Abstract
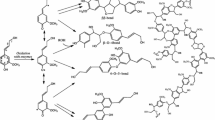
Lignin-carbohydrate complexes (LCCs) are one of the glycoconjugates in which hydrophobic lignin is chemically bound to hydrophilic polysaccharides in wood cell walls. So far, several types of lignin-carbohydrate linkages have been proposed. These include: (1) benzyl ether type (Fig. 4.1; Košíková et al. 1979; Yaku et al. 1981; Koshijima et al. 1984; Eriksson and Lindgren 1977; Watanabe et al. 1989; Ohnishi et al. 1992), in which the α-hydroxyl group of the lignin is etherified with the hydroxyl group of carbohydrate; (2) benzyl ester type (Fig. 4.2; Yaku et al. 1976; Eriksson et al. 1980; Takahashi and Koshijima 1988b; Obst 1982; Lundquist et al. 1983; Das et al. 1984; Watanabe and Koshijima 1988; Imamura et al. 1994), in which the α-hydroxyl group of the lignin is esterified with the carboxyl group of glucuronic acid residue; (3) glycoside type (Fig. 4.3: Hayashi 1961; Smelstorius 1974; Yaku et al. 1976; Joseleau and Kesraoui 1986; Kondo et al. 1990a; Ford 1990), in which alcoholic or phenolic hydroxyl groups of the lignin are glycosylated by the reducing end group of carbohydrates; and (4) acetal type, in which two hydroxyl groups of carbohydrate are linked to lignin by an acetal linkage (Fig. 4.4; Bolker and Sommerville 1963; Xie et al. 2000). Among the chemical linkages, the benzyl ether and ester types have been considered to be the most probable mode of linkage (Watanabe 1995).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Rights and permissions
Copyright information
© 2003 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Watanabe, T. (2003). Analysis of Native Bonds Between Lignin and Carbohydrate by Specific Chemical Reactions. In: Association Between Lignin and Carbohydrates in Wood and Other Plant Tissues. Springer Series in Wood Science. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-05191-7_4
Download citation
DOI: https://doi.org/10.1007/978-3-662-05191-7_4
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-07853-8
Online ISBN: 978-3-662-05191-7
eBook Packages: Springer Book Archive