Abstract
In various experiments, the freshwater snail species Lymnaea peregra (Müll.), Physa fontinalis (L.), Valvata piscinalis (Müll.) and Bithynia tentaculata (L.) significantly removed periphyton from glass slides, while the two tested crustacean species, Asellus aquaticus (L.) and Gammarus pulex (L.), did not. B. tentaculata removed similar amounts of periphyton accumulated in the field as it did of laboratory-cultured periphyton.
Removal rates ranged from 0.1–2.2 mg animal−1 day−1 ash-free dry weight (afdw) for different species, different temperatures and differing amounts of initially available periphyton. Differences in removal rate per individual snail between and within species could largely be accounted for by differences in snail biomass and activity. A significant effect of temperature on periphyton removal and activity was observed in only one out of four experiments. Apparently, an efficient temperature compensation mechanism is present in the tested species. The four species differed in grazing trail width, linear velocity, and also in the density and irradiance transmittance of the periphyton that is left in the grazing trail. Thus, differences in periphyton removal capacities of field populations of the different species are to be expected.
With similar periphyton density and taxonomic composition, velocity nor activity of L. peregra was influenced by the type of substratum (glass slides versus plant surface of Potamogeton pectinatus L.). Taxonomic composition of the periphyton did influence velocity and activity. Higher periphyton density resulted in increased periphyton removal in L. peregra (up to a plateau at 0.2 mg cm−2 afdw) and P. fontinalis but not in B. tentaculata and V. piscinalis.
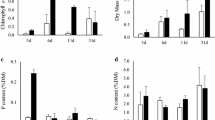
In a growth experiment lasting 9 weeks, the density of periphyton on P. pectinatus was reduced in the presence of L. peregra. Taxonomic composition of periphyton on grazed plants differed from that on control plants: dominance by tightly adhering unicellular green algae in the presence of snails versus filamentous Cyanobacteria in their absence. Ungrazed control plants with a higher periphyton cover produced more leaf material than plants with snails, at the expense of the tubers. In a second growth experiment with P. pectinatus in the presence of the grazers B. tentaculata, V. piscinalis and juvenile L. peregra, all snail species significantly reduced periphyton density. Contrary to the previous experiment, however, no differences in plant growth were apparent. This was due to a lower periphyton and phytoplankton density in the ungrazed controls as compared to the previous experiment. Irradiance reaching the plants in the first growth experiment was close to the compensation point for P. pectinatus of this age and acclimized to this irradiance, while in the second experiment net photosynthetic rates were estimated to be about twice as high. This resulted in a fivefold difference in newly formed biomass after 9 weeks.
The three tested species clearly showed different activity patterns. L. peregra remained active throughout the experiment and was present on the plants and aquarium walls in considerable numbers. V. piscinalis was present on the macrophytes for about one month, during which oviposition took place. Subsequent post-breeding mortality rapidly removed all adults of this semelparous species. B. tentaculata showed a somewhat intermediate pattern. In the first part of the experiment, densities on plants were similar to those of L. peregra, but after oviposition a large proportion of the animals moved to the sediment and burrowed themselves, whilst large-scale mortality did not take place.
Two methods were developed to estimate irradiance transmittance improvement due to periphyton removal. These methods were evaluated for different species and conditions. On glass slides, transmittance of the remaining periphyton after a period of exposure to grazing animals can be estimated without significant error from its biomass and a hyperbola relating irradiance attenuance (1-transmittance) to biomass (method A), provided that at least 60% of the biomass is removed by the grazers. Else, more detailed measurements of trail width, linear velocity and attenuance in grazing trails of individual snails are to be preferred (method B). When the two methods were evaluated on the basis of periphyton growth and removal rate on P. pectinatus in the first growth experiment, method B appeared to estimate the removed amounts more accurately.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Belcher, H.& Swale, E. 1976. A Beginner’s Guide to Freshwater Algae. Culture Centre of Algae and Protozoa, Cambridge, U.K., 47 pp.
Borum, J. 1985. Development of epiphytic communities on eelgrass (Zostera marina) along a nutrient gradient in a Danish estuary. Mar. Biol. 87: 211–218.
Bovbjerg, R.V. 1968. Responses to food in Lymnaeid snails. Physiol. Zool. 41: 412–423.
Brönmark, C. 1985. Interactions between macrophytes, epiphytes and herbivores: an experimental approach. Oikos 45: 26–30.
Brönmark, C. 1988. Effects of vertebrate predation on freshwater gastropods: an exclosure experiment. Hydrobiologia 169: 363–370.
Brown, K.M.& DeVries D.R. 1985. Predation and the distribution and abundance of a pulmonate snail. Oecologia 66: 93–99.
Calow, P. 1970. Studies on the natural diet of Lymnaea pereger obtusa (Kobelt) and its possible ecological implications. Proc. malac. Soc. Lond. 39: 203–215.
Calow, P. 1973a. Field observations and laboratory experiments on the general food requirements of two species of freshwater snail, Planorbis coniortus (Linn.) and Ancylus fluviatilis Müll. Proc. malac. Soc. Lond. 40: 483–489.
Calow, P. 1973b. On the regulatory nature of individual growth: some observations from freshwater snails. J. Zool. Lond. 170: 415–428.
Calow, P. 1975. The feeding strategies of two freshwater gastropods, Ancylus fluviatilis Müll, and Planorbis contortus Linn. (Pulmonata), in terms of ingestion rates and absorption efficiencies. Oecologia 20: 33–49.
Calow, P. 1978. The evolution of life-cycle strategies in freshwater gastropods. Malacologia 17: 351–364.
Calow, P. 1981. Adaptational aspects of growth and reproduction in Lymnaea peregra (Gastropoda: Pulmonata) from exposed and sheltered habitats. Malacologia 21: 5–13.
Calow, P.& Calow L.J. 1975. Cellulase activity and niche separation in freshwater gastropods. Nature 255: 478–480.
Cambridge, M.C., Chiffmgs, A.W., Brittan, C., Moore, L.,& McCombe A.J. 1986. The loss of seagrass in Cockburn Sound, Western Australia II. Possible causes of seagrass decline. Aquat. Bot. 24: 269–285.
Cattaneo, A. 1983. Grazing on epiphytes. Limnol. Oceanogr. 28: 124–132.
Cattaneo, A. 1987. Periphyton in lakes of different trophy. Can. J. Fish. Aquat. Sci. 44: 296–303.
Cattaneo, A.& Kalff, J. 1978. Seasonal changes in the epiphytic community of natural and artificial macrophytes in Lake Memphremagog (Que&Vt). Hydrobiologia 60: 135–144.
Cleland, D.M. 1954. A study of the habits of Valvata piscinalis (Müller) and the structure and function of the alimentary canal and reproductive system. Proc. malac. Soc. Lond. 30: 167–203.
Covich, A.P.& Knezevich, B. 1978. Size-selective predation by fish on thin-shelled gastropods (Lymnaea); the significance of floating vegetation (Trapa) as a physical refuge. Verh. internat. Verein. Limnol. 20: 2172–2177.
Cuker, B.E. 1983. Grazing and nutrient interactions in controlling the activity and composition of the epilithic algal community of an arctic lake. Limnol. Oceanogr. 28: 133–141.
Dussart, G.B.J. 1979. Life cycles and distribution of the aquatic gastropod molluscs Bithynia tentaculata (L.), Gyraulus albus (Muller), Planorbis planorbis (L.) and Lymnaea peregra (Muller) in relation to water chemistry. Hydrobiologia 67: 223–239.
Fretter, V.& Graham, A. 1962. British Prosobranch Molluscs. Ray Society, London, 755 pp.
Gilinsky, E. 1984. The role of fish predation and spatial heterogeneity in determining benthic community structure. Ecology 65: 455–468.
Gons, H.J. 1982. Structural and functional characteristics of epiphyton and epipelon in relation to their distribution in Lake Vechten. Hydrobiologia 95: 79–114.
Hassel, M.P. 1978. The Dynamics of Arthropod Predator Prey Systems. Monographs in Population Biology 13. Princeton University Press, Princeton, New Jersey, U.S.A., 237 pp.
Hootsmans, M.J.M.& Vermaat, J.E. 1985. The effect of periphyton grazing by three epifaunal species on the growth of Zosiera marina L. under experimental conditions. Aquat. Bot. 22: 83–88.
Howard, R.K.& Short, F.T. 1986. Seagrass growth and survivorship under the influence of epiphyte grazers. Aquat. Bot. 24: 287–302.
Hunter, D.R. 1980. Effects of grazing on the quantity and quality of freshwater aufwuchs. Hydrobiologia 69: 251–259.
Hylleberg, J. 1975. The effect of salinity and temperature on egestion in mud snails (Gastropoda: Hydrobiidae), a study on niche overlap. Oecologia 21: 279–289.
Irvine, K., Moss, B.& Balls, H. 1989. The loss of submerged plants with eutrophication. II. Relationships between fish and Zooplankton in a set of experimental ponds, and conclusions. Freshwat. Biol. 22: 89–107.
Kairesalo, T. 1984. The seasonal succession of epiphytic communities within an Equisetum fluviatile L. stand in Lake Pääjärvi, southern Finland. Int. Rev. ges. Hydrobiol. 69: 475–505.
Kairesalo, T.& Koskimies, I. 1987. Grazing by oligochaetes and snails on epiphytes. Freshwat. Biol. 17: 317–324.
Kirk, J.T.O. 1983. Light and Photosynthesis in Aquatic Ecosystems. Cambridge University Press, Cambridge, U.K., 401 pp.
Krebs, J.R.& Davies, N.B. 1984. Behavioural Ecology: An Evolutionary Approach. Second edition. Blackwell, Oxford, U.K., 514 pp.
Lammens, E.H.R.R. 1989. Causes and consequences of the success of Bream in Dutch eutrophic lakes. Hydrobiol. Bull. 23: 11–18.
Lodge, D.M. 1985. Macrophyte gastropod associations: observations and experiments on macrophyte choice by gastropods. Freshwat. Biol. 15: 695–708.
Lodge, D.M. 1986. Selective grazing on periphyton: a determinant of freshwater gastropod microdistributions. Freshwat. Biol. 16: 831–841.
Lodge, D.M.& Kelly, P. 1985. Habitat disturbance and the stability of freshwater gastropod populations. Oecologia 68: 111–117.
Lodge, D.M., Brown, K.M., Klosiewski, S.P., Stein, R.A., Covich, A.P., Leathers, B.K.& Brönmark, C. 1987. Distribution of freshwater snails: spatial scale and the relative importance of physicochemical and biotic factors. Amer. Malac. Bull. 5: 73–84.
Marcus, J.H., Sutcliffe, D.W.& Willoughby, L.G. 1978. Feeding and growth of Asellus aquaticus (Isopoda) on food items from the littoral of Windermere, including green leaves of Elodea canadensis. Freshwat. Biol. 8: 505–519.
Mason, C.F.& Bryant, R.J. 1975. Periphyton production and grazing by chironomids in Alderfen Broad, Norfolk. Freshwat. Biol. 5: 271–277.
McMahon, R.F., Hunter, R.D.& Russel-Hunter, W.D. 1974. Variation in aufwuchs at six freshwater habitats in terms of carbon biomass and of carbon:nitrogen ration. Hydrobiologia 45: 391–404.
Meulemans, J.T.& Heinis, F. 1983. Biomass and production of periphyton attached to dead reed stems in Lake Maarsseveen. In: Wetzel, R. (ed.), Periphyton of Freshwater Ecosystems, pp. 169–173. Dr W. Junk Publishers, The Hague, The Netherlands.
Meulemans, J.T.& Roos, P.J. 1985. Structure and architecture of the periphytic community on dead reed stems in Lake Maarsseveen. Arch. Hydrobiol. 102: 487–502.
Moed, J.R.& Hallegraef, G.M. 1978. Some problems in the estimation of chlorophyll-a and phaeopigments from pre-and post-acidification spectrophotometric measurements. Int. Revue ges. Hydrobiol. 63: 787–800.
Moore, J.W. 1975. The role of algae in the diet of Asellus aquaticus L. and Gammarus pulex L. J. Anim. Ecol. 44: 719–730.
Nederlandse Praktijkrichtlijn. 1984. NPR 6505, benodigdheden, werkwijze en medium voor het kweken van algen (Dutch Standards, materials and methods for the cultivation of algae, in Dutch). Nederlands Normalisatie Instituut, Delft, 4 pp.
Nicotri, M.E. 1977. Grazing effects of four marine intertidal herbivores on the microflora. Ecology 58: 1020–1032.
Norusis, M.J. 1986. SPSS-PC+ Manual. SPSS Inc., Chicago, 559 pp.
Orth, R.J.& Van Montfrans, J. 1984. Epiphyte seagrass relationships with an emphasis on the role of micrograzing: a review. Aquat. Bot. 18: 43–69.
Osenberg, C.W. 1989. Resource limitation, competition and the influence of life history in a freshwater snail community. Oecologia 79: 512–519.
Phillips, G.L., Eminson, D.F.& Moss, B. 1978. A mechanism to account for macrophyte decline in progressively eutrophicated waters. Aquat. Bot. 4: 103–125.
Reddingius, J., Schilstra, A.J.& Thomas, G. 1983. The grid method in estimating path length of a moving animal. J. Anim. Ecol. 52: 199–209.
Reynoldson, T.B.& Piearce, B. 1979. Predation on snails by two species of triclad and its bearing on the distribution of Planaria torva in Britain. J. Zool. London 189: 459–484.
Russel Hunter, W. 1961. Life cycles of four freshwater snails in limited populations in Loch Lomond, with a discussion of infraspecific variation. Proc. Zool. Soc. London 137: 135–171.
Russel-Hunter, W.D.& Eversole, A.G. 1976. Evidence for tissue degrowth in starved freshwater pulmonate snails (Helisoma trivolvis) from tissue, carbon, and nitrogen analyses. Com. Biochem. Physiol. 54A: 447–453.
Sand-Jensen, K. 1977. Effect of epiphytes on eelgrass photosynthesis. Aquat. Bot. 3: 55–63.
Sand-Jensen, K.& Borum, J. 1984. Epiphyte shading and its effect on photosynthesis and diel metabolism of Lobelia dortmanna L. during the spring bloom in a Danish lake. Aquat. Bot. 20: 109–119.
Silberstein, K., Chiffings, A.W.& McComb, A.J. 1986. The loss of seagrass in Cockburn Sound, Western Australia III. The effect of epiphytes on the productivity of Posidonia australis Hook. F. Aquat. Bot. 24: 355–371.
Soszka, G.J. 1975. The invertebrates on submerged macrophytes in three Masurian lakes. Ekol. Pol. 23: 371–391.
Steel, R.G.D.& Torrie, J.H. 1980. Principles and Procedures of Statistics, a Biometrical Approach. Second edition. Mc Graw Hill Book Company, Singapore, 633 pp.
Streble, H.& Krauter, D. 1985. Das Leben im Wassertropfen: Mikroflora und Mikrofauna des Süsswassers; ein Bestimmungsbuch. 7. Auflage, Kosmos — Franckh’sche Verlagshandlung, W. Keller&Co, Stuttgart, Germany, 369 pp.
Sumner, W.T.& Mclntire, C.D. 1982. Grazer-peripyton interactions in laboratory streams. Arch. Hydrobiol. 93: 135–157.
Van Dijk, G.M. 1991. Light climate and its impact on Potamogetonpectinatus L. in a shallow eutrophic lake. Ph.D. thesis, Wageningen Agricultural University, 125 pp.
Van Dijk, G.M.& Van Vierssen, W. 1991. Survival mechanisms of a Potamogeton pectinatus L. population under different light conditions in a shallow eutrophic lake (Lake Veluwe) in The Netherlands. Aquat. Bot. 39: 121–129.
Van Montfrans, J., Orth, R.J.& Vay, S.A. 1982. Preliminary studies of grazing by Bittium varium on eelgrass periphyton. Aquat. Bot. 14: 75–89.
Van Vierssen, W. 1982. The ecology of communities dominated by Zannichellia taxa in western Europe. II. Distribution, synecology and productivity aspects in relation to environmental factors. Aquat. Bot. 13: 385–483.
Van Vierssen, W.& Bij de Vaate, A. 1990. Licht en waterplanten, oorzaken van biomassafluctuaties van onderwatervegetaties in het Veluwemeer (Light and submerged macrophytes, causes for macrophyte biomass fluctuations in Lake Veluwe, in Dutch). Report of the Institute for Inland Water Management and Waste Water Treatment, Wageningen Agricultural University, Dept of Nature Conservation and the International Institute for Hydraulic and Environmental Engineering, 233 pp.
Vernon, L.P. 1960. Spectrophotometric determination of chlorophylls and pheophytins in plant extracts. Analytic Chemistry 32: 1144–1150.
Watson, M.A.& Casper, B.B. 1984. Morphogenetic constraints on patterns of carbon distribution in plants. Ann. Rev. Ecol. Syst. 15: 233–258.
Wetzel, R.G. 1983. Opening remarks. In: Wetzel, R.G. (ed.), Periphyton of Freshwater Ecosystems, pp. 3–4. Dr W. Junk Publishers, The Hague, The Netherlands.
Young, M.R. 1975. The life cycles of six species of freshwater molluscs in the Worcester-Birmingham canal. Proc. malac. Soc. London 41: 533–548.
Editor information
Rights and permissions
Copyright information
© 1994 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Vermaat, J.E. (1994). Periphyton removal by freshwater micrograzers. In: van Vierssen, W., Hootsmans, M., Vermaat, J. (eds) Lake Veluwe, a Macrophyte-dominated System under Eutrophication Stress. Geobotany, vol 21. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-2032-6_13
Download citation
DOI: https://doi.org/10.1007/978-94-011-2032-6_13
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-010-4899-6
Online ISBN: 978-94-011-2032-6
eBook Packages: Springer Book Archive