postpartum breast cancer. Cancer Res 81:3876–3889
Preston SEJ, Bartish M, Richard VR et al (2022) Phosphorylation of eIF4E in the stroma drives the production and spatial organisation of collagen type I in the mammary gland. Matrix Biol 111:264–288
Solon J, Levental I, Sengupta K et al (2007) Fibroblast adaptation and stiffness matching to soft elastic substrates. Biophys J 93:4453–4461
Parker MW, Rossi D, Peterson M et al (2014) Fibrotic extracellular matrix activates a profibrotic positive feedback loop. J Clin Invest 124:1622–1635
Financial Support
Work in SDR laboratory is funded by CIHR project grant PJT-178194, and this research was also funded by the Canadian Cancer Society (grant #707140). MB is supported by the international postdoc grant from the Swedish Research Council (VR). JSV is sponsored by an FRQS graduate fellowship.
Author information
Authors and Affiliations
Corresponding authors
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Bartish, M., Smith-Voudouris, J., del Rincón, S.V. (2023). Fibroblast Isolation from Mammary Gland Tissue and Syngeneic Murine Breast Cancer Models. In: Ursini-Siegel, J. (eds) The Tumor Microenvironment. Methods in Molecular Biology, vol 2614. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-2914-7_12
Download citation
DOI: https://doi.org/10.1007/978-1-0716-2914-7_12
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-2913-0
Online ISBN: 978-1-0716-2914-7
eBook Packages: Springer Protocols