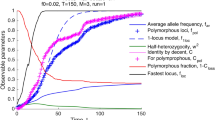
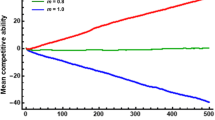
Abstract
In sexual populations, beneficial mutations that occur in different lineages may be recombined into a single lineage. In asexual populations, however, clones that carry such alternative beneficial mutations compete with one another and, thereby, interfere with the expected progression of a given mutation to fixation. From theoretical exploration of such ‘clonal interference’, we have derived (1) a fixation probability for beneficial mutations, (2) an expected substitution rate, (3) an expected coefficient of selection for realized substitutions, (4) an expected rate of fitness increase, (5) the probability that a beneficial mutation transiently achieves polymorphic frequency (≥ 1%), and (6) the probability that a beneficial mutation transiently achieves majority status. Based on (2) and (3), we were able to estimate the beneficial mutation rate and the distribution of mutational effects from changes in mean fitness in an evolving E. coli population.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Barton, N.H., 1993. The probability of fixation of a favoured allele in a subdivided population. Genet. Res. 62: 149–157.
Barton, N.H., 1994. The reduction in fixation probability caused by substitutions at linked loci. Genet. Res. 64: 199–208.
Barton, N.H., 1995. Linkage and the limits to natural selection. Genetics 140: 821–841.
Crow, J.F. & M. Kimura, 1965. Evolution in sexual and asexual populations. Am. Nat. 99: 439–450.
Crow, J.F. & M. Kimura, 1970. An Introduction to Population Genetics Theory. New York: Harper & Row.
Drake, J.W., 1991. A constant rate of spontaneous mutation in DNA-based microbes. Proc. Natl. Acad. Sci. USA 88: 7160–7164.
Elena, S.F., V.S. Cooper & R.E. Lenski, 1996. Punctuated evolution caused by selection of rare beneficial mutations. Science 272: 1802–1804.
Elena, S.F., L. Ekunwe, N. Hajela, S.A. Oden & R.E. Lenski, 1998. Distribution of fitness effects caused by random insertion muta tions in Escherichia coli. Genetica 102/103: 349–358.
Ewens, W.J., 1969. Population Genetics. London: Methuen Press.
Felsenstein, J., 1974. The evolutionary advantage of recombination. Genetics 78: 737–756.
Felsenstein, J., 1988. Sex and the evolution of recombination, pp. 74–86 in The Evolution of Sex, edited by R.E. Michod and B.R. Levin. Sunderland, Mass.: Sinauer Associates.
Fisher, R.A., 1930. The Genetical Theory of Natural Selection. Oxford: Oxford Univ. Press.
Gillespie, J.H., 1981. Mutation rate modification in a random envi ronment. Evolution 35: 468–476.
Gillespie, J.H., 1991. The Causes of Molecular Evolution. Oxford: Oxford Univ. Press.
Haigh, J., 1978. The accumulation of deleterious genes in a popula tion — Muller’s ratchet. Theor. Pop. Biol. 14: 251–267.
Haidane, J.B.S., 1927. The mathematical theory of natural and arti ficial selection. Proc. Camb. Phil. Soc. 23: 838–844.
Holmes, E.C., L.Q. Zhang, P. Simmonds, C.A. Ludlam & A.J.L. Brown, 1992. Convergent and divergent sequence evolution in the surface envelope glycoprotein of human immunodeficiency virus type 1 within a single infected patient. Proc. Natl. Acad. Sci. USA 89: 4835–4839.
Ishii, K., H. Matsuda, Y. Iwasa & A. Sasaki, 1989. Evolutionarily stable mutation rate in a periodically changing environment. Genetics 121: 163–174.
Keightley, P.D., 1991. Genetic variance and fixation probabilities at quantitative trait loci in mutation-selection balance. Genet. Res. 58: 139–144.
Kimura, M., 1979. Model of effectively neutral mutations in which selective constraint is incorporated. Proc. Natl. Acad. Sci. USA 76: 3440–3444.
Leigh, E.G., 1970. Natural selection and mutability. Am. Nat. 104: 301–305.
Lenski, R.E & M. Travisano, 1994. Dynamics of adaptation and diversification: a 10,000-generation experiment with bacterial populations. Proc. Natl. Acad. Sci. USA 91: 6808–6814.
Lenski, R.E., M.R. Rose, S.C. Simpson & S.C. Tadler, 1991. Longterm experimental evolution in Escherichia coli.I.Adaptation and divergence during 2000 generations. Am. Nat. 138: 1315–1341.
Manning, J.T. & D.J. Thompson, 1984. Muller’s ratchet accumula tion of favourable mutations. Acta Biotheor. 33: 219–225.
Maynard Smith, J., 1968. Evolution in sexual and asexual popula tions. Am. Nat. 102: 469–473.
Muller, H.J., 1932. Some genetic aspects of sex. Am. Nat. 8: 118–138.
Muller, H.J., 1964. The relation of recombination to mutational advance. Mutat. Res. 1: 2–9.
Otto, S.P. & M.C. Whitlock, 1997. The probability of fixation in populations of changing size. Genetics 146: 723–733.
Pamilo, P., M. Nei & W. Li, 1987. Accumulation of mutations in sexual and asexual populations. Genet. Res. 49: 135–146.
Peck, J.R., 1994. A ruby in the rubbish: beneficial mutations, delete rious mutations and the evolution of sex. Genetics 137: 597–606.
Peck, J.R., G. Barreau & S.C. Heath, 1997. Imperfect genes, Fisherian mutation and the evolution of sex. Genetics 145: 1171–1199.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1998 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Gerrish, P.J., Lenski, R.E. (1998). The fate of competing beneficial mutations in an asexual population. In: Woodruff, R.C., Thompson, J.N. (eds) Mutation and Evolution. Contemporary Issues in Genetics and Evolution, vol 7. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-5210-5_12
Download citation
DOI: https://doi.org/10.1007/978-94-011-5210-5_12
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-010-6193-3
Online ISBN: 978-94-011-5210-5
eBook Packages: Springer Book Archive