Abstract
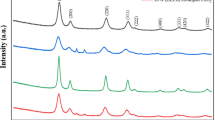
The reactivity of CeO2 is determined by grain size and oxygen vacancies, which can be achieved by doping elements with less oxidation state into CeO2. In this study nanocrystalline Ca-doped CeO2 sol was synthesized from the reaction of hydrate cerium (III) nitrate and calcium nitrate tetrahydrate in alcohol solution after being calcined at 600 °C. X-ray diffraction as well as selected area electron diffraction gave evidence that the synthesized Ca-doped CeO2 samples were well crystalline and had a cubic fluorite structure. TEM observation revealed that Ca-doped CeO2 was composed by nanoparticles with grain size around 8 nm. The Raman spectrum of pure CeO2 consists of a single triple degenerate F2g model characteristic of the fluorite-like structure. In the Ca-doped CeO2 sample, two additional low-intensity Raman bands were detected, thus confirming the formation of the solid solution. The synthesized nanometric powder is expected to be used in solid oxide fuel cells as well as in the catalytic treatment of automobile exhaust fumes.





Similar content being viewed by others
References
Patil KC, Hegde MS, Rattan T, Aruna ST (2008) Chemistry of nanocrystalline oxide materials: combustion synthesis, properties and applications. World Scientific, Singapore
Kiork RE, Othmer DF (1979) Encyclopedia of chemistry and technology, 3rd edn. Wiley, New York
Kirk NB, Wood JV (1994) Br Ceram Trans 93:25
Powell BR, Bloink RL, Erckel CC (1988) J Am Ceram Soc 71:104
Van Herle J, Horita T, Kawata T, Sakai N, Yokokawa H, Dokiya M (1997) J Am Ceram Soc 80:933
Blumenthal RN, Brugner FS, Garnier JE (1973) J Electrochem Soc 120:1230
Mogensen M, Sammes NM, Tompsett GA (2000) Solid State Ion 129:63
Li R, Yabe S, Yamashita M, Momose S, Yoshida S, Yin S, Sato T (2002) Solid State Ionics 151:235
Yabe S, Tsugio S (2002) J Solid State Chem 171:7
Eguchi K, Setoguchi T, Inoue T, Arai H (1992) Solid State Ion 52:165
Maricle DL, Swarr TE, Karavolis S (1992) Solid State Ion 52:173
Lee SW, Kim D, Won HJ, Chung WY (2006) Electron Mater Lett 2:53
Teoh LG (2012) J Sol-Gel Sci Technol 62:47
Yan M, Mori T, Ye F, Ou DR, Zou J, Drennan J (2008) J Euro Ceram Soc 28:2709
Godinho MJ, Goncalves RF, Santos LPS, Varela JA, Longo E, Leite ER (2007) Mater Lett 61:1904
Dikmen S, Shuk P, Greenblatt M, Gocmez H (2002) Solid State Sci 4:585
Lee JS, Lee JS, Choi SC (2005) Mater Lett 59:395
Hassanzadeh-Tabrizi SA, Mazaheri M, Aminzare M, Sadrnezhaad SK (2010) J Alloys Compd 491:499
Phonthammachai N, Rumruangwong M, Gulari E, Jamieson AM, Jitkarnka S, Wongkasemjit S (2004) Colloids Surf A 247:61
de Carolis S, Pascual JL, Pettersson LGM, Baudin M, Wo′jcik M, Palmqvist AEC, Muhammed M, Hermansson K (1999) J Phys Chem B 103:7627
Cullity BD (1978) Elements of X-ray diffraction. Addison-Wesley, Reading
Matta J, Courcot D, Abi-aad E, Aboukays A (2002) Chem Mater 14:4118
Reddy BM, Khan A, Lakshmanan P (2005) J Phys Chem B 109:3355
Lin XM, Li LP, Li GS, Su WH (2001) Mater Chem Phys 69:236
Pu ZY, Lu JQ, Luo MF, Xie YL (2007) J Phys Chem C 111:18695
McBride JR, Hass KC, Poindexter BD, Weber WH (1994) J Appl Phys 76:2435
Martinez-Arias A, Gamarra D, Fernandez-Garcia M, Wang XQ, Hanson JC, Rodriguez JA (2006) J Catal 240:1
Wang S, Wang W, Zuo J, Qian Y (2001) Mater Chem Phys 68:246
Acknowledgments
This work was financially supported by the National Science Council of Taiwan, the Republic of China, grant No. NSC 100-2622-E-020-004-CC3 and NSC 99-2622-E-020-002-CC3, which are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Teoh, L.G., Chiang, G.W. Preparation and characterization of nanocrystalline Ca-doped CeO2 by sol–gel process. J Sol-Gel Sci Technol 64, 530–533 (2012). https://doi.org/10.1007/s10971-012-2885-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-012-2885-5