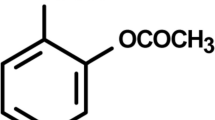
Abstract
Acetylsalicylic acid (aspirin) is unique—it contains two active moieties within one and the same molecule: the reactive acetyl group of the unmetabolized aspirin and the salicylate metabolite. Both have different pharmacokinetics and pharmacodynamics. Aspirin is rapidly hydrolyzed into inactive acetate and salicylate by aspirin “esterases,” preferentially in the intestinal epithelium, liver, and blood. The half-life of unmetabolized aspirin in blood is 20–30 min, the half-life of salicylate at analgesic doses of 1–2 g about 3 h. Different galenic preparations of aspirin are available with different pharmacokinetics, most notable a disintegrating formulation with markedly increased systemic bioavailability of unmetabolized aspirin. Pharmacological actions of aspirin, i.e., anti-inflammatory, analgesic, and antipyretic effects, are largely due to acetylation. This action is irreversible, i.e., the duration of action is determined by the turnover rate of the protein and not by the short half-life of aspirin in blood. The most important protein targets are the prostaglandin cyclooxygenases-1 (COX-1) and COX-2. Both enzymes are inhibited in vitro at comparable potency. In vivo; the inhibition of COX-2 is less pronounced, probably because of the rapid protein turnover rate of the enzyme and the short half-life of aspirin. In addition, acetylation of COX-2 allows for generation of15-(R)HETE and subsequent formation of “aspirin-triggered lipoxin” (ATL) by interaction with white cell lipoxygenases. In the cardiovascular system, aspirin also acetylates eNOS with subsequent upregulation of NO formation and enhanced expression of the antioxidant heme-oxygenase-1. Salicylate has some actions by its own, notably uncoupling of oxidative phosphorylation at low millimolar concentrations. This will contribute to the anti-inflammatory and, perhaps, antipyretic effects of aspirin.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Dreser H. Pharmakologisches über Aspirin (Acetylsalizylsäure). Pflügers Arch Physiol. 1899;76:306–18.
Vane JR. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat New Biol. 1971;231(25):232–5.
Roth GJ, Stanford N, Majerus PW. Acetylation of prostaglandin synthase by aspirin. Proc Natl Acad Sci U S A. 1975;72(8):3073–6.
Bateman LA, et al. An alkyne-aspirin chemical reporter for the detection of aspirin-dependent protein modification in living cells. J Am Chem Soc. 2013;135(39):14568–73.
Wang J, et al. Mapping sites of aspirin-induced acetylations in live cells by quantitative acid-cleavable activity-based protein profiling (QA-ABPP). Scientific Reports. 2015;5.
Nagelschmitz J, Blunk M, Krätschmar J, et al. Pharmacokinetics and pharmacodynamics of acetylsalicylic acid after intravenous and oral administration to healthy volunteers. Clin Pharmacol. 2013;5:1–9.
Shen J, et al. Model representation of salicylate pharmacokinetics using unbound plasma salicylate concentrations and metabolite urinary excretion rates following a single oral dose. J Pharmacokinet Biopharm. 1991;19(5):575–95.
Frantz B, O’Neill EA. The effect of sodium salicylate and aspirin on NF-kappa B. Science. 1995;270(5244):2017–9.
Schrör K, Rauch BH. Aspirin and lipid mediators in the cardiovascular system. Prostaglandins Other Lipid Mediat. 2015;121(Pt A):17–23.
Weyrich AS, Lindemann S, Zimmerman GA. The evolving role of platelets in inflammation. J Thromb Haemost. 2003;1(9):1897–905.
Hohlfeld T, Schrör K. Antiinflammatory effects of aspirin in ACS: relevant to its cardiocoronary actions? Thrombosis Haemost. 2015;114:469–77.
Patrignani P, Filabozzi P, Patrono C. Selective cumulative inhibition of platelet thromboxane production by low-dose aspirin in healthy subjects. J Clin Invest. 1982;69(6):1366–72.
Reilly IA, FitzGerald GA. Inhibition of thromboxane formation in vivo and ex vivo: implications for therapy with platelet inhibitory drugs. Blood. 1987;69(1):180–6.
Catella-Lawson F, et al. Cyclooxygenase inhibitors and the antiplatelet effects of aspirin. N Engl J Med. 2001;345(25):1809–17.
Saxena A, et al. Drug/drug interaction of common NSAIDs with antiplatelet effect of aspirin in human platelets. Eur J Pharmacol. 2013;721(1-3):215–24.
Hennekens CH, et al. Hypothesis formulation from subgroup analyses: nonadherence or nonsteroidal anti-inflammatory drug use explains the lack of clinical benefit of aspirin on first myocardial infarction attributed to “aspirin resistance”. Am Heart J. 2010;159(5):744–8.
Bhala N, et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: meta-analyses of individual participant data from randomised trials. Lancet. 2013;382(9894):769–79.
FitzGerald GA, et al. Endogenous biosynthesis of prostacyclin and thromboxane and platelet function during chronic administration of aspirin in man. J Clin Invest. 1983;71(3):676–88.
Higgs GA, et al. Pharmacokinetics of aspirin and salicylate in relation to inhibition of arachidonate cyclooxygenase and antiinflammatory activity. Proc Natl Acad Sci U S A. 1987;84(5):1417–20.
Hersh EV, Moore PA, Ross GL. Over-the-counter analgesics and antipyretics: a critical assessment. Clin Ther. 2000;22(5):500–48.
Schwartz JI, et al. Cyclooxygenase-2 inhibition by rofecoxib reverses naturally occurring fever in humans. Clin Pharmacol Ther. 1999;65(6):653–60.
Aronoff DM, et al. Inhibition of prostaglandin H2 synthases by salicylate is dependent on the oxidative state of the enzymes. J Pharmacol Exp Ther. 2003;304(2):589–95.
Mancini JA, et al. Mutation of serine-516 in human prostaglandin G/H synthase-2 to methionine or aspirin acetylation of this residue stimulates 15-R-HETE synthesis. FEBS Lett. 1994;342(1):33–7.
Lecomte M, et al. Acetylation of human prostaglandin endoperoxide synthase-2 (cyclooxygenase-2) by aspirin. J Biol Chem. 1994;269(18):13207–15.
Claria J, Serhan CN. Aspirin triggers previously undescribed bioactive eicosanoids by human endothelial cell-leukocyte interactions. Proc Natl Acad Sci U S A. 1995;92(21):9475–9.
Spite M, Serhan CN. Novel lipid mediators promote resolution of acute inflammation: impact of aspirin and statins. Circ Res. 2010;107(10):1170–84.
Morris T, et al. Effects of low-dose aspirin on acute inflammatory responses in humans. J Immunol. 2009;183(3):2089–96.
Taubert D, et al. Aspirin induces nitric oxide release from vascular endothelium: a novel mechanism of action. Br J Pharmacol. 2004;143(1):159–65.
Jung SB, et al. Histone deacetylase 3 antagonizes aspirin-stimulated endothelial nitric oxide production by reversing aspirin-induced lysine acetylation of endothelial nitric oxide synthase. Circ Res. 2010;107(7):877–87.
Kalgutkar AS, et al. Aspirin-like molecules that covalently inactivate cyclooxygenase-2. Science. 1998;280(5367):1268–70.
Grosser N, et al. Heme oxygenase-1 induction may explain the antioxidant profile of aspirin. Biochem Biophys Res Commun. 2003;308(4):956–60.
Grosser N, Schröder H. Aspirin protects endothelial cells from oxidant damage via the nitric oxide-cGMP pathway. Arterioscler Thromb Vasc Biol. 2003;23(8):1345–51.
Nascimento-Silva V, et al. Novel lipid mediator aspirin-triggered lipoxin A4 induces heme oxygenase-1 in endothelial cells. Am J Physiol Cell Physiol. 2005;289(3):C557–63.
Hennekens CH, et al. A randomized trial of aspirin at clinically relevant doses and nitric oxide formation in humans. J Cardiovasc Pharmacol Ther. 2010;15(4):344–8.
Hetzel S, et al. Aspirin increases nitric oxide formation in chronic stable coronary disease. J Cardiovasc Pharmacol Ther. 2013;18(3):217–21.
Hennekens CH. A randomized trial of aspirin at clinically relevant doses and nitric oxide formation in humans. J Cardiovasc Pharmacol. 2010;15.
Cronstein BN, Montesinos MC, Weissmann G. Salicylates and sulfasalazine, but not glucocorticoids, inhibit leukocyte accumulation by an adenosine-dependent mechanism that is independent of inhibition of prostaglandin synthesis and p105 of NFkappaB. Proc Natl Acad Sci U S A. 1999;96(11):6377–81.
Hasko G, et al. Adenosine receptors: therapeutic aspects for inflammatory and immune diseases. Nat Rev Drug Discov. 2008;7(9):759–70.
O’Brien M, et al. Aspirin attenuates platelet activation and immune activation in HIV-1-infected subjects on antiretroviral therapy: a pilot study. J Acquir Immune Defic Syndr. 2013;63(3):280–8.
Chiang N, et al. Aspirin triggers antiinflammatory 15-epi-lipoxin A4 and inhibits thromboxane in a randomized human trial. Proc Natl Acad Sci U S A. 2004;101(42):15178–83.
Edwards JE, et al. Oral aspirin in postoperative pain: a quantitative systematic review. Pain. 1999;81(3):289–97.
Scholz J, Woolf CJ. Can we conquer pain? Nat Neurosci. 2002;5(Suppl):1062–7.
Svensson CI, Zattoni M, Serhan CN. Lipoxins and aspirin-triggered lipoxin inhibit inflammatory pain processing. J Exp Med. 2007;204(2):245–52.
Vardeh D, et al. COX2 in CNS neural cells mediates mechanical inflammatory pain hypersensitivity in mice. J Clin Invest. 2009;119(2):287–94.
Anikwue R, et al. Decrease in efficacy and potency of nonsteroidal anti-inflammatory drugs by chronic delta(9)-tetrahydrocannabinol administration. J Pharmacol Exp Ther. 2002;303(1):340–6.
Ruggieri V, et al. The antinociceptive effect of acetylsalicylic acid is differently affected by a CB1 agonist or antagonist and involves the serotonergic system in rats. Life Sci. 2010;86(13-14):510–7.
Göbel H, et al. Acetylsalicylic acid activates antinociceptive brain-stem reflex activity in headache patients and in healthy subjects. Pain. 1992;48(2):187–95.
Diener HC, et al. Aspirin in the treatment of acute migraine attacks. Expert Rev Neurother. 2006;6(4):563–73.
Adler RD, et al. The effect of salicylate on pyrogen-induced fever in man. Clin Sci. 1969;37(1):91–7.
Rosendorff C, Cranston WI. Effects of salicylate on human temperature regulation. Clin Sci. 1968;35(1):81–91.
MacDonald TM, Wei L. Effect of ibuprofen on cardioprotective effect of aspirin. Lancet. 2003;361(9357):573–4.
Hohlfeld T, Saxena A, Schror K. High on treatment platelet reactivity against aspirin by non-steroidal anti-inflammatory drugs--pharmacological mechanisms and clinical relevance. Thromb Haemost. 2013;109(5):825–33.
Mason WD, Winer N. Influence of food on aspirin absorption from tablets and buffered solutions. J Pharm Sci. 1983;72(7):819–21.
Voelker M, Hammer M. Dissolution and pharmacokinetics of a novel micronized aspirin formulation. Inflammopharmacology. 2012;20(4):225–31.
Graham DY, Smith JL. Aspirin and the stomach. Ann Intern Med. 1986;104(3):390–8.
Cryer B, Feldman M. Effects of very low dose daily, long-term aspirin therapy on gastric, duodenal, and rectal prostaglandin levels and on mucosal injury in healthy humans. Gastroenterology. 1999;117(1):17–25.
Rowland M, et al. Absorption kinetics of aspirin in man following oral administration of an aqueous solution. J Pharm Sci. 1972;61(3):379–85.
Rowland M, et al. Kinetics of acetylsalicylic acid disposition in man. Nature. 1967;215(5099):413–4.
Warner TD, et al. Influence of plasma protein on the potencies of inhibitors of cyclooxygenase-1 and -2. FASEB J. 2006;20(3):542–4.
Harris PA, Riegelman S. Influence of the route of administration on the area under the plasma concentration-time curve. J Pharm Sci. 1969;58(1):71–5.
Pedersen AK, FitzGerald GA. Dose-related kinetics of aspirin. Presystemic acetylation of platelet cyclooxygenase. N Engl J Med. 1984;311(19):1206–11.
Zhou Y, Boudreau DM, Freedman AN. Trends in the use of aspirin and nonsteroidal anti-inflammatory drugs in the general U.S. population. Pharmacoepidemiol Drug Saf. 2013;23(1):43–50.
Adebayo GI, Williams J, Healy S. Aspirin esterase activity—evidence for skewed distribution in healthy volunteers. Eur J Intern Med. 2007;18(4):299–303.
Rowland M, Riegelman S. Pharmacokinetics of acetylsalicylic acid and salicylic acid after intravenous administration in man. J Pharm Sci. 1968;57:1313–9.
Kuehl GE, et al. Glucuronidation of the aspirin metabolite salicylic acid by expressed UDP-glucuronosyltransferases and human liver microsomes. Drug Metab Dispos. 2006;34(2):199–202.
Chen Y, et al. UGT1A6 polymorphism and salicylic acid glucuronidation following aspirin. Pharmacogenet Genomics. 2007;17(8):571–9.
Osawa K, et al. Association between polymorphisms in UDP-glucuronosyltransferase 1A6 and 1A7 and colorectal cancer risk. Asian Pac J Cancer Prev. 2012;13(5):2311–4.
Bedford C, Cummings AJ, Martin BK. A kinetic study of the elimination of salicylate in man. Br J Pharmacol Chemother. 1965;24:418–31.
Notarianni LJ, Ogunbona FA, Oldham HG. Glycine conjugation of salicylic acid after aspirin overdose. Br J Clin Pharmacol. 1983;15.
Forman WB, Davidson ED, Webster Jr LT. Enzymatic conversion of salicylate to salicylurate. Mol Pharmacol. 1971;7(3):247–59.
Wilson JT, et al. Gentisuric acid: metabolic formation in animals and identification as a metabolite of aspirin in man. Clin Pharmacol Ther. 1978;23(6):635–43.
Cham BE, et al. Simultaneous liquid-chromatographic quantitation of salicylic acid, salicyluric acid, and gentisic acid in urine. Clin Chem. 1980;26(1):111–4.
Caldwell J, Gorman JO’, Smith RL. Inter-individual differences in the glycine conjugation of salicylic acid [proceedings]. Br J Clin Pharmacol, 1980. 9(1): 114p.
Steiner TJ, Lange R, Voelker M. Aspirin in episodic tension-type headache: placebo-controlled dose-ranging comparison with paracetamol. Cephalalgia. 2003;23(1):59–66.
Martinez-Martin P, et al. Efficacy and safety of metamizol vs. acetylsalicylic acid in patients with moderate episodic tension-type headache: a randomized, double-blind, placebo- and active-controlled, multicentre study. Cephalalgia. 2001;21(5):604–10.
Gatoulis SC, Voelker M, Fisher M. Assessment of the efficacy and safety profiles of aspirin and acetaminophen with codeine: results from 2 randomized, controlled trials in individuals with tension-type headache and postoperative dental pain. Clin Ther. 2012;34(1):138–48.
MacGregor EA, Dowson A, Davies PT. Mouth-dispersible aspirin in the treatment of migraine: a placebo-controlled study. Headache. 2002;42(4):249–55.
Lange R, Schwarz JA, Hohn M. Acetylsalicylic acid effervescent 1000 mg (Aspirin) in acute migraine attacks; a multicentre, randomized, double-blind, single-dose, placebo-controlled parallel group study. Cephalalgia. 2000;20(7):663–7.
Diener HC, et al. Placebo-controlled comparison of effervescent acetylsalicylic acid, sumatriptan and ibuprofen in the treatment of migraine attacks. Cephalalgia. 2004;24(11):947–54.
Diener HC, et al. Efficacy of 1,000 mg effervescent acetylsalicylic acid and sumatriptan in treating associated migraine symptoms. Eur Neurol. 2004;52(1):50–6.
Lipton RB, et al. Aspirin is efficacious for the treatment of acute migraine. Headache. 2005;45(4):283–92.
Lampl C, Voelker M, Diener HC. Efficacy and safety of 1,000 mg effervescent aspirin: individual patient data meta-analysis of three trials in migraine headache and migraine accompanying symptoms. J Neurol. 2007;254(6):705–12.
Eccles R, et al. Effects of acetylsalicylic acid on sore throat pain and other pain symptoms associated with acute upper respiratory tract infection. Pain Med. 2003;4(2):118–24.
Zhang WY, Li Wan A. Po, Efficacy of minor analgesics in primary dysmenorrhoea: a systematic review. Br J Obstet Gynaecol. 1998;105(7):780–9.
Forbes JA, et al. Evaluation of an ibuprofen controlled-release tablet and placebo in postoperative oral surgery pain. Pharmacotherapy. 1991;11(3):242–8.
Forbes JA, et al. Analgesic efficacy of bromfenac, ibuprofen, and aspirin in postoperative oral surgery pain. Clin Pharmacol Ther. 1992;51(3):343–52.
Cooper SA. Comparative analgesic efficacies of aspirin and acetaminophen. Arch Intern Med. 1981;141(3):282–5.
Cooper SA, Voelker M. Evaluation of onset of pain relief from micronized aspirin in a dental pain model. Inflammopharmacology. 2012;20(4):233–42.
Bachert C, et al. Aspirin compared with acetaminophen in the treatment of fever and other symptoms of upper respiratory tract infection in adults: a multicenter, randomized, double-blind, double-dummy, placebo-controlled, parallel-group, single-dose, 6-hour dose-ranging study. Clin Ther. 2005;27(7):993–1003.
Kanani K, Gatoulis SC, Voelker M. Influence of differing analgesic formulations of aspirin on pharmacokinetic parameters. Pharmaceutics. 2015;7(3):188–98.
Lecchi M, et al. Pharmacokinetics and safety of a new aspirin formulation for the acute treatment of primary headaches. Expert Opin Drug Metab Toxicol. 2014;10(10):1381–95.
Muir N, et al. Comparative bioavailability of aspirin and paracetamol following single dose administration of soluble and plain tablets. Curr Med Res Opin. 1997;13(9):491–500.
Muir N, et al. The influence of dosage form on aspirin kinetics: implications for acute cardiovascular use. Curr Med Res Opin. 1997;13(10):547–53.
Sagar KA, Smyth MR. A comparative bioavailability study of different aspirin formulations using on-line multidimensional chromatography. J Pharm Biomed Anal. 1999;21(2):383–92.
Brandon RA, et al. A new formulation of aspirin: bioavailability and analgesic efficacy in migraine attacks. Cephalalgia. 1986;6(1):19–27.
Stillings M, et al. Comparison of the pharmacokinetic profiles of soluble aspirin and solid paracetamol tablets in fed and fasted volunteers. Curr Med Res Opin. 2000;16(2):115–24.
Voelker M, Centofani R. A bioavailability study of fast release aspirin under fasting and fed conditions and regular aspirin under fed conditions. Bayer Study Report. PH-36651, 2011-11-24, 2011.
Zeymer U, Gebert I, Roitenberg A, Hohlfeld T. Prospective, randomized comparison of 500 mg and 250 mg acetylsalicylic acid i.v. and 300 mg p.o. in patients with acute coronary syndrome, measured by time-dependent thromboxane inhibition (ACUTE). Abstr. AHA Scientific Sessions, Nov 7–11, Orlando, FL, USA, 2015.
Limmroth V, May A, Diener H. Lysine-acetylsalicylic acid in acute migraine attacks. Eur Neurol. 1999;41(2):88–93.
Leniger T, et al. Comparison of intravenous valproate with intravenous lysine-acetylsalicylic acid in acute migraine attacks. Headache. 2005;45(1):42–6.
Weatherall MW, et al. Intravenous aspirin (lysine acetylsalicylate) in the inpatient management of headache. Neurology. 2010;75(12):1098–103.
Neurologie, D.G.f., Therapie der Migräne. 2012.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Schrör, K., Voelker, M. (2016). NSAIDS and Aspirin: Recent Advances and Implications for Clinical Management. In: Lanas, A. (eds) NSAIDs and Aspirin. Springer, Cham. https://doi.org/10.1007/978-3-319-33889-7_7
Download citation
DOI: https://doi.org/10.1007/978-3-319-33889-7_7
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-33887-3
Online ISBN: 978-3-319-33889-7
eBook Packages: MedicineMedicine (R0)