Abstract
Most cochlear implants (CIs) stimulate the auditory nerve with trains of symmetric biphasic pulses consisting of two phases of opposite polarity. Animal and human studies have shown that both polarities can elicit neural responses. In human CI listeners, studies have shown that at suprathreshold levels, the anodic phase is more effective than the cathodic phase. In contrast, animal studies usually show the opposite trend. Although the reason for this discrepancy remains unclear, computational modelling results have proposed that the degeneration of the peripheral processes of the neurons could lead to a higher efficiency of anodic stimulation. We tested this hypothesis in ten guinea pigs who were deafened with an injection of sysomycin and implanted with a single ball electrode inserted in the first turn of the cochlea. Animals were tested at regular intervals between 1 week after deafening and up to 1 year for some of them. Our hypothesis was that if the effect of polarity is determined by the presence or absence of peripheral processes, the difference in polarity efficiency should change over time because of a progressive neural degeneration. Stimuli consisted of charge-balanced symmetric and asymmetric pulses allowing us to observe the response to each polarity individually. For all stimuli, the inferior colliculus evoked potential was measured. Results show that the cathodic phase was more effective than the anodic phase and that this remained so even several months after deafening. This suggests that neural degeneration cannot entirely account for the higher efficiency of anodic stimulation observed in human CI listeners.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Most contemporary cochlear implants (CIs) stimulate the auditory nerve with trains of symmetric biphasic pulses consisting of two phases of opposite polarity presented in short succession. Although both polarities can elicit neural responses (Miller et al. 1998, 1999; Klop et al. 2004; Undurraga et al. 2013), they are not equally efficient. Animal recordings performed at the level of the auditory nerve usually show longer latencies and lower thresholds for cathodic than for anodic stimulation (e.g., Miller et al. 1999). This observation is consistent with theoretical models of extracellular electrical stimulation of the nerve showing that a cathodic current produces a larger depolarization and elicits action potentials more peripherally than an anodic current of the same level (Rattay 1989).
In past years, studies performed with human CI listeners have shown different results. While at hearing threshold, no consistent difference between polarities has been observed, a large number of studies have reported that anodic stimulation is more effective than cathodic stimulation at suprathreshold levels (Macherey et al. 2006, 2008; Undurraga et al. 2013; Carlyon et al. 2013). This has been shown psychophysically in loudness balancing, pitch ranking and masking experiments and electrophysiologically in measures of electrically-evoked compound action potentials and auditory brainstem responses. Although this result contrasts with the conclusions of most animal reports, an exception is the study by Miller et al. (1998) who found a similar trend in their guinea pigs. Because the results of this specific study are at odds with other measurements performed in the guinea pig (Klop et al. 2004) and with all measurements performed in cats, the first aim of the present study was to clarify the effect of polarity in guinea pigs. This was done by measuring the inferior colliculus evoked potential in response to intracochlear electrical stimulation using various pulse shapes differing in polarity and in current level.
Although the reason for the higher sensitivity of human CI users to anodic stimulation remains unknown, predictions from two different -among the few- computational models of the implanted cochlea offer possible explanations (Macherey et al. 2006). First, the modeling results of Rattay et al. (2001) suggest that polarity sensitivity may strongly depend on the state of degeneration and demyelination of the peripheral processes of the neurons. Specifically, the model predicts lower anodic thresholds for degenerated peripheral processes. Although post-mortem studies have shown that such degeneration can occur in CI listeners, there is no currently available method to evaluate the presence of peripheral processes in patients. The second aim of this study was to investigate polarity sensitivity in guinea pigs as a function of time after deafening. Our reasoning was based on the observation that spiral ganglion neurons degenerate over time in deafened animals and that this degeneration first affects the peripheral processes (Koitchev et al. 1982; Cazals et al. 1983). If this degeneration is responsible for the higher anodic sensitivity, we hypothesized that the effect of polarity would change over time.
An alternative explanation for the higher anodic sensitivity at high current levels comes from a model of the implanted guinea-pig cochlea (Frijns et al. 1996). This model predicts that cathodic stimulation should elicit action potentials at the periphery of the neurons but that above a certain current level, the propagation of these action potentials may be blocked by the hyperpolarization produced more centrally on the fibers. This blocking was not observed in computer simulations using anodic stimuli because action potentials were in this case elicited at a more central locus on the fibers. A third aim of the present study was to investigate if this behavior could be observed at the level of the inferior colliculus of the guinea pigs. If, at some level in the dynamic range, action potentials are being blocked for cathodic stimulation, this should be reflected by a slower rate of increase of the neural response for cathodic than for anodic stimulation and/or potentially by a non-monotonic growth of response amplitude as a function of current level.
Finally, most animal studies on polarity sensitivity have been performed using monophasic pulses which cannot safely be used in humans. This is why studies in CI subjects use charge-balanced asymmetric waveforms which are hypothesized to make one polarity dominate over the other. Some of these waveforms have the advantage of requiring lower current levels than the commonly-used symmetric waveform to elicit auditory sensations in implant listeners (Macherey et al. 2006). The fourth aim of the present study was to evaluate the efficiency of these different pulse shapes and to compare it to human psychophysical data obtained previously.
2 Methods
2.1 Animal Preparation
Ten Albino guinea pigs (G1–G10) of the Dunkin-Hartley strain were used in these studies. The experimental procedures were carried out in accordance with French national legislation (JO 87–848, European Communities Council Directive (2010/63/EU,74) and local ethics committee “Direction Départementale de la Protection des Populations”, with permit number 00002.01.
In a first step, using stereotaxic procedures, the animals were chronically implanted with an electrode in the right inferior colliculus (IC). This technique allows easy and stable recordings over months as indicated in several previous experiments (e.g., Popelár et al. 1994). Using a micromanipulator, a Teflon-coated platinum electrode (127 mm in diameter obtained from A-M systems) was lowered into the IC. Then the electrode was fixed to the skull with acrylic cement and soldered to a small connector to which two screws in the frontal bones were also connected and served as reference and ground, respectively. The ensemble was finally fixed to the surface of the skull with acrylic cement.
After a recovery period of about 1 week, the animal was sedated with xylazine (5 mg/kg), a small loudspeaker (MF1 from TDT) was placed at 1 cm from its left ear pinna and the animal’s head connector was linked to the input of an amplifier (A-M systems model 1700). Tone pips with 2 ms linear rise/fall times at octave frequencies between 2 to 32 kHz were presented at levels of 90–0 dB SPL in 10-dB steps. In response to sound stimuli, electrophysiological responses were amplified 10,000 times and filtered between 100 and 5 kHz, then they were averaged over 100–300 times (CED 1401 A/D converter).
About a week later, the animal was anesthetized and the left ear bulla was approached from behind the pinna. The round window was gently pierced and a mixture of sisomycin at a concentration of 50 mg/ml was injected into the round window niche until the bulla was completely filled with the mixture. The animal was left so for 2 h and the sisomycin was then aspirated from the bulla. This sisomycin treatment is known from previous experiments (Cazals et al. 1983) to completely destroy cochlear hair cells and lead to progressive spiral ganglion cells degeneration over months. Then a teflon-coated platinum wire (200 mm in diameter) with a tip bare over about 0.5 mm was introduced through the round window over a length of about 2 mm (about the 16–32 kHz tonotopy). Because of deficiencies in handling and care in animals’ quarter during the months of the experiment, for some animals, the head connector came off the skull, and the animal was operated again, sometimes replacing the cochlear implant electrode.
2.2 Stimuli
During the experiment, electrical stimulation was delivered using a stimulus isolator current generator (AM systems model 2200), the output of which was connected through the head connector to the cochlear implant electrode (active) and to one screw in the frontal bone (reference). This generator delivered a current proportional to an arbitrary input voltage waveform. The voltage waveform was the output of an Advanced Bionics HiRes90k test implant connected to a purely resistive load. This allowed us to use pulse shapes similar to those used in previous human CI studies. The stimuli were designed and controlled using the BEDCS software (Advanced Bionics).
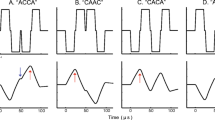
Stimuli were trains of electrical pulses delivered at a rate of 8 Hz. The four pulse shapes illustrated in Fig. 1 were tested, including symmetric biphasic (SYM), reversed pseudomonophasic (RPS), reversed pseudomonophasic with an interphase gap of 6.5 ms (RPS-IPG) and triphasic (TRI) pulses. The top pulse shapes are assumed to favor cathodic stimulation, hence the “C” at the end of their acronyms while the bottom pulse shapes are assumed to favor anodic stimulation, hence the “A”. The recordings were synchronized to the onset of the cathodic phase for the top pulse shapes and to the onset of the anodic phase for the bottom pulse shapes.
Schematic representation of the electrical pulse shapes used for stimulation, including symmetric biphasic cathodic-first (SYM-C) and anodic-first (SYM-A), Reversed pseudomonophasic with a short, high-amplitude cathodic (RPS-C) or anodic (RPS-A) phase, Reversed pseudomonophasic with a 6.5-ms interphase gap (RPS-IPG-C and RPS-IPG-A) and triphasic with a cathodic (TRI-C) or anodic (TRI-A) central phase
The duration of the phases of SYM, of the short, high-amplitude phases of RPS and RPS-IPG and of the central phase of TRI was 86 μs. The long, low-amplitude phases of RPS and RPS-IPG were 8 times longer and lower in amplitude to maintain the total charge balanced. The first and third phases of TRI had a duration of 43 μs and the same amplitude as the central phase.
2.3 Sessions
Over months, several sessions of electrical stimulation from the cochlear implant and recording from the right IC were performed. Each session lasted about 1 h during which the animals were sedated. In each session and for each pulse shape, a threshold level was first defined as the level producing a detectable IC evoked potential after averaging; the maximum level of stimulation was defined as the level triggering a detectable motor response of the animal’s left cheek. Between these two values, a series of current levels was defined in 1.5-dB steps. For each pulse shape and each level tested, the recordings alternated between polarities. Between 50 and 100 averages were obtained for each combination of pulse shape, polarity and level and the corresponding IC evoked potentials were recorded.
3 Results
3.1 Morphology and Response Amplitude of the IC Evoked Potential
The bottom traces in Figs. 2a and b show examples of IC evoked potentials obtained for subject G2 2 weeks after deafening in response to RPS-IPG-C and RPS-IPG-A, respectively. For comparison, the top traces in each panel show recordings obtained from the same subject in response to 16-kHz tone pips before deafening.
a Inferior colliculus evoked potential obtained 2 weeks after deafening for G2 in response to RPS-IPG-C pulses. The top trace shows the response obtained before deafening for an acoustic tone pip presented at 90 dB SPL. b Same as a for RPS-IPG-A and for a 60-dB SPL tone pip. c Response amplitude (N1-P2) as a function of current level for G2 2 weeks after deafening. d Same as c 7 months after deafening
The morphology of the response was similar for acoustic and electric stimulation and consisted of a first positive peak (P1) followed by a negative peak (N1) and a second positive peak (P2). The amplitude of the response was arbitrarily defined as the difference in voltage between the negative peak and the second positive peak. The response amplitude varied from about 20 to 600 μV depending on subjects and pulse shapes. The dynamic range, as defined by the difference between the maximum stimulation level and the minimum level that produced a visually detectable response ranged from about 3 to 18 dB, which compare well with usual values of dynamic range for individual CI electrodes.
Figure 2c shows the response amplitude as a function of current level for the same subject and the same pulse shape. It can be seen that at all current levels, the response was larger for the cathodic (RPS-IPG-C) than for the anodic (RPS-IPG-A) pulse. As depicted in the next section, this trend was observed in most of our subjects. Because the RPS-IPG stimulus had a large interphase gap between the two phases, the responses illustrated here presumably represent the responses to each individual polarity. Input/output growth functions from the same animal collected 7 months after deafening are shown in Fig. 2d. The effect of polarity was consistent with that obtained just after deafening. Matching the response amplitudes obtained for electrical stimulation to those measured acoustically before deafening, the difference in response amplitude between the two polarities was, for this animal, equivalent to an acoustic level difference of about 30 dB.
Figure 3 shows the amplitude of the response for cathodic stimulation as a function of that for anodic stimulation for the four pulse shapes SYM, RPS, RPS-IPG and TRI and all subjects and levels tested. A very large majority of the data points lie above the diagonal. This illustrates that so-called cathodic pulses usually produced a larger response than anodic pulses for all pulse shapes. For subjects G8-G10 who had several electrodes implanted in the IC, only the recordings from the electrode giving the largest responses are shown. However, the effects of pulse shape and polarity remained consistent across the different recording sites.
3.2 Effect of Polarity as a Function of Time
For a given pulse shape and session, the input/output growth functions obtained for the two polarities were analyzed to determine the current levels needed for each of them to reach an arbitrarily-defined IC response amplitude. This arbitrary amplitude was equal to 50 % of the maximum response amplitude obtained for this particular pulse shape, subject and session and could, therefore, differ from one session to the next.
Figure 4a shows the difference in current level between RPS-C and RPS-A when both produced the same response amplitude. This quantity referred to as the “polarity effect” is illustrated as a function of time after deafening for the different subjects. Although only the data for RPS are shown, similar results were obtained for RPS-IPG and TRI. If neural degeneration had an effect on polarity sensitivity, we would expect the polarity effect to increase over time and to reach positive values when fibers are degenerated. This does not seem to be the case as the level difference between polarities rather tends to decrease right after deafening for several subjects and then to remain roughly constant.
a Difference in current level between RPS-C and RPS-A required to obtain the same response amplitude in the IC. b Star symbols: mean polarity effect across guinea-pigs subjects (+/‑1 s.e.). Filled symbols: mean polarity effects obtained at most comfortable loudness for similar pulse shapes in previous human studies using electrodes in the middle of the array. Squares: Macherey et al. 2006; circles: Undurraga et al. 2013; triangles: Carlyon et al. 2013; diamonds: Macherey et al. 2008. Human data were obtained with non-reversed versions of RPS and RPS-IPG, meaning that the short, high-amplitude phase was presented first. The three data points for comparison with TRI correspond to the ‘TP’ condition of Carlyon et al. 2013 for Advanced Bionics (1st point) and Med-EL (2nd point) subjects and to their ‘QP’ condition for Cochlear subjects (3rd point). c Example of non-monotonic growth function obtained for RPS in G7. d Difference in current level between each cathodic asymmetric pulse shape and SYM-C needed to obtain the same response amplitude. Star symbols: results of the present study averaged across subjects (+/‑1 s.e.). Filled symbols show corresponding human psychophysical data measured at most comfortable loudness for anodic pulse shapes. Triangles correspond to the TP condition for Advanced Bionics (1st point) and Med-EL (2nd point) subjects
Given the absence of an effect of duration of deafness, the polarity effects obtained in all sessions were averaged for each animal and each pulse shape. As shown by the star symbols in Fig. 4b, the magnitude of the polarity effect differed across pulse shapes. The mean across subjects was ‑1.4, ‑3.1, ‑5.3 and ‑2.3 dB for SYM, RPS, RPS-IPG and TRI, respectively. Paired-sample t-tests with Bonferroni correction showed that this effect was larger for RPS than for SYM (t(8) = 3.6, p < 0.05) and also for RPS-IPG than for SYM (t(9) = 3.7, p < 0.05). For comparison, filled symbols show the polarity effects obtained using loudness-balancing experiments in human CI listeners for similar pulse shapes (Macherey et al. 2006, 2008; Undurraga et al. 2013; Carlyon et al. 2013).
Although the polarity effect is opposite in sign for the two species, it is interesting to note that its magnitude is similar for RPS and for TRI. For RPS-IPG, the smaller effect obtained psychophysically may be due to the fact that the delayed long, low-amplitude phase contributed to perception whereas it did not contribute to the neural responses recorded here.
Closer inspection of the data revealed that the input/output growth functions sometimes had non-monotonic shapes. An example obtained for G7 1 month after deafening is illustrated in Fig. 4c for RPS-A and RPS-C. The function for RPS-C shows, at some point in the dynamic range, a decrease in response amplitude with increases in level. In this case, the cathodic pulse was more effective than the anodic pulse at low levels but less effective at high levels. Such non-monotonicities were observed in seven of the ten subjects but only in about 13 % of the cathodic and 4 % of the anodic growth functions. Furthermore, they did not always occur for the same pulse shape across sessions.
3.3 Effect of Pulse Shape
The difference in level between SYM-C and the various asymmetric pulse shapes RPS-C, RPS-IPG-C and TRI-C when they all produced the same response amplitude was determined. The response amplitude at which the pulse shapes were compared was equal to 50 % of the maximum amplitude of SYM-C. Given there was no effect of duration of deafness on the magnitude of these effects, the data were averaged across sessions for each animal and analyzed in a one-way repeated measures ANOVA. The star symbols in Fig. 4d show the mean across animal subjects. The effect of pulse shape was highly significant (F(3,18) = 22.76, p < 0.001). RPS-IPG required 2.2 dB less current than SYM (p < 0.05) while TRI required 2.7 dB more. There was no significant difference between SYM and RPS. For comparison, the filled symbols show the effect of pulse shape as observed in human CI listeners. For these data, however, the level difference was obtained using loudness balancing between anodic pulse shapes. Here again, the magnitude of the effect of pulse shape is similar in both species.
4 Discussion
Most guinea-pigs tested in the present study showed higher sensitivity to cathodic stimulation. This effect of polarity was consistent across all levels tested, ranging from threshold to maximum level. This observation corroborates the results obtained in most previous animal experiments performed in cats and guinea pigs.
Duration of deafness did not influence the effect of polarity, suggesting that neural degeneration may not entirely account for the higher sensitivity to anodic stimulation observed in humans. However, the morphology of the nerve fibers and the cochlear geometry are different in the two species and may play an additional role on neural excitation (Rattay et al. 2001).
In few cases, input/output growth functions were non-monotonic, showing a decrease in response amplitude with increases in level. Similar non-monotonic functions have already been observed at the level of the auditory nerve (Miller et al. 1998). They may reflect the blocking of action potentials along the nerve fibers (Frijns et al. 1996) and, therefore, remain a possible candidate explanation for the higher efficiency of anodic stimulation at suprathreshold levels in humans.
Finally, the difference in efficiency between the various pulse shapes was consistent with results obtained in previous animal and human experiments (Miller et al. 2001; Carlyon et al. 2013), showing that, to elicit responses of equal amplitude, pseudomonophasic pulses with a gap between the two phases require a lower current level than symmetric pulses while triphasic pulses require a higher current level.
References
Carlyon RP, Deeks JM, Macherey O (2013) Polarity effects on place pitch and loudness for three cochlear-implant designs and at different cochlear sites. J Acoust Soc Am 134(1):503–509
Cazals Y, Aran JM, Charlet de Sauvage R (1983) Artificial activation and degeneration of the cochlear nerve in guinea pigs. Arch Otorhinolaryngol 238(1):1–8
Frijns JH, de Snoo SL, ten Kate JH (1996) Spatial selectivity in a rotationally symmetric model of the electrically stimulated cochlea. Hear Res 95(1–2):33–48
Klop WM, Hartlooper A, Briaire JJ, Frijns JH (2004) A new method for dealing with the stimulus artefact in electrically evoked compound action potential measurements. Acta Otolaryngol 124(2):137–143
Koitchev K, Guilhaume A, Cazals Y, Aran JM (1982) Spiral ganglion changes after massive aminoglycoside treatment in the guinea pig. Counts and ultrastructure. Acta Otolaryngol 94(5–6):431–438
Macherey O, Carlyon RP, van Wieringen A, Deeks JM, Wouters J (2008) Higher sensitivity of human auditory nerve fibers to positive electrical currents. J Assoc Res Otolaryngol 9(2):241–251
Macherey O, van Wieringen A, Carlyon RP, Deeks JM, Wouters J (2006) Asymmetric pulses in cochlear implants: effects of pulse shape, polarity, and rate. J Assoc Res Otolaryngol 7(3):254–266
Miller CA, Abbas PJ, Robinson BK, Rubinstein JT, Matsuoka AJ (1999) Electrically evoked single-fiber action potentials from cat: responses to monopolar, monophasic stimulation. Hear Res 130(1–2):197–218
Miller CA, Abbas PJ, Rubinstein JT, Robinson BK, Matsuoka AJ, Woodworth G (1998) Electrically evoked compound action potentials of guinea pig and cat: responses to monopolar, monophasic stimulation. Hear Res 119(1–2):142–154
Miller CA1, Robinson BK, Rubinstein JT, Abbas PJ, Runge-Samuelson CL (2001) Auditory nerve responses to monophasic and biphasic electric stimuli. Hear Res 151:79–94
Popelár J, Erre JP, Aran JM, Cazals Y (1994) Plastic changes in ipsi-contralateral differences of auditory cortex and inferior colliculus evoked potentials after injury to one ear in the adult guinea pig. Hear Res 72(1–2):125–134
Rattay F (1989) Analysis of models for extracellular fiber stimulation. IEEE Trans Biomed Eng 36(7):676–682
Rattay F, Lutter P, Felix H (2001) A model of the electrically excited human cochlear neuron. I. Contribution of neural substructures to the generation and propagation of spikes. Hear Res 153(1–2):43–63
Undurraga JA, Carlyon RP, Wouters J, van Wieringen A (2013) The polarity sensitivity of the electrically stimulated human auditory nerve measured at the level of the brainstem. J Assoc Res Otolaryngol 14(3):359–377
Acknowledgments
The authors acknowledge funding from the Agence Nationale de la Recherche (grant numbers ANR-11-PDOC-0022 and ANR-TECSAN-040-STESU).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
<SimplePara><Emphasis Type="Bold">Open Access</Emphasis> This chapter is distributed under the terms of the Creative Commons Attribution-Noncommercial 2.5 License (http://creativecommons.org/licenses/by-nc/2.5/) which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.</SimplePara> <SimplePara>The images or other third party material in this chapter are included in the work's Creative Commons license, unless indicated otherwise in the credit line; if such material is not included in the work's Creative Commons license and the respective action is not permitted by statutory regulation, users will need to obtain permission from the license holder to duplicate, adapt or reproduce the material.</SimplePara>
Copyright information
© 2016 The Author(s)
About this paper
Cite this paper
Macherey, O., Cazals, Y. (2016). Effects of Pulse Shape and Polarity on Sensitivity to Cochlear Implant Stimulation: A Chronic Study in Guinea Pigs. In: van Dijk, P., Başkent, D., Gaudrain, E., de Kleine, E., Wagner, A., Lanting, C. (eds) Physiology, Psychoacoustics and Cognition in Normal and Impaired Hearing. Advances in Experimental Medicine and Biology, vol 894. Springer, Cham. https://doi.org/10.1007/978-3-319-25474-6_15
Download citation
DOI: https://doi.org/10.1007/978-3-319-25474-6_15
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-25472-2
Online ISBN: 978-3-319-25474-6
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)