Abstract
Ischemic disorders are the main cause of death in the Western world. With more patients surviving their acute myocardial infarction and an aging population, congestive heart failure is the rising health problem. At present, heart transplantation remains the only curative treatment for end stage heart failure. The discrepancy between demand and supply of donor organs does not fill the clinical need. This explains the huge efforts made in the field of stem cell research trying to establish alternative methods for endogenous tissue regeneration and to find sources for tissue replacement. In contrast to adult stem cells mainly acting in a paracrine fashion pluripotent stem cells have the potential to generate transplantable myocardial and vascular tissue.
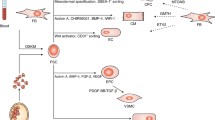
Due to the low percentage of cardiovascular progenitor cells in pluripotent stem cell cultures, various approaches using exogenous factors aim for their amplification and purification in vitro. However, one future key technology may be genetic forward programming based on profound understanding of differentiation pathways in order to direct stem cell differentiation towards cardiovascular fates. In this regard, subtype specific programming has already been achieved by overexpression of distinct early cardiovascular transcription factors leading to populations of either predominantly early/intermediate type cardiomyocytes or differentiated ventricular myocardial cells, respectively. In addition, techniques for gentle purification of myocardial and vascular progenitor cells will have to be further refined in order to enable the generation of highly specific, pure and safe cell populations for transplantations and for tissue engineering.
In contrast, circulating bone marrow-derived progenitor cells have the potential not to replace diseased cardiovascular tissue but to stimulate its endogenous regeneration. Various approaches have recently been introduced to mobilise these cells from their physiological niche and to facilitate a sufficient recruitment into the diseased tissue. While the therapeutic impact of these cells is already being investigated in clinical trials at present, further methodological refinements may once allow their use in clinical routine.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Abbasi M, Javan H, Alizadeh B, Afzalnia S (2011) Can intracoronary stem cell injection permanently improve cardiac function after myocardial infarction? Interact Cardiovasc Thorac Surg 12(2):229–231. doi:10.1510/icvts.2010.252106
Akins RE, Boyce RA, Madonna ML, Schroedl NA, Gonda SR, McLaughlin TA, Hartzell CR (1999) Cardiac organogenesis in vitro: reestablishment of three-dimensional tissue architecture by dissociated neonatal rat ventricular cells. Tissue Eng 5(2):103–118
Assmus B, Leistner DM, Schachinger V, Erbs S, Elsasser A, Haberbosch W, Hambrecht R, Sedding D, Yu J, Corti R, Mathey DG, Barth C, Mayer-Wehrstein C, Burck I, Sueselbeck T, Dill T, Hamm CW, Tonn T, Dimmeler S, Zeiher AM, Group R-AS (2014) Long-term clinical outcome after intracoronary application of bone marrow-derived mononuclear cells for acute myocardial infarction: migratory capacity of administered cells determines event-free survival. Eur Heart J 35(19):1275–1283. doi:10.1093/eurheartj/ehu062
Balsam LB, Wagers AJ, Christensen JL, Kofidis T, Weissman IL, Robbins RC (2004) Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature 428(6983):668–673. doi:10.1038/nature02460, nature02460 [pii]
Ban K, Wile B, Kim S, Park HJ, Byun J, Cho KW, Saafir T, Song MK, Yu SP, Wagner M, Bao G, Yoon YS (2013) Purification of cardiomyocytes from differentiating pluripotent stem cells using molecular beacons that target cardiomyocyte-specific mRNA. Circulation 128(17):1897–1909. doi:10.1161/CIRCULATIONAHA.113.004228
Bel A, Planat-Bernard V, Saito A, Bonnevie L, Bellamy V, Sabbah L, Bellabas L, Brinon B, Vanneaux V, Pradeau P, Peyrard S, Larghero J, Pouly J, Binder P, Garcia S, Shimizu T, Sawa Y, Okano T, Bruneval P, Desnos M, Hagege AA, Casteilla L, Puceat M, Menasche P (2010) Composite cell sheets: a further step toward safe and effective myocardial regeneration by cardiac progenitors derived from embryonic stem cells. Circulation 122(11):S118–S123. doi:122/11_suppl_1/S118 [pii], 10.1161/CIRCULATIONAHA.109.927293
Bondue A, Lapouge G, Paulissen C, Semeraro C, Iacovino M, Kyba M, Blanpain C (2008) Mesp1 acts as a master regulator of multipotent cardiovascular progenitor specification. Cell Stem Cell 3(1):69–84
Boyle A, Colvin-Adams M (2004) Recipient selection and management. Semin Thorac Cardiovasc Surg 16(4):358–363
Brenner C, Franz WM (2014) Pluripotent-stem-cell-derived epicardial cells: a step toward artificial cardiac tissue. Cell Stem Cell 15(5):533–534. doi:10.1016/j.stem.2014.10.007
Brenner C, Krankel N, Kuhlenthal S, Israel L, Remm F, Fischer C, Herbach N, Speer T, Grabmaier U, Laskowski A, Gross L, Theiss H, Wanke R, Landmesser U, Franz WM (2014) Short-term inhibition of DPP-4 enhances endothelial regeneration after acute arterial injury via enhanced recruitment of circulating progenitor cells. Int J Cardiol 177(1):266–275. doi:10.1016/j.ijcard.2014.09.016
Buckingham M, Meilhac S, Zaffran S (2005) Building the mammalian heart from two sources of myocardial cells. Nat Rev Genet 6(11):826–835
Christopherson KW 2nd, Hangoc G, Mantel CR, Broxmeyer HE (2004) Modulation of hematopoietic stem cell homing and engraftment by CD26. Science 305(5686):1000–1003. doi:10.1126/science.1097071
David R, Groebner M, Franz WM (2005) Magnetic cell sorting purification of differentiated embryonic stem cells stably expressing truncated human CD4 as surface marker. Stem Cells 23(4):477–482
David R, Brenner C, Stieber J, Schwarz F, Brunner S, Vollmer M, Mentele E, Muller-Hocker J, Kitajima S, Lickert H, Rupp R, Franz WM (2008a) MesP1 drives vertebrate cardiovascular differentiation through Dkk-1-mediated blockade of Wnt-signalling. Nat Cell Biol 10(3):338–345
David R, Theiss H, Franz WM (2008b) Connexin 40 promoter-based enrichment of embryonic stem cell-derived cardiovascular progenitor cells. Cells Tissues Organs 188(1–2):62–69
David R, Stieber J, Fischer E, Brunner S, Brenner C, Pfeiler S, Schwarz F, Franz WM (2009) Forward programming of pluripotent stem cells towards distinct cardiovascular cell types. Cardiovasc Res 84(2):263–272
David R, Jarsch V, Schwarz F, Nathan P, Gegg M, Lickert H, Franz WM (2011) Induction of MesP1 by Brachyury(T) generates the common multipotent cardiovascular stem cell. Cardiovasc Res 92(1):115–122. doi:cvr158 [pii], 10.1093/cvr/cvr158
Deindl E, Zaruba MM, Brunner S, Huber B, Mehl U, Assmann G, Hoefer IE, Mueller-Hoecker J, Franz WM (2006) G-CSF administration after myocardial infarction in mice attenuates late ischemic cardiomyopathy by enhanced arteriogenesis. FASEB J 20(7):956–958
DESTATIS (2013) Number of deaths up 2 % on a year earlier. Press release from 12 December 2013, German federal statistical office (422)
Eisenberg LM, Eisenberg CA (2006) Wnt signal transduction and the formation of the myocardium. Dev Biol 293(2):305–315
Engelmann MG, Theiss HD, Theiss C, Henschel V, Huber A, Wintersperger BJ, Schoenberg SO, Steinbeck G, Franz WM (2009) G-CSF in patients suffering from late revascularised ST elevation myocardial infarction: final 1-year-results of the G-CSF-STEMI trial. Int J Cardiol 144(3):399–404
Gassanov N, Er F, Zagidullin N, Hoppe UC (2004) Endothelin induces differentiation of ANP-EGFP expressing embryonic stem cells towards a pacemaker phenotype. FASEB J 18(14):1710–1712
Gerecht-Nir S, David R, Zaruba M, Franz WM, Itskovitz-Eldor J (2003) Human embryonic stem cells for cardiovascular repair. Cardiovasc Res 58(2):313–323
Gnecchi M, Zhang Z, Ni A, Dzau VJ (2008) Paracrine mechanisms in adult stem cell signaling and therapy. Circ Res 103(11):1204–1219. doi:10.1161/CIRCRESAHA.108.176826
Graichen R, Xu X, Braam SR, Balakrishnan T, Norfiza S, Sieh S, Soo SY, Tham SC, Mummery C, Colman A, Zweigerdt R, Davidson BP (2008) Enhanced cardiomyogenesis of human embryonic stem cells by a small molecular inhibitor of p38 MAPK. Differentiation 76(4):357–370
Grepin C, Nemer G, Nemer M (1997) Enhanced cardiogenesis in embryonic stem cells overexpressing the GATA-4 transcription factor. Development 124(12):2387–2395
Gyongyosi M, Wojakowski W, Lemarchand P, Lunde K, Tendera M, Bartunek J, Marban E, Assmus B, Henry TD, Traverse JH, Moye LA, Surder D, Corti R, Huikuri H, Miettinen J, Wohrle J, Obradovic S, Roncalli J, Malliaras K, Pokushalov E, Romanov A, Kastrup J, Bergmann MW, Atsma DE, Diederichsen A, Edes I, Benedek I, Benedek T, Pejkov H, Nyolczas N, Pavo N, Bergler-Klein J, Pavo IJ, Sylven C, Berti S, Navarese EP, Maurer G, Investigators* A (2015) Meta-Analysis of Cell-based CaRdiac stUdiEs (ACCRUE) in patients with acute myocardial infarction based on individual patient data. Circ Res 116(8):1346–1360. doi:10.1161/CIRCRESAHA.116.304346
Hunt SA (1998) Current status of cardiac transplantation. JAMA 280(19):1692–1698
Izarra A, Moscoso I, Canon S, Carreiro C, Fondevila D, Martin-Caballero J, Blanca V, Valiente I, Diez-Juan A, Bernad A (2014) miRNA-1 and miRNA-133a are involved in early commitment of pluripotent stem cells and demonstrate antagonistic roles in the regulation of cardiac differentiation. J Tissue Eng Regen Med. doi:10.1002/term.1977
Jung JJ, Husse B, Rimmbach C, Krebs S, Stieber J, Steinhoff G, Dendorfer A, Franz WM, David R (2014) Programming and isolation of highly pure physiologically and pharmacologically functional sinus-nodal bodies from pluripotent stem cells. Stem Cell Rep 2(5):592–605. doi:10.1016/j.stemcr.2014.03.006
Kanno S, Kim PK, Sallam K, Lei J, Billiar TR, Shears LL 2nd (2004) Nitric oxide facilitates cardiomyogenesis in mouse embryonic stem cells. Proc Natl Acad Sci U S A 101(33):12277–12281
Kattman SJ, Huber TL, Keller GM (2006) Multipotent flk-1+ cardiovascular progenitor cells give rise to the cardiomyocyte, endothelial, and vascular smooth muscle lineages. Dev Cell 11(5):723–732
Kelly RG, Brown NA, Buckingham ME (2001) The arterial pole of the mouse heart forms from Fgf10-expressing cells in pharyngeal mesoderm. Dev Cell 1(3):435–440
Kessler PD, Byrne BJ (1999) Myoblast cell grafting into heart muscle: cellular biology and potential applications. Annu Rev Physiol 61:219–242
Kitajima S, Takagi A, Inoue T, Saga Y (2000) MesP1 and MesP2 are essential for the development of cardiac mesoderm. Development 127(15):3215–3226
Klug MG, Soonpaa MH, Koh GY, Field LJ (1996) Genetically selected cardiomyocytes from differentiating embryonic stem cells form stable intracardiac grafts. J Clin Invest 98(1):216–224
Kolossov E, Lu Z, Drobinskaya I, Gassanov N, Duan Y, Sauer H, Manzke O, Bloch W, Bohlen H, Hescheler J, Fleischmann BK (2005) Identification and characterization of embryonic stem cell-derived pacemaker and atrial cardiomyocytes. FASEB J 19(6):577–579
Kraehenbuehl TP, Ferreira LS, Hayward AM, Nahrendorf M, van der Vlies AJ, Vasile E, Weissleder R, Langer R, Hubbell JA (2011) Human embryonic stem cell-derived microvascular grafts for cardiac tissue preservation after myocardial infarction. Biomaterials 32(4):1102–1109. doi:S0142-9612(10)01290-1 [pii], 10.1016/j.biomaterials.2010.10.005
Lam JT, Moretti A, Laugwitz KL (2009) Multipotent progenitor cells in regenerative cardiovascular medicine. Pediatr Cardiol 30(5):690–698
Larsen W (1998) Essentials of human embryology. Library of congress cataloging-in-publication data, vol 2. Churchill Livingstone, New York
Laugwitz KL, Moretti A, Caron L, Nakano A, Chien KR (2008) Islet1 cardiovascular progenitors: a single source for heart lineages? Development 135(2):193–205
Leal J, Luengo-Fernandez R, Gray A, Petersen S, Rayner M (2006) Economic burden of cardiovascular diseases in the enlarged European Union. Eur Heart J 27(13):1610–1619. doi:ehi733 [pii], 10.1093/eurheartj/ehi733
Lee MS, Makkar RR (2004) Stem-cell transplantation in myocardial infarction: a status report. Ann Intern Med 140(9):729–737
Liao SY, Liu Y, Siu CW, Zhang Y, Lai WH, Au KW, Lee YK, Chan YC, Yip PM, Wu EX, Wu Y, Lau CP, Li RA, Tse HF (2010) Proarrhythmic risk of embryonic stem cell-derived cardiomyocyte transplantation in infarcted myocardium. Heart Rhythm 7(12):1852–1859. doi:S1547-5271(10)00904-5 [pii], 10.1016/j.hrthm.2010.09.006
Lin Q, Fu Q, Zhang Y, Wang H, Liu Z, Zhou J, Duan C, Wang Y, Wu K, Wang C (2010) Tumourigenesis in the infarcted rat heart is eliminated through differentiation and enrichment of the transplanted embryonic stem cells. Eur J Heart Fail 12(11):1179–1185. doi:hfq144 [pii], 10.1093/eurjhf/hfq144
Lindsley RC, Gill JG, Murphy TL, Langer EM, Cai M, Mashayekhi M, Wang W, Niwa N, Nerbonne JM, Kyba M, Murphy KM (2008) Mesp1 coordinately regulates cardiovascular fate restriction and epithelial-mesenchymal transition in differentiating ESCs. Cell Stem Cell 3(1):55–68
Liu HS, Jan MS, Chou CK, Chen PH, Ke NJ (1999) Is green fluorescent protein toxic to the living cells? Biochem Biophys Res Commun 260(3):712–717
Lyons I, Parsons LM, Hartley L, Li R, Andrews JE, Robb L, Harvey RP (1995) Myogenic and morphogenetic defects in the heart tubes of murine embryos lacking the homeo box gene Nkx2-5. Genes Dev 9(13):1654–1666
Maltsev VA, Rohwedel J, Hescheler J, Wobus AM (1993) Embryonic stem cells differentiate in vitro into cardiomyocytes representing sinusnodal, atrial and ventricular cell types. Mech Dev 44(1):41–50
Masumoto H, Ikuno T, Takeda M, Fukushima H, Marui A, Katayama S, Shimizu T, Ikeda T, Okano T, Sakata R, Yamashita JK (2014) Human iPS cell-engineered cardiac tissue sheets with cardiomyocytes and vascular cells for cardiac regeneration. Sci Rep 4:6716. doi:10.1038/srep06716
Meilhac SM, Esner M, Kelly RG, Nicolas JF, Buckingham ME (2004) The clonal origin of myocardial cells in different regions of the embryonic mouse heart. Dev Cell 6(5):685–698
Menasche P (2005) Skeletal myoblast for cell therapy. Coron Artery Dis 16(2):105–110
Menasche P, Hagege AA, Vilquin JT, Desnos M, Abergel E, Pouzet B, Bel A, Sarateanu S, Scorsin M, Schwartz K, Bruneval P, Benbunan M, Marolleau JP, Duboc D (2003) Autologous skeletal myoblast transplantation for severe postinfarction left ventricular dysfunction. J Am Coll Cardiol 41(7):1078–1083
Müller M, Fleischmann BK, Selbert S, Ji GJ, Endl E, Middeler G, Muller OJ, Schlenke P, Frese S, Wobus AM, Hescheler J, Katus HA, Franz WM (2000) Selection of ventricular-like cardiomyocytes from ES cells in vitro. FASEB J 14(15):2540–2548
Mummery CL, Ward D, Passier R (2007) Differentiation of human embryonic stem cells to cardiomyocytes by coculture with endoderm in serum-free medium. Curr Protoc Stem Cell Biol Chapter 1:Unit 1F 2
Murry CE, Soonpaa MH, Reinecke H, Nakajima H, Nakajima HO, Rubart M, Pasumarthi KB, Virag JI, Bartelmez SH, Poppa V, Bradford G, Dowell JD, Williams DA, Field LJ (2004) Haematopoietic stem cells do not transdifferentiate into cardiac myocytes in myocardial infarcts. Nature 428(6983):664–668
Nelson TJ, Faustino RS, Chiriac A, Crespo-Diaz R, Behfar A, Terzic A (2008) CXCR4+/FLK-1+ biomarkers select a cardiopoietic lineage from embryonic stem cells. Stem Cells 26(6):1464–1473
Neumayer HH (2005) Introducing everolimus (Certican) in organ transplantation: an overview of preclinical and early clinical developments. Transplantation 79(9 Suppl):S72–S75
Okano T, Yamada N, Okuhara M, Sakai H, Sakurai Y (1995) Mechanism of cell detachment from temperature-modulated, hydrophilic-hydrophobic polymer surfaces. Biomaterials 16(4):297–303
Orlic D, Kajstura J, Chimenti S, Bodine DM, Leri A, Anversa P (2001) Transplanted adult bone marrow cells repair myocardial infarcts in mice. Ann N Y Acad Sci 938:221–229; discussion 229–230
Paquin J, Danalache BA, Jankowski M, McCann SM, Gutkowska J (2002) Oxytocin induces differentiation of P19 embryonic stem cells to cardiomyocytes. Proc Natl Acad Sci U S A 99(14):9550–9555
Potta SP, Liang H, Winkler J, Doss MX, Chen S, Wagh V, Pfannkuche K, Hescheler J, Sachinidis A (2010) Isolation and functional characterization of alpha-smooth muscle actin expressing cardiomyocytes from embryonic stem cells. Cell Physiol Biochem 25(6):595–604. doi:000315078 [pii], 10.1159/000315078
Roggia C, Ukena C, Bohm M, Kilter H (2006) Hepatocyte growth factor (HGF) enhances cardiac commitment of differentiating embryonic stem cells by activating PI3 kinase. Exp Cell Res 313(5):921–930
Sadler T (1998) Medizinische Embryologie – Die normale menschliche Entwicklung und ihre Fehlbildungen (trans: Drews U), 9th edn
Saga Y, Miyagawa-Tomita S, Takagi A, Kitajima S, Miyazaki J, Inoue T (1999) MesP1 is expressed in the heart precursor cells and required for the formation of a single heart tube. Development 126(15):3437–3447
Saga Y, Kitajima S, Miyagawa-Tomita S (2000) Mesp1 expression is the earliest sign of cardiovascular development. Trends Cardiovasc Med 10(8):345–352
Schachinger V, Erbs S, Elsasser A, Haberbosch W, Hambrecht R, Holschermann H, Yu J, Corti R, Mathey DG, Hamm CW, Suselbeck T, Assmus B, Tonn T, Dimmeler S, Zeiher AM (2006a) Intracoronary bone marrow-derived progenitor cells in acute myocardial infarction. N Engl J Med 355(12):1210–1221
Schachinger V, Erbs S, Elsasser A, Haberbosch W, Hambrecht R, Holschermann H, Yu J, Corti R, Mathey DG, Hamm CW, Suselbeck T, Assmus B, Tonn T, Dimmeler S, Zeiher AM, Investigators R-A (2006b) Intracoronary bone marrow-derived progenitor cells in acute myocardial infarction. N Engl J Med 355(12):1210–1221. doi:10.1056/NEJMoa060186
Sharma A, Li G, Rajarajan K, Hamaguchi R, Burridge PW, Wu SM (2015) Derivation of highly purified cardiomyocytes from human induced pluripotent stem cells using small molecule-modulated differentiation and subsequent glucose starvation. J Vis Exp 97: e52628. doi:10.3791/52628
Shiba Y, Hauch KD, Laflamme MA (2009) Cardiac applications for human pluripotent stem cells. Curr Pharm Des 15(24):2791–2806
Singh AM, Li FQ, Hamazaki T, Kasahara H, Takemaru KI, Terada N (2007) Chibby, an antagonist of the Wnt/{beta}-catenin pathway, facilitates cardiomyocyte differentiation of murine embryonic stem cells. Circulation 115(5):617–626
Singla DK, Long X, Glass C, Singla RD, Yan B (2011) iPS cells repair and regenerate infarcted myocardium. Mol Pharm. doi:10.1021/mp2001704
Soonpaa MH, Koh GY, Klug MG, Field LJ (1994) Formation of nascent intercalated disks between grafted fetal cardiomyocytes and host myocardium. Science 264(5155):98–101
Srivastava D, Ivey KN (2006) Potential of stem-cell-based therapies for heart disease. Nature 441(7097):1097–1099
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126(4):663–676
Takahashi T, Lord B, Schulze PC, Fryer RM, Sarang SS, Gullans SR, Lee RT (2003) Ascorbic acid enhances differentiation of embryonic stem cells into cardiac myocytes. Circulation 107(14):1912–1916
Theiss HD, Brenner C, Engelmann MG, Zaruba MM, Huber B, Henschel V, Mansmann U, Wintersperger B, Reiser M, Steinbeck G, Franz WM (2010) Safety and efficacy of SITAgliptin plus GRanulocyte-colony-stimulating factor in patients suffering from Acute Myocardial Infarction (SITAGRAMI-Trial)-rationale, design and first interim analysis. Int J Cardiol 145(2):282–284. doi:S0167-5273(09)01501-0 [pii], 10.1016/j.ijcard.2009.09.555
Theiss HD, Vallaster M, Rischpler C, Krieg L, Zaruba MM, Brunner S, Vanchev Y, Fischer R, Grobner M, Huber B, Wollenweber T, Assmann G, Mueller-Hoecker J, Hacker M, Franz WM (2011) Dual stem cell therapy after myocardial infarction acts specifically by enhanced homing via the SDF-1/CXCR4 axis. Stem Cell Res 7(3):244–255. doi:10.1016/j.scr.2011.05.003
Theiss HD, Gross L, Vallaster M, David R, Brunner S, Brenner C, Nathan P, Assmann G, Mueller-Hoecker J, Vogeser M, Steinbeck G, Franz WM (2013) Antidiabetic gliptins in combination with G-CSF enhances myocardial function and survival after acute myocardial infarction. Int J Cardiol 168(4):3359–3369. doi:10.1016/j.ijcard.2013.04.121
Tulloch NL, Muskheli V, Razumova MV, Korte FS, Regnier M, Hauch KD, Pabon L, Reinecke H, Murry CE (2011) Growth of engineered human myocardium with mechanical loading and vascular coculture. Circ Res 109(1):47–59. doi:CIRCRESAHA.110.237206 [pii], 10.1161/CIRCRESAHA.110.237206
Ventura C, Maioli M, Asara Y, Santoni D, Scarlata I, Cantoni S, Perbellini A (2004) Butyric and retinoic mixed ester of hyaluronan. A novel differentiating glycoconjugate affording a high throughput of cardiogenesis in embryonic stem cells. J Biol Chem 279(22):23574–23579
Ventura C, Maioli M, Asara Y, Santoni D, Mesirca P, Remondini D, Bersani F (2005) Turning on stem cell cardiogenesis with extremely low frequency magnetic fields. FASEB J 19(1):155–157
Wiese C, Nikolova T, Zahanich I, Sulzbacher S, Fuchs J, Yamanaka S, Graf E, Ravens U, Boheler KR, Wobus AM (2011) Differentiation induction of mouse embryonic stem cells into sinus node-like cells by suramin. Int J Cardiol 147(1):95–111. doi:S0167-5273(09)00871-7 [pii], 10.1016/j.ijcard.2009.08.021
Witty AD, Mihic A, Tam RY, Fisher SA, Mikryukov A, Shoichet MS, Li RK, Kattman SJ, Keller G (2014) Generation of the epicardial lineage from human pluripotent stem cells. Nat Biotechnol 32(10):1026–1035. doi:10.1038/nbt.3002
Wobus AM, Kaomei G, Shan J, Wellner MC, Rohwedel J, Ji G, Fleischmann B, Katus HA, Hescheler J, Franz WM (1997) Retinoic acid accelerates embryonic stem cell-derived cardiac differentiation and enhances development of ventricular cardiomyocytes. J Mol Cell Cardiol 29(6):1525–1539
Wollert KC, Meyer GP, Lotz J, Ringes-Lichtenberg S, Lippolt P, Breidenbach C, Fichtner S, Korte T, Hornig B, Messinger D, Arseniev L, Hertenstein B, Ganser A, Drexler H (2004) Intracoronary autologous bone-marrow cell transfer after myocardial infarction: the BOOST randomised controlled clinical trial. Lancet 364(9429):141–148
Wu SM (2008) Mesp1 at the heart of mesoderm lineage specification. Cell Stem Cell 3(1):1–2
Xu XQ, Graichen R, Soo SY, Balakrishnan T, Rahmat SN, Sieh S, Tham SC, Freund C, Moore J, Mummery C, Colman A, Zweigerdt R, Davidson BP (2008) Chemically defined medium supporting cardiomyocyte differentiation of human embryonic stem cells. Differentiation 76(9):958–970
Yamanaka S (2007) Strategies and new developments in the generation of patient-specific pluripotent stem cells. Cell Stem Cell 1(1):39–49
Yuasa S, Itabashi Y, Koshimizu U, Tanaka T, Sugimura K, Kinoshita M, Hattori F, Fukami SI, Shimazaki T, Okano H, Ogawa S, Fukuda K (2005) Transient inhibition of BMP signaling by Noggin induces cardiomyocyte differentiation of mouse embryonic stem cells. Nat Biotechnol 23(5):607–611
Zaffran S, Kelly RG, Meilhac SM, Buckingham ME, Brown NA (2004) Right ventricular myocardium derives from the anterior heart field. Circ Res 95(3):261–268
Zandstra PW, Bauwens C, Yin T, Liu Q, Schiller H, Zweigerdt R, Pasumarthi KB, Field LJ (2003) Scalable production of embryonic stem cell-derived cardiomyocytes. Tissue Eng 9(4):767–778
Zaruba MM, Theiss HD, Vallaster M, Mehl U, Brunner S, David R, Fischer R, Krieg L, Hirsch E, Huber B, Nathan P, Israel L, Imhof A, Herbach N, Assmann G, Wanke R, Mueller-Hoecker J, Steinbeck G, Franz WM (2009) Synergy between CD26/DPP-IV inhibition and G-CSF improves cardiac function after acute myocardial infarction. Cell Stem Cell 4(4):313–323
Zimmermann WH, Cesnjevar R (2009) Cardiac tissue engineering: implications for pediatric heart surgery. Pediatr Cardiol 30(5):716–723
Zweigerdt R, Burg M, Willbold E, Abts H, Ruediger M (2003) Generation of confluent cardiomyocyte monolayers derived from embryonic stem cells in suspension: a cell source for new therapies and screening strategies. Cytotherapy 5(5):399–413
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Brenner, C., David, R., Franz, WM. (2016). Stem Cells for Cardiovascular Regeneration. In: Steinhoff, G. (eds) Regenerative Medicine - from Protocol to Patient. Springer, Cham. https://doi.org/10.1007/978-3-319-27610-6_6
Download citation
DOI: https://doi.org/10.1007/978-3-319-27610-6_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-27608-3
Online ISBN: 978-3-319-27610-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)