Abstract
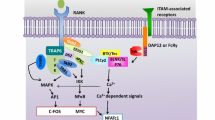
The protein Src homology 3 domain-binding protein 2 (SH3BP2) is a regulator of tumor necrosis factor (TNF)-alpha generation in macrophages and of osteoclast differentiation. SH3BP2 regulates bone marrow monocyte responses to macrophage and osteoclast differentiation signals downstream of the receptors for macrophage colony-stimulating factor and receptor activator of nuclear factor kappaB ligand. SH3BP2 is a potential target for the development of novel anti-TNF-alpha therapeutic interventions as well as a target for suppression of osteoclastogenesis.
Similar content being viewed by others
References
Ueki Y, Lin C, Senoo M, Ebihara T, Agata N, Onji M, Saheki Y, Kawai T, Mukherjee PM, Reichenberger E, Olsen B (2007) Increased myeloid cell responses to M-CSF and RANKL cause bone loss and inflammation in SH3BP2 “cherubism” mice. Cell 128:71–83
LeBras S, Moon C, Foucault I, Breittmayer J, Deckert M (2007) Abl-SH3 binding protein 2,3BP2, interacts with CIN85 and HIP-55. FEBS Lett 581:967–974
Ueki Y, Tiziani V, Santanna C, Fukai N, Maulik C, Garfinkle J, Ninomiya C, doAmaral C, Peters H, Habal M, Rhee-Morris L, Doss JB, Kreiborg S, Olsen BR, Reichenberger E (2001) Mutations in the gene encoding c-Abl-binding protein SH3BP2 cause cherubism. Nat Genet 28:125–126
Ren R, Mayer BJ, Cicchetti P, Baltimore D (1993) Identification of a ten-amino acid proline-rich SH3 binding site. Science 259:1157–1161
Deckert M, Tartare-Deckert S, Hernandez J, Rottapel R, Altman A (1996) Adapter function for the Syk kinases-interacting protein 3BP2 in IL-2 gene activation. Immunity 9:595–605
Foucault I, Liu YC, Bernard A, Deckert M (2003) The chaperone protein 14–3–3 interacts with 3BP2/SH3BP2 and regulates its adapter function. J Biol Chem 278:7146–7153
Maeno K, Sada K, Kyo S, Miah SM, Kawauchi-Kamata K, Qu X, Shi Y, Yamamura H (2003) Adapter protein 3BP2 is a potential ligand of Src homology 2 and 3 domains of Lyn protein-tyrosine kinase. J Biol Chem 278:24912–24920
Foucault I, LeBras S, Charvet C, Moon C, Altman A, Deckert M (2005) The adapter protein 3BP2 associates with VAV guanine nucleotide exchange factors to regulate NFAT activation by the B-cell antigen receptor. Blood 105:1106–1113
Dumitru CD, Ceci JD, Tsatsanis C, Kontoyiannis D, Stamatakis K, Lin JH, Patriotis C, Jenkins NA, Copeland NG, Kollias G, Tsichlis PN (2000) TNF-alpha induction by LPS is regulated posttranscriptionally via a Tpl2/ERK-dependent pathway. Cell 103:1071–1083
Chen G, Dimitriou ID, LaRose J, Ilangumaran S, Yeh W, Doody G, Turner M, Gommerman J, Rottapel R (2007) The 3BP2 adapter protein is required for optimal B-cell activation and thymus-independent type 2 humoral response. Mol Cell Biol 27(8):3109–3122
de la Fuente MA, Kumar L, Lu B, Geha RS (2006) 3BP2 deficiency impairs response of B cells, but not T cells, to antigen receptor ligation. Mol Cell Biol 26:5214–5225
Leitman SA, Yin L, Levine MA (2008) SH3BP2 is an activator of NFAT activity and osteoclastogenesis. Biochem Biophys Res Commun 371(4):644–648
Author information
Authors and Affiliations
Corresponding author
Additional information
SH3BP2 is a critical regulator of macrophage and osteoclast response to M-CSF and RANKL stimulation.
Rights and permissions
About this article
Cite this article
McMahon, M.S., Ueki, Y. SH3BP2 is a Critical Regulator of Macrophage and Osteoclast Response to M-CSF and RANKL Stimulation. HSS Jrnl 5, 49–50 (2009). https://doi.org/10.1007/s11420-008-9091-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11420-008-9091-6