Abstract
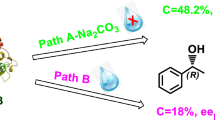
The enzymatic hydrolysis of a series of carboxylic esters by carboxylesterase NP has been investigated in order to determine the scope and limitations of this enzyme. α-Substituted propionates were hydrolysed with high enantioselectivity when an aromatic moiety was part of the α-substituent. Enantioselective hydrolysis could be accomplished with several α-arylpropionates, α-aryloxypropionates and N-arylalanine esters. The propionate esters yielded the propionic acids as (S)-enantiomers, whereas the alanine esters yielded the (R)- or (D)-enantiomers. Without α-aryl substituent the enzymatic hydrolysis of the propionates occurred at a lower rate without acceptable enantioselectivity.
In addition to α-substituted propionates only a few other esters were hydrolysed with high enantioselectivity by carboxylesterase NP, such as some prochiral di-substituted malonates, 1-Phenylethyl acetate was the only substrate with the chirality in the alcohol part of the ester that was found to be hydrolysed enantioselectively.
Carboxylesterase NP proves to be a powerful enzyme for the kinetic resolution of propionate esters with an aromatic ring containing α-substituent.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Literature
Davies, H.G.; Green, R.H.; Kelly, D.R.; Roberts, S.M. Biotransformations in Preparative Organic Chemistry; Katritzky, A.R.; Mett-Cohn, O.; Rees, C.W., Eds.; Academic Press: London, 1989.
Cheetham, P.S.J. Enzyme Microb. Technol. 1987, 9, 194–213.
Li-Ming-Zhu; Tedford, M.C. Tetrahedron 1990, 46, 6587–6611.
Gardossi, L.; Bianchi, D.; Klibanov, A.M. J. Am. Chem. Soc. 1991, 113, 6328–6329.
Burgess, K.; Jennings, L.D. J. Am. Chem. Soc. 1991, 113, 6129–6139.
Klibanov, A.M. Acc. Chem. Res. 1990, 23, 114–120.
Kalaritis, P.; Regenye, R.W.; Partridge, J.J.; Coffen, D.L. J. Org. Chem. 1990, 55, 812–815.
Gastel, F.J.C. van; Klunder, A.J.M.; Zwanenburg, B. Recl. Trav. Chim. Pays-Bas 1991, 110, 175–184.
Ariens, A.J. Eur. J. Clin. Pharmacol 1984, 26, 663.
Kooreman, H.J. Synthesis of biologically active natural products 3, 52–59.
Mutsaers, J.H.G.M.; Kooreman, H.J. Recl. Trav. Chim. Bays-Bas 1991, 110, 185–188.
Harrison, I.T.; Lewis, B.; Nelson, P.; Rooks, W.; Roszkowski, A.; Tamalonis, A.; Fried, J.H. J. Med. Chem. 1970, 13, 203.
Patent Syntex USP 3637767.
Baker, J.T. Application note No.2.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1992 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Smeets, J.W.H., Kieboom, A.P.G. (1992). Enzymatic enantioselective ester hydrolysis by carboxylesterase NP. In: Servi, S. (eds) Microbial Reagents in Organic Synthesis. NATO ASI Series, vol 381. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-2444-7_22
Download citation
DOI: https://doi.org/10.1007/978-94-011-2444-7_22
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-010-5078-4
Online ISBN: 978-94-011-2444-7
eBook Packages: Springer Book Archive