Abstract
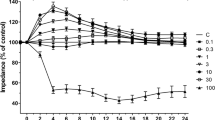
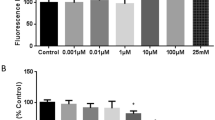
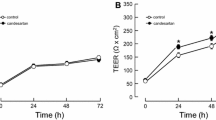
Ischemic alterations in the glutathione (GSH) redox system of the blood-brain barrier (BBB) may facilitate oxidative injury and formation of vasogenic brain edema. In this study, both the intra- and extracellular GSH contents of human cerebromicrovascular endothelial cells (HCEC) were reduced by 35% after exposing the cells to 4 h in vitro ischemia and 24 h-recovery. The intracellular/extracel-lular GSH ratio was not affected, indicating a constant rate of GSH efflux. The activities of the peroxide detoxifying enzymes, glutathione peroxidase and glutathione S-transferase, increased by 35%-50%, whereas the GSH regenerating enzyme, glutathione reductase, remained unchanged in ischemic HCEC. γ-glutamyl transpeptidase (GGTP), a GSH catabolizing enzyme enriched in brain capillaries, was reduced by 30–50% in ischemic HCEC. The effect of in vitro ischemia on HCEC permeability was assessed by measuring sodium fluorescein clearance across a compartmentalized in vitro BBB model. Sodium fluorescein clearance across HCEC monolayers exposed to leukotriene C4 in the presence of the GGTP inhibitor, acivicin (1 µM), or after in vitro ischemia was increased by 60% and 30%, respectively, suggesting that oxidative stress and loss of GGTP may ‘unmask’ BBB permeabilizing actions of leukotrienes. These results indicate that oxidative stress and loss of GGTP activity in HCEC contribute to ischemic BBB disruption and vasogenic brain edema.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Baba T, Black KL, Ikezaki K, Chen K, Becker DP (1991) Intracarotid infusion of leukotriene C4 selectively increases blood-brain barrier permeability after focal ischemia in rats. J Cerebral Blood Flow Metab 11: 638–643
Baba T, Chio C-C, Black KL (1992) The effect of 5-lipoxygenase inhibition on blood-brain barrier permeability in experimental brain tumors. J Neurosurg 77: 403–406
Black KL (1995) Biochemical opening of the blood brain barrier. Adv Drug Delivery Rev 15: 37–52
Black KL, Baba T, Pardridge WM (1994) Enzymatic barrier protects brain capillaries from leukotrience C4. J Neurosurg. 81: 745–751
Black KL, Hoff JT, McGillicuddy JE, Gebarski SS (1986) Increased leukotrience C4 and vasogenic edema surrounding brain tumors in humans. Ann Neurol 19: 592–595
Bradford MM (1976) A rapid and sensitive method of quantification of microgram quantities of proteins utilizing the principle of protein dye binding. Anal Biochem 72: 248–254
Carlberg I, Mannervik B SPIVn (1985) Glutathione reductase. Methods enzymol. In: Meister A (ed) Academic Press, New York, 113:484–490
Chan PH, Schmidley JW, Fishman RA, Longar SM (1984) Brain injury, edema and vascular permeability changes induced by oxygen-derived free radicals. Neurology 34: 315–320
Claesson H-E, Haeggstrom J (1988) Human endothelial cells stimulate leukotriene synthesis and convert granulocyte released leukotriene A4 into leukotrienes B4, C4, D4, and E4. Eur J Bio-chem 173: 93–100
Dehouck MP, Jolliet-Riant P, Bree F, Fruchart JC, Cechelli R, Tillement J-P (1992) Drug transfer across the blood-brain barrier: correlation between in vitro and in vivo models. J Neuro-chem 58: 1790–1797
delZoppo GJ (1994) Microvascular changes during cerebral ischemia and reperfusion. Cerebrovasc Brain Metab Rev 6:47–96
Flohe L, Gunzler A SPIVnz (1984) Assays of glutathione peroxidase. Methods in enzymology. In: Packer L (ed) Academic Press, New York, 105:114–121
Frey A SPIVn (1993) Gamma-glutamyl transpeptidase: molecular cloning and structural and functional features of a blood-brain barrier marker protein. The blood-brain barrier cellular and molecular biology. In: Pardridge WM (ed) Raven Press, New York, pp 339–368
Gidday JM, Beetsch JW, Park TS (1999) Endogenous glutathione protects cerebral endothelial cells from traumatic injury. J Neurotrauma 16: 27–36
Habig EH, Pabst MJ, Jakoby WB (1974) Glutathione-S-trans-ferases: the first enzymatic step in mercaptopuric acid formation. J Biol Chem 249: 7130–7139
Hurst RD, Heales SJ, Dobbie MS, Barker JE, Clark JB (1998) Decreased endothelial cell glutathione and increased sensitivity to oxidative stress in an in vitro blood-brain barrier model system. Brain Res 802: 232–240
Juurlink BHJ, Schultke, E, Hertz L (1996) Glutathione release and catabolism during energy substrate restriction in astrocytes. Brain Res 710: 229–233
Kokura S, Wolf RE, Yoshikawa T, Granger DN, Aw TY (1999) Molecular mechanisms of neutrophil-endothelial cell adhesion induced by redox imbalance. Circ Res 84: 516–524
Lindgren JA, Karnushina I, Clesson H-E (1989) Role of brain microvessels and choroid plexus in cerebral metabolism of leukotrienes. Ann NY Acad Sci 559: 112–120
Moskovitz MA, Kiwak KJ, Hekimian K, Levine L (1984) Synthesis of compounds with properties of leukotriene C4 and D4 in gerbil brains after ischemia and reperfusion. Science 224: 886–888
Muruganandam A, Herx LM, Monette R, Durkin JP, Stanimirovic DB (1997) Development of immortalized human cere-bromicrovascular endothelial cell line as an in vitro model of the human blood-brain barrier. FASEB J 13: 1187–1197
Plateel M, Dehouck M-P, Torpier G, Cecchelli R, Teissier E (1995) Hypoxia increases the susceptibility to oxidant stress and the permeability of the blood-brain barrier endothelial cell monolayer. J Neurochem 65: 2138–2145
Rabin O, Piciotti M, Drieu K, Bourre JM, Roux F (1996) Effects of anoxia and reoxygenation on antioxidant enzyme activities in immortalized brain endothelial cells. In Vitro Cell Dev 32: 221–224
Samuelsson B (1983) Leukotrienes: mediators of immediate hypersensitivity and inflammation. Science 220: 568–575
Stanimirovic D, Morley P, Ball R, Hamel E, Mealing G, Durkin JP (1996) Angiotensin II-induced fluid phase endocytosis in human cerebromicrovascular endothelial cells is regulated by the inositol-phosphate signaling pathway. J Cell Physiol 169: 455–467
Stanimirovic D, Shapiro A, Wong J, Hutchison J, Durkin J (1997) The induction of ICAM-1 in human cerebromicrovascular endothelial cells (HCEC) by ischemia-like conditions promotes enhanced neutrophil/HCEC adhesion. J Neuro-immunol 76(1–2): 193–205
Stanimirovic DB, Wong J, Ball R, Durkin JP (1995) Free radical-induced endothelial membrane dysfunction at the site of blood brain barrier: relationship between lipid peroxidation, Na, K-ATPase activity and 51Cr release. Neurochem Res 20: 1417–1427
Strasser A, Stanimirovic DB, Kawai N, McCarron RM, Spatz M (1997) Hypoxia modulates free radical formation in brain microvascular endothelium. Acta Neurochir [Suppl] (Wien) 70: 8–11
Unterberg A, Schmidt W, Wahl M, Ellis EF, Marmarou A, Baethmann A (1991) Evidence against leukotrienes as mediators of brain edema. J Neurosurg 74: 773–780
Wei EP, Ellison M, Kontos HA, Povlishock JT (1986) O2 radicals in arachidonate-induced increased blood-brain barrier permeability to proteins. Am J Physiol 251: H693–H699.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2000 Springer-Verlag Wien
About this paper
Cite this paper
Muruganandam, A., Smith, C., Ball, R., Herring, T., Stanimirovic, D. (2000). Glutathione Homeostasis and Leukotriene-Induced Permeability in Human Blood-Brain Barrier Endothelial Cells Subjected to in Vitro Ischemia. In: Mendelow, A.D., et al. Brain Edema XI. Acta Neurochirurgica Supplements, vol 76. Springer, Vienna. https://doi.org/10.1007/978-3-7091-6346-7_6
Download citation
DOI: https://doi.org/10.1007/978-3-7091-6346-7_6
Publisher Name: Springer, Vienna
Print ISBN: 978-3-7091-7257-5
Online ISBN: 978-3-7091-6346-7
eBook Packages: Springer Book Archive