Abstract
Forests are one of the most important habitats for insectivorous bats as they offer the potential for both roosting and foraging. We reviewed silvicultural literature from North America, Australia, and Europe and found that diverse research approaches have revealed commonalities in bat responses to forest silviculture . Almost all silvicultural treatments evaluated were compatible with some use by forest bats, though different bat ensembles respond in different ways. Ensemble ecomorphology was a consistent predictor of how bats respond to vegetative clutter and its dynamic changes as forests regenerate and develop a dense structure following harvesting. Sustaining high levels of bat diversity in timber production forests requires a mix of silvicultural treatments and exclusion areas staggered across the landscape, regardless of forest type or geographic region. Use of edge habitats, exclusion areas/set-asides, and riparian corridors for roosting and foraging by bats were consistent themes in the literature reviewed, and these habitat elements need to be considered in forest planning. Densities of hollow or dead trees sufficient to support large populations of roosting bats are unknown and remain a major knowledge gap, but will likely be species contingent. New paradigm shifts in forest management away from the use of even-aged systems to multi-spatial scale retention of mature forest including trees with cavities should be beneficial to bats, which are influenced by landscape-scale management. Such an approach is already in use in some regions, though there is a limited guidance on what constitutes a reasonable landscape threshold for retention. The effectiveness of such an approach will require long-term monitoring and research, especially with population studies which are currently lacking.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
1 Introduction
Forests are one of the most important habitats for bats as they offer the potential for both roosting and foraging, and most species are reliant on forests for at least some part of their life cycle. Humans are also heavily reliant on the resources produced by forests, in particular timber. Consequently, forests are highly managed and modified in many areas. Understanding the effect that human manipulation of forested landscapes has on the resources required by bats is therefore of great importance to their conservation.
The use of silvicultural techniques to manipulate tree stands for timber production or biodiversity conservation goals presents several challenges. Forest bats are mobile and, as with forest birds, can use a large three-dimensional space to meet their life requisites (Kroll et al. 2012). Therefore, stand-level considerations alone are insufficient in sustaining habitat conditions for many forest bats as landscape-level needs are of equal or greater concern (Duchamp et al. 2007). Secondly, forest bats require roosting sites, high-quality foraging habitats, drinking sites, and features that provide connectivity among landscape elements. Providing all of these habitat requirements for an entire assemblage of bats simultaneously on a managed forested landscape is a difficult challenge, necessitating hierarchical approaches that assess spatial juxtaposition of habitat elements on the landscape and that implement silvicultural systems using multiple treatments applied both within and among stands.
Silvicultural practices vary greatly around the world. For example, in the northern hemisphere, clear felling typically results in cleared areas of 40–180 ha surrounded by relatively even-aged forests (Thomas 1988; Grindal and Brigham 1999; Swystun et al. 2001). In parts of Europe and North America, however, patch sizes are considerably smaller and some countries have abandoned clear felling altogether, favouring a more selective logging approach. Similarly, in parts of Australia, where broad scale clear-fall techniques are not utilised, selective logging results in a multi-aged forest (Nicholson 1999).
A key feature of insectivorous bats is their sophisticated sensory system, which enables them to navigate and forage in the dark. The foraging efficiency of echolocating bats is constrained by variations in vegetation because the echoes returning from prey need to be distinguished from background echoes returning from vegetation. These ‘clutter’ echoes can mask the echoes of prey making foraging inefficient in situations where vegetation is dense (Schnitzler et al. 2003). Forest bat species differ in echolocation signal design and wing morphology and this influences their flight behaviour and their tolerance to clutter, allowing classification into three broad foraging ensembles: (1) closed-space species are slow flying and highly manoeuvrable bats that can forage close to vegetation; (2) edge-space species exploit edge habitat and other linear features; and (3) open-space foragers have lower manoeuvrability and fly faster above the forest canopy or within large gaps in the forest. Changes to forest structure that influence the degree of clutter can, therefore, alter the availability of foraging habitat for each ensemble.
Our aim in this chapter was to explore how insectivorous bats respond to different silvicultural approaches used in forests around the world, incorporating studies within natural, or semi-natural, forests to intensive management within plantation forestry. We focus on three broad areas: North America, Australasia (including New Zealand), and Europe and refer the reader to Meyer et al. (2016) (Chap. 3) for tropical forests. While the majority of studies included in this review are published in scientific journals, we also include information from the grey literature (e.g. reports, conference proceedings, and unpublished theses) and some unpublished data where appropriate.
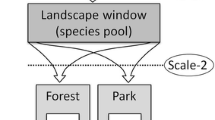
We look to highlight both commonalities and differences in the various approaches to the issue in different regions. We suggest that ecomorphology is one of the keys to understanding how bats use their environment and we use ecomorphological traits as a framework for predicting how the three broad functional ensembles of bats respond to forest logging (Hanspach et al. 2012; Luck et al. 2013). Conceptual models have been proposed previously for the relationship between the abundance of bats and key ecological resources manipulated by forest management (Fig. 5.1; Hayes and Loeb 2007). These posit the influence of thresholds for certain variables such as water availability, where further increases do not result in increased bat abundance. We assess the extent to which these models fit current data and extend them to (1) consider the time since logging as a response variable and (2) include an ecomorphological framework for the response of bats. We emphasise the importance of a long-term perspective when assessing bat responses in forests given that forests are long-lived ecosystems that undergo dynamic changes after disturbance. Finally, we consider the merits of multi-spatial scale management for bats and recommend future areas of research to advance the effective management of this diverse and functionally important group. There is some specialised terminology within this chapter that may be unfamiliar to those new to silvicultural literature, so we have provided a glossary at the end of the chapter with definitions. While the term woodland is often used to describe vegetation communities comprising trees but with a more open and lower canopy cover than forests, this definition varies by country. Here, we use the term forest to encompass the various definitions of woodland.
Conceptual models illustrating hypothesised relationships among the abundance of bats and ecological resources within forests (Hayes and Loeb 2007)
2 Major Forest Areas
2.1 North America
Management of forests in North America is undergoing a renaissance, of sorts, as threats associated with habitat loss and fragmentation, climate change, increased fire frequency, and introduction of forest insect pests are leading to paradigm shifts in how forests should be managed to sustain biodiversity, increase carbon sequestration, and maintain the capacity for resource extraction (Boerner et al. 2008; Parks and Bernier 2010; Moore et al. 2012). Historically, even-aged management was practiced across the continent with clearcuts, shelterwood cuts, seed-tree cuts, and deferment cuts all used in management of forests regardless of region or forest type. These practices have reached their zenith in south-eastern pine plantations where production forestry has led to short rotation harvests of monotypic stands of loblolly pine, Pinus taeda (Wear and Greis 2013). More recently, silvicultural approaches have focused on mimicking natural disturbance events or ecologically based forestry (Mitchell et al. 2002; Long 2009), resulting in application of uneven-aged or multi-aged silvicultural systems (O’Hara 2002, 2009), and prescribed fires (Boerner et al. 2008), in both pine and hardwood forests. North America is >24 million km2 in total land surface and lies entirely within the northern hemisphere. The continent supports a rich diversity of plant species across eight major forest types (Young and Geise 2003, Fig. 5.2) with each type encompassing from 1 to 8 subtypes (SAF 2010). Latitude plays a prominent role in the distribution of forest types across the continent, with a north-to-south pattern of northern coniferous, northern hardwood, central broad-leaved, oak–pine, bottomland hardwood, and tropical forests (Young and Geise 2003). Two other forest types, Pacific coast and Rocky Mountain, are distributed largely in a north–south direction paralleling several mountain ranges and, thus, cross a greater expanse of latitudes. The northern coniferous and boreal forest, dominated by spruce, fir, and larch, covers the largest extent of North American land surface of any forest type, followed by Rocky Mountain and central broad-leaved forests. Rocky Mountain forests are dominated by pines across much of their range, with central broad-leaved forests supporting oaks, hickories, maple, and beech. Bottomland hardwoods, comprising gums, bald cypress, oaks, and willows, represent the smallest land area of any major forest type in North America (1.25 million ha remaining; Mississippi Museum of Natural Science 2005). Globally, North America has experienced some of the greatest forest losses with a 5.1 % decline in forest land cover from 2000 to 2005 (Hansen et al. 2010). Declines in forest cover have been greatest in the south-eastern USA, where 3.5 million ha have been lost from 1992 to 2001 (World Resources Institute 2014). Recent shifts in the region-wide approach to management of south-eastern bottomland hardwood forests, however, have brought about a reversal in the trend of loss of these forests (USDA Forest Service 2009; Miller et al. 2011).
a Standing dead ponderosa pine (Pinus ponderosa) used as a roost tree by long-legged myotis (Myotis volans) in Oregon, b forested landscape treated using clearcut logging in Idaho with natural regeneration present, c stand of dead trees in California typical of habitats used by bark- and cavity-roosting bats in western coniferous forests, and d bottomland hardwood forest in Western Kentucky, with hollow roost tree of Rafinesque’s big-eared bat (Corynorhinus rafinesquii) in the centre. Photograph credits M. Baker (a), M. Lacki (b, c), and J. Johnson (d)
2.2 Europe
Europe consists of 50 countries and is just over 10 million km2 in land area. Forests cover approximately 45 % of the land area, most of which is found within the Russian Federation which comprises 40 % of the land area of Europe (FAO 2012). Europe’s native forest is very diverse with 13 broad categories encompassing 74 types (EEA 2006). Boreal forest consisting primarily of spruce or pine species dominates in northerly latitudes that comprise Scandinavia (Fig. 5.3). This is replaced by hemiboreal forest and nemoral coniferous and mixed broad-leaved/coniferous forest in southern Sweden and much of eastern central Europe, with alpine coniferous forest along the mountain ranges. Moving west, mesophytic deciduous and beech forest dominates, but there is increasing amounts of plantation forest. In the southern parts of Europe coniferous (pines, firs, junipers, cypress, cedar), broadleaved (oak, chestnut) and evergreen broadleaved forests are the main wooded habitats. Parts of Europe have undergone extensive deforestation and cover has been fragmented and depleted for several centuries. While 26 % of Europe’s forest area is classified as primary, this falls to <3 % excluding the Russian Federation, and approximately 52 % of all forests in Europe are now designated primarily for production (FAO 2012). In Europe, as in North America and Australia, there is growing interest in silvicultural practices that mimic natural forest ecosystem processes with the aim of developing mixed, structurally diverse stands (Lähde et al. 1999). This is a result of a move away from treating forests, particularly plantations, solely as a resource for timber, and an increased emphasis on sustainable management for multiple objectives including biodiversity conservation and recreation (Mason and Quine 1995). In practice, this has meant a reduction in clear felling, although this varies greatly between countries. For example, it has been largely phased out in Switzerland and Slovenia, but is still the primary form of logging in the UK (Fries et al. 1997; Mason et al. 1999), but recent modifications include retaining stands with longer rotations where possible (Mason and Quine 1995), reducing the removal of deadwood (Humphrey and Bailey 2012), and techniques geared to mimic natural disturbance such as prescribed burning.
a New Forest, United Kingdom: wood pasture, a historical European land management system providing shelter and forage for grazing animals as well as timber products, b double-leadered Corsican pines (Pinus nigra ssp. laricio) are used as roost sites by Natterer’s bats (M. nattereri) in Tentsmuir forest in Scotland, UK; c wooded landscape, including olive groves, used extensively in southern Italy by Rhinolophus euryale; d typical Bechstein’s bat (Myotis bechsteinii) foraging habitat in England, UK: a mixture of oak (Quercus robur) and hazel (Corylus avellana) woodland. Photograph credits J Sjolund, G Mortimer (b), D Russo (c), F Greenaway (d)
2.3 Australia
It is estimated that forests covered about a third of the Australian continent at the time of European settlement in 1788, but by the mid-2000s this had been reduced to about 19 % cover (Montreal Process Implementation Group for Australia 2008). Five million hectares of forest are classified as old growth (22 %) and over 70 % of these occur in conservation reserves. Timber harvesting on public land is now restricted to 9.4 million ha, or about 25 % of the areas potentially suitable for timber production, and much of this has been previously logged. Eucalypts dominate the forests of Australia, and they are highly diverse comprising 500–600 species (Fig. 5.4, Florence 1996). Eucalypt forests range from those with a high diversity of eucalypt species to those dominated by one or a few species, the latter most often occurring in the tall wet forests of temperate southern Australia, including Tasmania (Florence 1996). These different eucalypt species and forest communities grow on different soils, under varying climates and natural disturbance regimes that in turn influence the variety of silvicultural practices applied. Fire is also a driving force behind the distribution and composition of eucalypt forests, and it occurs as massive wildfires that sweep across the landscape and less intensive prescribed burns that aim to reduce fuel loads and minimise damaging wildfires. To some extent, silvicultural practices aim to mimic these disturbance events and maximise regeneration after harvest.
Eucalypt forests of Australia: a narrow vehicle tracks through regrowth wet sclerophyll forest are used extensively by bats; b recently thinned regrowth forest potentially increases flight space and foraging opportunities for bats; c senescing crown of a Blackbutt Eucalyptus pilularus supports multiple hollow branches where bats, including maternity colonies, selectively roost; d an old-growth, spotted gum forest, Corymbia maculata, supports high densities of hollows and an open zone above a dense understorey/shrub layer, providing a variety of niches for foraging and roosting bats. Photographs B. Law
Silviculture of Australian eucalypts is thus highly variable, although the techniques applied largely resemble those used elsewhere around the globe. For example, silviculture varies from clearcut practices in the tall wet eucalypt forests of temperate southern Australia (Tasmania and Victoria) to group selection and single tree selection in warm temperate and subtropical areas to the north. Clearcuts aim to mimic broadly the massive stand replacement events created by wildfires, which are an irregular feature of tall eucalypt forests in Australia. However, one important difference between clearcuts and wildfires is that wildfires leave legacies in the form of dead trees with hollows that can remain standing for decades. Regrowth after harvesting may take many decades to self-thin sufficiently for the forest to begin to resemble the openness of mature or unlogged forest (Florence 1996). Selective logging can occur at a range of intensities that are almost a continuum from very low levels of tree removal targeting specific size/species of trees with ~10 % of tree basal area removed to almost a seed-tree retention silviculture with >60 % of stand basal area removed. In selectively harvested forests, nominal ‘rotations’ are about 60–80 years though these develop from repeated logging visits to the same coupes every 10–30 years to produce a dynamic of multi-aged mosaics of even-aged regeneration cohorts (Curtin et al. 1991). Selective logging is most commonly applied to forests comprising mixed eucalypt species and uneven ages. Rainforest has a restricted occurrence in Australia, and logging of this forest type is no longer permitted.
3 Complexity of Bat Habitat Needs
3.1 Mature, Large Diameter Trees
Older age classes of trees, especially old-growth forests , have historically been viewed as important habitats for bats (Altringham 1996; Fisher and Wilkinson 2005; Hayes and Loeb 2007) and are likely to contain a greater diversity and abundance of insect prey (e.g. Fuentes-Montemayor et al. 2012; Lintott et al. 2014). Early studies demonstrated variation in bat activity across stands of different age classes, with the levels of bat activity higher in older, mature stands than young stands (Thomas 1988; Erickson and West 1996; Crampton and Barclay 1998; Law and Chidel 2002). Older forests possess canopies that are more fully developed than regenerating or early-seral forests, with complex crown architecture (Wunder and Carey 1996). Old-growth forests are also likely to contain a larger number of microhabitats which are associated with higher bat species richness and higher levels of activity in common and Nathusius pipistrelles, Pipistrellus pipistrellus and P. nathusii, in oak, Quercus spp., forests in southern France managed for coppice (Regnery et al. 2013a). In a parallel study, time since cutting was the best predictor of the number of tree microhabitats which were 13 times more abundant in stands >90 years post-cutting, than those <30 years in age (Regnery et al. 2013b).
Considerable research has been undertaken on roost selection since pioneering radio-tracking studies in Australia (Lunney et al. 1988; Taylor and Savva 1988). A consistent trend throughout the world is that most bats prefer to roost in larger diameter trees (>30 cm, Russo et al. 2004; ~80 cm, Baker and Lacki 2006; see also Kalcounis-Rüppell et al. 2005), often in older forest stands or mature forests (Lunney et al. 1988; Taylor and Savva 1988; Brigham et al. 1997; Betts 1998; Crampton and Barclay 1998; Sedgeley and O’Donnell 1999; Law and Anderson 2000; Lumsden et al. 2002; Mazurek and Zielinski 2004; Russo et al. 2004, 2010; Ruczyński et al. 2010). Such trees have a greater likelihood of supporting larger populations of roosting bats and persist for longer than smaller diameter dead trees (Lacki et al. 2012); thus, their identification and provision in residual patches during timber harvesting is important. Where mature forest is absent across large areas at least some species find roosts in scattered hollow trees in regrowth forest where habitat trees were not specifically retained, indicating that bats typically roost in the largest available trees. One Australian study found that the 4-g eastern forest bat, Vespadelus pumilus, which ranges over relatively small areas, maintains similar sizes of maternity colonies in the scarce roosts remaining within regrowth forest compared to maternity colonies in old-growth forest (Law and Anderson 2000). Russo et al. (2010) found evidence of roost selection flexibility in barbastelle bats, Barbastella barbastellus; dead and dying trees, a favoured roost site for this species, were six times more common in unmanaged than managed European beech, Fagus sylvatica, forests in central Italy. Bats, however, were able to roost within managed forest, albeit in smaller numbers by exploiting roost sites in live trees and rock crevices. Few studies have investigated roost selection in younger forest where roosts are scarce, so generalisations are difficult (although see section on Plantations below).
3.2 Deadwood Availability and Hollow Tree Density
Until the late twentieth century, in many parts of Europe and North America, deadwood in managed forests was removed due to concerns over forest health. While this is still common practice in some areas, the key role played by dead and decaying wood in the functioning and productivity of forest ecosystems, and its importance for biodiversity, has gained increasing recognition over the past 20 years (Humphrey 2005). In Australia, deadwood removal has been confined to plantations, though recognition of the importance of specifically retaining old trees with hollows in managed forests originated in the 1980s. A preference for roosts in dead and dying trees has been noted for Barbastella and Nyctalus species in Europe (Russo et al. 2004; Ruczyński and Bogdanowicz 2008; Hillen et al. 2010), and high densities of dead trees appear to be strongly correlated with the presence of roosts of bark and cavity-roosting bats in forested ecosystems across North America (Mattson et al. 1996; Sasse and Pekins 1996; Rabe et al. 1998; Waldien et al. 2000; Cryan et al. 2001; Bernardos et al. 2004; Broders and Forbes 2004; Miles et al. 2006; Perry and Thill 2007b; Arnett and Hayes 2009).
The importance of high roost density has also been reported in Australia. In dry Jarrah forest of Western Australia, both Gould’s long-eared bat, Nyctophilus gouldi, and the southern forest bat, Vespadelus regulus, preferred roosting in older forest that contained a much higher density of trees with hollows (16–32 trees ha−1) than shelterwood creation and gap release sites (8–12 trees ha−1) (Webala et al. 2010). These mature forest hollow tree densities are comparable to average densities of live and dead hollow trees in roost areas used by Gould’s wattled bat, Chalinolobus gouldii, (17 ha−1) and the lesser long-eared bat, N. geoffroyi, (18 ha−1) in a fragmented landscape in south-eastern Australia (Lumsden et al. 2002). Greater densities of hollow trees likely facilitate roost switching in bark and cavity-roosting bats or fission–fusion behaviours (Kerth and König 1999; Willis and Brigham 2004). These behaviours lead to complex patterns of use and movement among available roost trees by colonies of forest bats. The variation in numbers of roosts between core and peripheral areas of roost networks is further influenced by the density and spatial distribution of available roost trees, as demonstrated for Rafinesque’s big-eared bat, Corynorhinus rafinesquii, in south-eastern bottomland hardwood forests of North America (Johnson et al. 2012b). Roost networks of northern long-eared bat, Myotis septentrionalis, in actively managed forests were scale-free and connected to a single central-node roost tree (Johnson et al. 2012a). A similar pattern was observed for the open-space foraging white-striped free-tail bat, Tadarida australis, in south-east Queensland (Rhodes et al. 2006). Given these patterns, we postulate that implementation of silvicultural systems, which promote retention of higher densities of dead and old living trees across forested ecosystems, should benefit bark- and cavity-roosting bats and facilitate ‘natural patterns’ in colony behaviours, social interactions, and the use of roost networks.
3.3 Understory Vegetation
The extent and composition of understory vegetation in forests strongly influences insect prey availability, the ability of bats to access the forest interior, and the microclimates available and is also likely to affect risk of predation. The degree to which understory cover affects the use of forests by bats depends greatly on their wing morphology and foraging behaviour, with some bats benefitting from a more open forest with little in the way of cover, while other species rely heavily on a well-developed dense understory (e.g. Hill and Greenaway 2008; Müller et al. 2012). Vegetation structure revealed by LiDAR in Germany indicated that while high levels of understory cover were preferred by edge-space and gleaning species, open-space foragers were more associated with relatively open forest stands (Jung et al. 2012). Foraging intensity also varies with canopy height, with the activity of open-space foragers highest above the canopy (Kalcounis et al. 1999; Müller et al. 2013), although few studies have surveyed bats at those heights. Similarly, in forest fragments in Scotland (UK), high activity levels of edge-space species, e.g. Pipistrellus spp., are related to low tree densities and an open understory, while closed-space gleaning species, e.g. Natterer’s bat, Myotis nattereri, showed the opposite trend. These studies are supported by numerous species-specific studies. For example, roosts of Bechstein’s bat, Myotis bechsteinii, and the barbastelle bat, B. barbastellus, are strongly associated with areas of thick understory (Greenaway and Hill 2004), and core foraging areas for brown long-eared bat, Plecotus auritus, a closed-space species, were associated with more cover and a well-developed understory layer more than peripheral areas (Murphy et al. 2012). An Australian study of vertical stratification (excluding above the canopy) in spotted gum forest also found the understorey to support the greatest insect abundance, although bat activity was up to 11 times greater in the canopy where there was less clutter and presumably insects were more accessible (Adams et al. 2009). There was no evidence that any one ensemble or ensemble species foraged exclusively at a particular height, although the open-space ensemble was most activity in the canopy.
3.4 Slope and Aspect
Slope and aspect influence roost selection in forest bats by creating variation in the amount and extent of solar heating at roosting sites due to differences in shading effects and the length of the day that roosts are in direct sunlight. Studies have demonstrated the importance of both slope position and reproductive stage in roost selection. For example, long-legged myotis, Myotis volans, in the north-western USA switch between riparian bottoms and upper-slope positions during pregnancy, but select roosts in upper-slope positions during lactation, where they would be exposed to greater solar radiation (Baker and Lacki 2006). Studies of bats in south-eastern forests of North America have also observed preferences for roosting in upper-slope positions by foliage-roosting eastern red bat, Lasiurus borealis, and bark- and cavity-roosting bats (Myotis and Eptesicus) (Hutchinson and Lacki 2000; Lacki and Schwierjohann 2001; Perry et al. 2008), suggesting that higher slopes are important for roost selection in some forest bat species in both eastern and western parts of North America and should be accounted for in forest planning. Use of lower slope positions and riparian corridors for roosting is common in several bats in eastern and south-eastern forests, however, including bark- and cavity-roosting (Watrous et al. 2006; Perry and Thill 2008; Fleming et al. 2013) and foliage-roosting species (Perry et al. 2007a; Hein et al. 2008b; O’Keefe et al. 2009). Roosting on lower slopes was also found in a subtropical Australian forest, where lactating eastern forest bats, V. pumilus, roost in hollow trees in riparian zones during early summer, but shift up-slope during autumn when bats begin to mate (Law and Anderson 2000). Riparian zones may provide more buffered conditions for maternity roosts in warm, subtropical locations.
In the northern hemisphere, selection of south-eastern-facing (Willis and Brigham 2005), south-facing (Klug et al. 2012), and eastern-facing (Perry and Thill 2007a) sides of tree canopies by hoary bats, Lasiurus cinereus, is associated with positive energy savings and is hypothesised to facilitate rapid growth of young (Klug et al. 2012). Eastern red bat, L. borealis, another foliage-roosting species, was observed using the south aspect of tree canopies that were also located in south-facing slope positions (Mormann and Robbins 2007). Collectively, these behaviours suggest consideration be given to creating and maintaining edge habitats for foliage-roosting bats at the landscape scale, especially along south-facing slopes in the northern hemisphere in areas with sufficient topographic relief.
3.5 Forest Edge
Loss and fragmentation of forest habitat are accompanied by an increase in the ratio of forest edge to interior forest, and the response of bats to this can vary among species. Roosting ecology and edge-affinity have been identified as good predictors of the sensitivity of individual bat species to habitat fragmentation; ‘forest interior’ species (often tree-roosting bats) are negatively affected by fragmentation, as opposed to species which show affinity for forest edges (Meyer et al. 2008).
Edge habitats can influence roosting behaviour in bark- and cavity-roosting Myotis species differently. Indiana bat, M. sodalis, and northern long-eared bat, M. septentrionalis, two species with overlapping distributions in North America and similar preferences for roosting in dead trees (Foster and Kurta 1999; Lacki et al. 2009), choose roosts differently in the same forested landscapes. M. sodalis prefers roosts in edge habitats with low vegetative clutter and higher solar exposure of roost trees and M. septentrionalis selects roosts in shaded environments within intact forests (Carter and Feldhamer 2005). Russo et al. (2007) found that barbastelle bat, B. barbastellus, emerged later from tree roosts in more open forests, probably as a result of increased predation risks, and suggested that it was important to ensure canopy heterogeneity to provide a range of roosting conditions. Edge effects also influence foraging behaviour in forest bats although results from studies comparing bat activity at the edge compared to forest interior show contrasting results; all five species spanning the open/edge-space/closed-space spectra that were assessed in forests in Canada showed higher activity at the forest edge than in the interior (Jantzen and Fenton 2013). Bat activity was also high along coupe edges 5–8 years after clear fell in Tasmania (Law and Law 2011), partly because bats avoided the large harvested gaps in these coupes. In contrast, of three species surveyed within forest fragments on farmland in the UK, one edge-space species showed similar levels of activity at edge verses interior while the other two species (one edge-space and one closed-space) showed higher levels of activity within the forest interior (Fuentes-Montemayor et al. 2013). In Australia, harvested forests are often interspersed with old logging trails and fire trails, providing ‘edge habitats’ that facilitate the use of dense forest regenerating after harvest by bats that possess a range of traits (Crome and Richards 1988; Law and Chidel 2002; Webala et al. 2011). Activity on trails in regrowth forest is as high as it is in mature forest. Most importantly, foraging activity is typically much higher on forest trails than within the forest remote from trails or along narrow riparian zones (Law and Chidel 2002; Lloyd et al. 2006; Webala et al. 2011). Use of trails as linear edges in regenerating forest has also been reported in North America (Menzel et al. 2002). These observations highlight the importance of edge habitats to many bat species within each ensemble, in all the regions covered in this chapter.
4 Bat Responses to Silvicultural Treatments
Silviculture involves a diverse range of techniques to manipulate growth conditions, extract resources, and facilitate regeneration within forests. These influence the composition and density of tree species present, the extent and composition of the understorey vegetation and ultimately the resources available for bats. Here, we focus on the techniques for which there is at least some information on the response of bats to (1) different logging strategies, (2) thinning regimes, and (3) the use of harvest exclusion areas. We also examine the use of timber plantations by bats which, in some regions, is the focus of silvicultural activities. There is very little information on the effects of other techniques such as coppice and the use of chemical applications (e.g. herbicides to clear vegetation), and we highlight important knowledge gaps in the concluding section.
4.1 Logging
Historically, the strategy for logging in forest managed for timber extraction was to remove all trees within an area (clearcuts) as this is considered the most economically profitable method. In production State Forests in Australia, selective harvesting was most common before World War II, but it was subsequently recognised that this adversely affected the regeneration and growth of many of the fastest growing, commercial species, which subsequently led to increased intensity of harvests. Recent concern over the environmental (including biodiversity loss and soil erosion) and visual impacts, however, has led to increased use of more selective forms of logging including variable retention and group selection techniques, which are reviewed here.
A review of published data sets on response of forest bats to silvicultural logging indicates that there are major gaps in our understanding of relationships of bats with timber harvesting practices (Table 5.1). In particular, there is a notable lack of long-term, longitudinal studies that track changes in bat assemblages and their forest habitat over time. Studies on bats in even-aged systems have largely focused on responses to clearcuts with limited exploration of two-age systems such as seed tree, shelterwood, or deferment harvests (Owen et al. 2004; Titchenell et al. 2011). Clearcut harvests have been used with less frequency, especially on public lands, for some time now (USDA and USDI 1994), though they still persist in cool temperate forests, such as those of Tasmania (Law 1996), and some European countries. Patterns in bat responses to clearcuts are still helpful, however, in understanding the potential effects on bats of future directions in forest management based on even-aged systems. Bat responses to uneven-aged systems, such as small cutblocks, patch cuts, or group selection harvests, have received greater attention and have been evaluated across multiple bat species and forest types, so inferences can be drawn on the efficacy of these silvicultural systems for bats. In North America, more studies have evaluated bat response to thinning than any other silvicultural treatment, with thinning often applied in combination with other treatments on the same landscape (Erickson and West 1996; Patriquin and Barclay 2003; Loeb and Waldrop 2008; Perry et al. 2008). Studies of treatment combinations are important as future directions in the management of forests in North America are emphasising multi-treatment prescriptions (Aubry et al. 2009; Harrod et al. 2009; Hessburg et al. 2010), to increase structural habitat complexity, both vertically and horizontally, while reducing the impact of insect infestations and the threats of wildfire and global climate change (Boerner et al. 2008; Parks and Bernier 2010; Duerr and Mistretta 2013). Some forest management strategies specifically target bats, though often bats are catered for under broad forest prescriptions that aim to accommodate the needs of a range of forest-dependent species in an area (Law 2004).
There is a surprising lack of European studies on the effects of any logging strategy on bats and the only study found for this review which directly related to this issue was one on the effects of salvage logging (see Sect. 4.1.4). This is especially concerning given the strict protection afforded to all bat species and particularly bat roosts under the EU Habitat Directive; this prohibits deliberate disturbance of all bats during any stage of their life cycle as well as the destruction of breeding sites or resting areas. As such, the timing of forest harvesting needs to consider whether bats may be roosting in targeted areas (e.g. Forestry Commission 2005). There are no such restrictions in Australia; though for New Zealand pine plantations, Borkin et al. (2011) recommends that harvests should be planned when bats are not heavily pregnant nor have non-volant dependents. In eastern North America, logging is currently restricted from 15 October through 31 March across the distribution of the endangered Indiana bat, Myotis sodalis, as this bat uses live and dead trees as maternity sites during the growing season (USFWS 2009). Restrictions are further constrained to a start date of 15 November within 16 km of known hibernacula of the species (USFWS 2009). The implications of white-nose syndrome and the extensive mortalities of cave-hibernating bats in North America (USFWS 2012) are likely to add species of forest bats to the threatened and endangered species list in the USA, leading to further restrictions on logging. Missing in all of the dialogue, however, is any direct link of impact, or mortality of bats, during logging operations and studies of these potential impacts are needed (but see Borkin et al. 2011).
4.1.1 Clearcut and Deferment Harvests
Response of forest bats to clearcut harvests has been mixed across forest types and species of bats (Table 5.1). For example, three studies each in different locations within the Pacific coast forest type found no response to clearcuts by little brown bats, Myotis lucifugus, in British Columbia (Lunde and Harestad 1986), a decrease in overall bat activity over clearcuts in south-eastern Alaskan rainforests (Parker et al. 1996), and an increase in activity of big brown bats, Eptesicus fuscus, silver-haired bats, Lasionycteris noctivagans, and Townsend’s big-eared bats, Corynorhinus townsendii, in clearcuts in western Washington (Erickson and West 1996). Patterns in bat activity recorded in and around clearcut harvests are influenced by three factors: the number of years post-harvest when data were collected, the size and shape of cutblocks studied, and the assemblage of bat species present in the area. When reported, the age of clearcut stands in North America evaluated post-harvest ranged from 1.5 to 17 years. This range in age is wide and likely spans considerable variation in above-ground habitat structure due to differences in the amounts of regeneration present; thus, a varied response by bats across studies and geographic locations should be expected. In montane eucalypt forests of south-eastern Australia, bat activity peaked in 165-year-old wildfire regrowth rather than in younger regrowth from clear-felling operations (Brown et al. 1997). Unfortunately, the size and shape of clearcuts studied are rarely reported so an evaluation of the effects of cut size and shape on bat activity cannot be made.
Focusing on traits is likely to provide more insights into the response of bats to the large gaps created by clearcut harvests. In North America, two trends are evident. First, the creation of less obstructed flight space over clearcut stands generally leads to increased levels of activity of edge/open-space bats that possess moderate to high aspect ratios and often higher wing loadings (Lacki et al. 2007). This mix of bats includes the foliage-roosting Lasiurus species, along with others (Lasionycteris and Eptesicus) (Table 5.1). The length of years post-harvest at which this increase in bat activity is sustained is less clear and likely is affected by tree species composition and the speed at which regeneration proceeds in harvested stands at a particular geographic location. Second, the response to clearcut harvests between Myotis species varies both within and among species (Patriquin and Barclay 2003), with some increase in activity associated with linear edge habitats at the periphery of cuts but reduced activity in the centre of harvested stands, except where residual patches are left behind (Hogberg et al. 2002). As our ability to distinguish among Myotis species increases with technological advances in acoustic detectors and software packages (Britzke et al. 2011), resolution among the full suite of Myotis bats in North America should become possible allowing for a more in-depth and complete evaluation of bat response to edge effects in actively managed forests.
Data on bat responses to even-aged systems other than clearcuts are sorely lacking. A study of bat activity in deferment harvests found high levels of activity of silver-haired bats, L. noctivagans, in stands with 6–10 m2/ha of basal area remaining (Owen et al. 2004), and the only study examining bat activity in shelterwood harvests (30 to 50 % reductions in basal area) observed higher levels of activity in three species of bats that have wing morphologies and echolocation call structures possessed by edge/open-space bats (Titchenell et al. 2011). Patterns of habitat use by radio-tagged northern long-eared bats, M. septentrionalis, a closed-space bat, showed this species spent limited time in deferment harvest stands, especially harvested sites with more open canopies and less cluttered foraging space (Owen et al. 2003).
For roosting bats, gap release and shelterwood systems retain tall and large diameter hollow-bearing trees within stands possessing less clutter than surrounding forest regenerating after harvest and these offer potential roosts for bats. However, in Western Australia, southern forest bat, V. regulus, avoided locating roosts in shelterwood treatments when older forest was available nearby (Webala et al. 2010). In general, remnant trees in these silvicultural treatments, including retained ‘habitat trees’, were not preferred as roost sites by V. regulus, though a second species (N. gouldi) frequently used such trees. One possible reason for avoiding using ‘habitat trees’ as roosts was the relatively low density of hollow roosts (see 3.2 Deadwood availability and hollow tree density).
4.1.2 Variable Retention Harvests
Variable retention has recently been proposed as an alternative to standard clearcuts, whereby old-growth elements are retained within the clearcut coupe (Baker and Read 2011). Variable retention increases the availability of edges, for example, around retained patches (aggregates ) of undisturbed forest within the clear-fell coupe and along coupe boundaries as well as increasing the area of open space. Open- and edge-space ensembles would be expected to benefit from this treatment. The 200-ha Silvicultural Systems Trial, in Tasmania, provides one of the main experimental sites in Australia for investigating responses to variable retentions. Bat activity was similar in control coupes of 45- to 60-m-tall old-growth Messmate Stringybark, Eucalyptus obliqua, forest, compared to variable retention coupes 5–8 years after logging (Law and Law 2011). Activity was lower above the dense young regeneration of clear-fell-burn-sow (no retention) coupes and marginally lower for dispersed tree retention coupes. This suggests that the retention of old-growth elements as aggregates or patches moderates the unsuitable young regrowth zone for total bat activity, while retention of dispersed individual trees is less effective. Surprisingly, bat activity was low at the retained aggregates themselves, both in their centre and along the edge, and it is not known to what extent bats roost in these locations. Overall the results are consistent with conceptual models (Fig. 5.1), whereby activity is predicted to be higher in areas of medium clutter levels and where hollow abundance is high. Individual bat taxa responded to treatments consistent with predictions from ecomorphology . Closed-space bats were less active in clearcuts than unharvested forest, large edge-space bats were more active in clearcuts (especially along edges), and smaller edge-space bats were less influenced by patch type and location within coupes; consistent with other studies of forest clearcuts from North America (Grindal and Brigham 1999; Menzel et al. 2002; Patriquin and Barclay 2003).
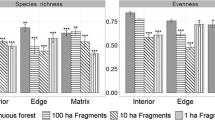
The age of regenerating forest is likely to be an important influence on how bats respond to variable retention. An unreplicated, operational scale (100-ha forest blocks) experiment established in 1984 in the temperate forests of southern New South Wales (Waratah Creek) (Kavanagh and Webb 1998) was sampled acoustically for bats after 18 years of regrowth. Treatments retained different amounts of tree canopy within four different forest blocks comprising 100 % (control), 50 % (0.5 ha patches in a chessboard pattern), 25, or 10 % tree canopy retention. Control sites supported 2–4 times more activity than logged sites, with 10 % retention supporting the lowest activity level with just 50 bat passes per night of sampling (Fig. 5.5; B. Law, unpubl. data). Thus, bat activity remained low even 18 years after logging and the amount of canopy retained within a block had little impact on activity, except for the block with the most intensive logging which supported the lowest activity level. As expected, the activity of closed-space bats was similar, though low, between the control and treatments, after 18 years. Activity of edge-space bats was three times lower in logged stands, suggesting a loss of edges and spaces between trees, especially in the treatment where logging was most intense. Logging treatments had little effect on open-space bats that forage above the canopy, except that activity was lower where logging intensity was greatest.
Total bat activity (762 passes, 10 taxa) recorded 18 years after logging in an unreplicated, variable intensity logging experiment in New South Wales, Australia. Data are mean number of passes per night for two Anabat detectors deployed per forest block (~100 ha) over two entire nights of recording and exclude activity on trails (B. Law, unpubl. data). Different bat ensembles are open-space, edge-space, closed-space, and unknown
4.1.3 Group Selection Harvests
Changes in ensemble activity with group selection harvest are likely to depend on gap size, with an increase in edge-space activity if gaps are small and open-space activity if gaps are large. Immediately after harvest, closed-space bats are expected to decline, but we predict subsequent recovery if the retention of roost trees is catered for. All studies examining bat responses in North America to group selection harvests, canopy gaps, or small cutblocks consistently reported increases in activity of bats, primarily open/edge-space species, with the opening up of forest canopies, regardless of forest type or assemblage of bats present (Table 5.1). The one exception was a decline in activity of Myotis bats in canopy gaps in forests of the central Appalachian Mountains, with this drop off in use inversely correlated with increasing diameter of canopy gaps (Ford et al. 2005). In this study, the maximum gap diameter examined was 33.5 m in width, with the decline in activity with increasing gap size largely attributable to response of closed-space Myotis species. Studies in oak–pine forests in Arkansas have demonstrated the use of dead and live trees along gap edges for roosting by several bat species (Perry and Thill 2007b; Perry et al. 2008), demonstrating the importance of maintaining canopy gaps in managed forests. The almost universal response by bats of increased activity with canopy gap formation means this silvicultural treatment holds much promise for management of foraging habitat of bats in the short-term. Use of gaps by forest bats following a decade or more of successional change is likely to be different, however, with overall declines in activity plausible as open/edge-space species disappear or decline in abundance with increasing gap clutter. Such temporal changes need to be identified along with the optimal gap size(s) and the density of gaps required by different species of bats to permit commercially viable, sustained yield harvests while fostering high levels of bat activity and provision of roosting habitat in managed forests.
In contrast to many North American studies that have been undertaken in gaps soon after harvesting, in Australia, most bat research has focused on the use of older regrowth regenerating from group selection harvest, particularly characterising bat species by their traits in relation to the use of these dense stands. There is a general pattern of forest clutter increasing over time after group selection harvest so that old regrowth (>30 years) has significant higher clutter levels than young or older forest, which constrains use by bats to closed-space species with a low wing aspect ratio (Law and Chidel 2002; Webala et al. 2011). Less manoeuvrable edge-space species with a high wing aspect ratio tend to be scarce in regrowth forest (except on flyways provided by tracks and creeks), although their activity is greater in the subcanopy and canopy than understorey (Adams et al. 2009). Vegetation is more cluttered in regrowth at these upper heights (closer stems and less vertical space in the subcanopy), and this leads to less bat activity in such situations (Adams et al. 2009). It is not known whether open-space and low-frequency edge-space species are active above the canopy of these young forests, although this was confirmed by Müller et al. (2013) for mature forests in Europe.
4.1.4 Salvage Logging
Salvage logging involves the removal of dead wood after a natural disturbance (e.g. windthrow, forest fires, and insect outbreaks) and has been employed even in protected forests, provoking some controversy. To our knowledge, no research has examined the implications for roost availability of this practice, although removal of standing dead wood will inevitably reduce the abundance and diversity of roosts and would have a considerable impact when carried-out over large scales (Lindenmayer and Noss 2006). We found two studies which investigated changes in bat activity following salvage operations. In Germany, closed-space species reduced their activity in both types of forest clearing (bark beetle and logging), while the activity of open-space species slightly increased, and edge-adapted species showed a mixed response (Mehr et al. 2012). These results are similar to a study in Oregon where the highest bat activity was in the more intensely logged sites (Hayes 2009).
4.2 Recovery Times After Timber Harvest
Long-term studies are largely missing from assessments of the response of bats to silvicultural methods. A typical approach uses chrono-sequences or snapshots of comparisons between different silvicultural methods or logging histories and makes the assumption that the matching of treatments is equal and evenly distributed across the same environmental niche and landscape context. Most importantly, a one-year snapshot may not be representative of temporal variation and dynamism over a longer period (Recher et al. 1983; Maron et al. 2005); thus, conservation plans developed from snap-shots can have limitations. Long-term studies are ideal for tracking changes to vegetation structure as forests regenerate after harvesting and how different ensembles of bats respond to these dynamics.
One longitudinal study in Australian eucalypt forests, initiated in 1998, has been investigating alternate-coupe-integrated harvesting for woodchips and saw-logs, and although currently unpublished, a summary is presented here (B. Law and M. Chidel, unpubl. data). Alternate-coupe harvesting divides management units (e.g. 200-ha areas) into small (~15 ha) coupes that are alternately harvested in a chessboard fashion, every 20 years. In 1998, bat activity was recorded after 22 years of regrowth from the first cycle of logged coupes (Law and Chidel 2001). Bat activity in the cluttered regrowth was about half that of adjacent, more open unlogged coupes. This effect was most notable for less manoeuvrable, open- and edge-space vespertilionids that were more active in unlogged coupes.
The site was then sampled at intervals over 13 years following the second round of alternate-coupe logging (B. Law and M. Chidel, unpubl. data; Fig. 5.6). During this period, total bat activity remained low in old regrowth coupes (22 years old in 1998). Activity in unlogged controls remained similar to the initial samples taken prior to second round harvesting. Within the recently logged coupes, activity peaked soon after logging in the large gaps, but it quickly declined and remained at low levels (similar to that found in old regrowth coupes) once young regenerating eucalypts established within eight years of logging. In terms of clutter and total bat activity, these results are only partly consistent with the conceptual models of Hayes and Loeb (2007). The model predicts low bat activity when clutter is very low, yet this was not the case in this study, possibly because gaps were patchy within the 15-ha coupes due to the requirement for retention of 5 habitat trees per ha plus equivalent numbers of recruits, indicating that gap size or scale is likely to be an important issue influencing activity. High activity at intermediate clutter levels (unlogged coupes) and low activity at high clutter levels (old regrowth coupes) are consistent with the model. The response of individual species and ensembles are yet to be analysed for this study.
Changes in total bat activity over 14 years in an alternate-coupe logging system in southern Australia (B. Law and M. Chidel, unpubl. data). The dashed vertical line indicates second round logging of the alternate unlogged coupes in 1999, which took place 23 years after the first round of logging of adjacent coupes in 1976. All but two unlogged coupes were harvested in 1999 and are thereafter referred to as recently logged coupes. Bat activity is a log transformation of the number of passes per night (±95 % confidence limits) after adjusting with mean nightly temperature as a covariate
The lack of recovery after 36 years in old regrowth coupes is consistent with a number of other studies where low activity persisted for more than 30 years after disturbance (Brown et al. 1997; Adams et al. 2009; Webala et al. 2011), but differs from selective harvesting of wet sclerophyll forest in subtropical Queensland where recovery of bat activity was apparent in a site logged 33 years previously (de Oliveira et al. 1999). It is important to note that none of these studies consider activity levels on tracks, riparian zones, or other areas of retention that potentially could ameliorate the effects of clutter from dense regrowth and loss of tree hollows.
4.3 Thinning Young Forests
The goal of thinning is to improve the quality and growth of the remaining trees (especially diameter) by reducing the density of trees in a stand. Reducing tree density will decrease canopy cover, at least initially, with increased light levels reaching the forest floor and thus influencing understory cover. Adams and Law (2011) reviewed the literature on thinning and bats and proposed hypotheses for testing that included: (1) activity of edge- and open-space species will increase from pretreatment levels where thinning reduces stem separation to 7 m (~200 stems per ha) but will remain at low levels where average stem separation is less than 3 m (~1100 stems per ha); (2) highly cluttered forests will have low bat activity away from flyways, regardless of the number of potential roosting sites and the abundance of insects, while bat activity in open forests will be highest where roost availability and insect abundance are high.
Consistent with the hypotheses, bat responses to silvicultural thinning have been examined across several forest types in North America with increases in bat activity associated with thinning in Pacific coast (Erickson and West 1996; Humes et al. 1999) and southern oak–pine (Loeb and Waldrop 2008) forests, but not in northern red pine, Pinus resinosa, plantations (Tibbels and Kurta 2003) or northern coniferous forests (Patriquin and Barclay 2003). An explanation for these differences is not readily clear, as the extent of thinning is not always reported in metrics that can be compared among study sites, and the suite of bat species present varies among locations. Further, data for bat activity within the Myotis genus could not be resolved to the species level with technologies used, preventing an evaluation of responses by ensemble. Patterns in roost selection of Lasiurus species in southern oak–pine forests indicate that thinned stands are frequently selected by these bats for roosting. Thus, as with clearcut harvests and larger-sized canopy gaps, stands thinned to basal areas <14 m2/ha appear to be well suited to less manoeuvrable edge-space Lasiurus species by providing suitable roosting and foraging habitats (Perry and Thill 2007a; Perry et al. 2007a, 2008).
The response of bats to forest thinning has received little attention in Australia. A preliminary study found high variability in activity for all bats and ensembles between thinned and unthinned eucalypt stands and among vegetation layers within the forest (Adams and Law 2011). Unexpectedly, thinned regrowth had a higher percentage cover for the shrub layer, and the vertical gap between canopy and understory trees was halved, which represented an increase in clutter in the zone where bats frequently fly and this could have undermined any benefit of wider stem spacings. However, the variability in bat activity within the thinned/control treatments was too high to unequivocally state that thinning had no effect.
While thinning is a commonly employed silvicultural technique across Europe, there has been no study of its effects on bat activity, occurrence, or species richness. There are, however, a few studies which have looked at effects of tree density on bats, thereby providing indirect evidence on likely effects of thinning. For example, in one study, where tree density varied between 180 and 2500 stems per ha in mixed deciduous/coniferous fragments within agricultural landscapes in Scotland (UK), activity of the soprano pipistrelle, Pipistrellus pygmaeus (an edge-space forager), decreased with increased tree density. In contrast, the abundance and activity of Myotis spp., and the abundance of Diptera, both increased with tree density (Fuentes-Montemayor et al. 2013). This mirrors findings by Müller et al. (2012) where the activity of closed-space foragers and prey abundance increased at higher vegetation densities, while the activity of open-space foragers, and to a lesser extent, edge-space foragers declined.
4.4 Harvest Exclusion Areas
Given the low levels of bat activity observed in young regenerating forest after logging, mitigations are needed to ameliorate the effect of high clutter levels and lower numbers of tree hollows. Edge habitat, such as tracks and clearcut boundaries, is extensively used by a range of bat species (Sect. 3.5). In Australia, harvest exclusion areas that support naturally open, undisturbed forest constitute a much greater proportion of the forest landscape compared to forest tracks and are therefore expected to be more important at ameliorating logging impacts on bats given that they also provide roosts in the hollows of old trees. Provided attention is paid to the size and location of harvest exclusion areas these can play a vital role in landscape connectivity, acting as corridors across forested landscapes, permitting bats to reach otherwise isolated blocks of preferred habitat within landscapes where fragmentation has altered the matrix and created an abundance of suboptimal habitat blocks. As the extent of habitat fragmentation increases, so does the importance of corridors on the landscape (Duchamp et al. 2007). Indiana bats, M. sodalis, preferred to fly along wooded corridors and avoided open fields in Michigan, even though commuting distances increased by more than 50 % (Murray and Kurta 2004), with similar results for Pipistrellus spp. in the UK (Downs and Racey 2006). Activity of bats in heavily fragmented, pine plantations in South Carolina demonstrated more use by bats of edges along corridors than habitats within the corridor interior or nearby stands of timber (Hein et al. 2009a), with bat activity directly correlated with the height of the corridor overstorey.
Riparian corridors in timber production forests are often excluded from harvesting in order to ameliorate impacts of harvesting on water quality as well as providing unharvested productive habitat for biodiversity. Riparian corridors are important areas of bat foraging activity (Hayes and Adam 1996; Zimmerman and Glanz 2000; Brigham 2007), with male and female bats segregating themselves along corridor reaches in upland landscapes, with males more abundant at higher elevations (Grindal et al. 1999; Senior et al. 2005). Activity of bats along riparian corridors appears to be scale-dependent, with vegetation architecture, i.e. shrub and tree cover, influencing the use of foraging space by bats at the local, or finest spatial, scale more than landscape habitat measures or abundance of insect prey (Ober and Hayes 2008). Abundance of Lepidoptera was high in riparian corridors in Arkansas prompting the authors to hypothesise that Ozark big-eared bat, Corynorhinus townsendii ingens, a moth strategist (Dodd and Lacki 2007), feeds extensively in and around riparian corridors in the Ozark Mountains (Dodd et al. 2008). Use of best management practices along streamside management zones for sustaining healthy, riparian ecosystems is a well-established forest management practice in many regions of North America (Stringer and Perkins 2001; Lee et al. 2004). Regardless, data on how these practices influence habitat use by forest bats in riparian areas remain limited, with experimental studies sorely needed on the effects of habitat quality within corridors (stand age and composition) and corridor dimensions (size and width) on roosting and foraging ecology of bats. One study in Australia demonstrated that bat activity, foraging rates, and species richness in riparian corridors within selectively harvested eucalypt forest was maintained at levels similar to riparian areas in mature forest (Lloyd et al. 2006). Higher activity was recorded on larger rather than smaller order streams, a pattern also not affected by harvesting history. Such results highlight the benefits of buffers , with riparian areas effectively providing habitat for foraging and commuting bats in selectively logged forests where clutter levels are likely to be high.
Mitigating the loss of roosting habitat in hollow-bearing trees is arguably even more important than maintaining suitable foraging habitat. Forested corridors are critical habitat elements for North American foliage-roosting bats by providing both roosting and foraging opportunities. Male Seminole bats, Lasiurus seminolus, in south-eastern loblolly pine, P. taeda, plantations chose roost trees in forested corridors within harvest exclusion zones over 60 % of the time, even though corridors represented only 11 % of the landscape area (Hein et al. 2008a). Corridors were 100 to 200 m in width and comprised largely of older-aged forests in riparian and upland slope positions. Use of forested corridors for roosting has been observed in other foliage-roosting species in south-eastern forests, with tri-coloured bats, Perimyotis subflavus, selecting riparian corridors (O’Keefe et al. 2009), male evening bats, Nycticeius humeralis, choosing upland corridors of mature forest (Hein et al. 2009b), and eastern red bats, L. borealis, roosting in the vicinity of gated roads (O’Keefe et al. 2009). Greenbelts in riparian corridors, or unharvested inclusions of mature mixed-pine hardwoods ≥50 years in age, were important roosting habitats for these same species in southern oak–pine forests of Arkansas (Perry et al. 2007b; Perry and Thill 2008).
Harvest exclusion areas, especially those surrounding streams, are commonly used as roosting habitat by many tree hollow roosting Australian bats such as Gould’s long-eared bat, N. gouldi, eastern forest bat, V. pumilus, and southern forest bat, V. regulus (Lunney et al. 1988; Law and Anderson 2000; Webala et al. 2010). A range of factors will influence the pattern of roosting close to creek-lines, but a large pool of older and mature trees in a variety of decay classes is likely to be important. Riparian areas often support a different vegetation type, with rainforest being particularly common in Australia. The specialist golden-tipped bat, Kerivoula papuensis, preferentially roosts in the suspended nests of small birds within riparian rainforest and such areas are excluded from harvesting (Schulz 2000; Law and Chidel 2004).
Jarrah forest in Western Australia offers one example of providing pools of mature trees using zoning. Since 2004, Fauna Habitat Zones (i.e. areas of mature forest >200 ha set 2–4 km apart within areas available for logging) have been retained for species, including bats, that rely on blocks of forest supporting mature forest attributes or characteristics (Webala et al. 2010). In some forest blocks, approximately 54 % of the total area (11,740 ha) is currently reserved from logging as conservation reserves, informal reserves (riparian buffers, diverse ecotype zones, road reserves), old-growth forest, and fauna habitat zones. Of these, about 39 % are permanently reserved, including riparian buffers, from logging in the future. Testing the effectiveness of this level of retention remains a priority for forest bat research. Collectively, these findings indicate that forested corridors are important habitat elements for roosting bats in forests across the globe.
4.5 Plantations
There is no internationally agreed definition of forest plantation and many very old forests we may think of as natural have been planted. However, for the purposes of this review, the term plantation is used to mean forests planted primarily for timber extraction using intensive management techniques. Timber plantations are perhaps the most extreme form of silviculture as they require replanting of typically exotic trees, with site and soil preparation required over large scales. Seedlings are planted at high densities to maximise growth and form of trees, and this has the consequence of producing high levels of clutter as the trees grow. All the silvicultural practices outlined in this section are also applicable to plantation forests. The response of bats has been documented in eucalypt plantations in Australia and pine plantations in New Zealand. As expected, bat activity in young plantations of eucalypts (<10 years) is typically low and considerably less than that found in nearby forest, and, somewhat surprisingly, activity is similar to levels over adjacent cleared farms (Law and Chidel 2006; Law et al. 2011). Bat activity is higher in older eucalypt plantations (~25 years), especially where drought and lack of maintenance leads to tree mortality and the creation of gaps (Law and Chidel 2006). Closed-space species (Nyctophilus) show some association with plantations as do open-space species (Mormopterus ridei), which presumably use the space above plantations together with adjacent open paddocks. Radio-tracked bats avoid roosting in young eucalypt plantations where tree hollows are absent, even though decorticating bark is present (Law et al. 2011).
Despite limitations in habitat quality, plantation forests provide large areas of additional habitat for threatened long-tailed bats, Chalinolobus tuberculatus, in New Zealand (Borkin and Parsons 2011a). Borkin and Parsons (2011b) found these bats roosting in crevices, fissures, and small hollows in the oldest stands of Monterey pine, Pinus radiata, plantations (25–30 years), with females choosing to roost within 150 m of waterways. In these plantations, bats selected home ranges with higher proportions of relatively old stands than available (Borkin and Parsons 2011a). Males selected edges with open unplanted areas within their home ranges, which females avoided, instead selecting older stands for foraging. Borkin et al. (2011) also documented the response to the clear-fell harvest of a pine plantation and found a pattern of declining numbers of roosts used, as well as smaller roosting areas and colony sizes. Over 3 years, 21 % of known roosts were lost with 15 % due to forestry operations and 6 % due to natural tree fall. To mitigate harvest operations, it was suggested that some suitable foraging and roosting areas should be retained within bat home ranges. Borkin et al. (2011) further suggested that priority management for this declining New Zealand bat should focus on plantation areas closest to water and harvests should be planned when bats are not heavily pregnant nor have non-volant dependents.
Pine plantations in the south-eastern USA are actively managed landscapes with extensive amounts of fragmentation and edge development. Nevertheless, these landscapes often support a diverse bat assemblage, in part due to enhanced foraging conditions along edge interfaces and to suitable foraging and roosting habitats along forested-riparian corridors (Miller 2003; Elmore et al. 2004; Hein et al. 2008b, 2009a). Experimental studies have demonstrated that activity of bats is affected by edge habitats, with highest levels of activity occurring along the edge interface regardless of echolocation call structure or wing morphology (Jantzen and Fenton 2013). Tree canopies also serve as edge interfaces in forested environments, with more manoeuvrable, high-frequency bats foraging along canopies and edges more often than less manoeuvrable, low-frequency bats (Pettit and Wilkins 2012). Relationships of age, formation, and structural characteristics of edge habitats with activity of foraging bats are complex, with newly formed, high-contrast edges supporting higher bat activity and stronger depth of edge influence, than older more developed, cantilevered edges which possess less contrast between adjacent habitats (Jantzen and Fenton 2013). Regardless, data indicate that managed forests with an abundance of edge habitat, typical of plantation forests in south-eastern North America, can support a diverse assemblage of forest bat species.
Spruce, pine, and fir species account for the largest share of the forest plantation area in Europe, with Eucalyptus species introduced from Australia common in the south. While eucalypt plantations appear to be avoided by some bats (Di Salvo et al. 2009), positive selection was found for the Mediterranean horseshoe bat, Rhinolophus euryale, in the Basque country (Aihartza et al. 2003). In Spain, R. euryale and Mehely’s horseshoe bat, R. mehelyi, both closed-space foragers, were radio-tracked foraging in eucalypt plantations and dehesa (managed oak savanna) in proportion to, or greater than, their availability (Russo et al. 2005a, b). Numerous acoustic and radio-tracking studies have documented avoidance of bats from non-native coniferous plantations in Europe (e.g. Entwhistle et al. 1996; Walsh and Harris 1996). Perhaps as a consequence of this, the effects of plantation forestry practices on bat populations in Europe have been largely ignored, and surprisingly little is known about the use of timber plantations by bats. However, several long-running artificial ‘bat box’ schemes operated by the UK’s Forestry Commission have indicated that some plantations contain large roosting bat populations (Park et al. 1998). Radio-tracking of Natterer’s bat, Myotis nattereri, a species previously associated primarily with deciduous forests has uncovered the extensive use of areas used for commercial forestry, both for roosting and foraging (Mortimer 2006). This study conducted in a plantation in Scotland found that M. nattereri preferentially foraged within areas of Corsican pine, Pinus nigra var. maritima, and roosted in cavities formed from live double-leadered Corsican pine (Mortimer 2006). Given life-history parameters of the bats studied (survival, population densities) were similar or higher than those described within deciduous forests, and that double-leadered trees are usually targeted for removal by foresters as uneconomic, such findings illustrate the importance of studies in plantation forests. A high percentage of open ground in some planted forests can benefit species that specialise on the predation of ground dwelling prey. Greater mouse-eared bat, Myotis myotis, for example, while often associated with deciduous forests, was found preferentially foraging in mature spruce monocultures with a high percentage of open ground in Germany, and intensively managed orchards and lowland forests with no undergrowth in Switzerland (Arlettaz 1999; Zahn et al. 2004). These studies collectively suggest that it is the forest structure that may be more important than tree species composition in many cases. Therefore, it seems clear that timber plantations have the potential to be of value to bats, but we lack an understanding of how populations of different species are affected by current silviculture practices.
4.6 Prey
The response of bat prey is also a critical issue when evaluating silvicultural treatments. Lepidoptera (moths–a fundamentally important prey group of bats) in temperate zone forests of North America differ little in species richness between stands regenerating after harvest and stands that remain unharvested (Burford et al. 1999; Summerville and Crist 2002; Dodd et al. 2008). Group selection logging of Australian eucalypt forests has found greater insect biomass in old regrowth Jarrah forest (>30 years since logging) than younger forest treatments (Webala et al. 2011) and a similar trend was found in spotted gum forests in eastern Australia (Adams et al. 2009). An additive effect of insect abundance and an index of vegetation openness in the spotted gum forests influenced bat activity, especially edge-space species with medium to high echolocation frequency. High values of insects and openness correlated with high levels of bat activity (Adams et al. 2009). Thus, dense clutter appears to constrain activity of some species even where insect abundance is high. This varies between bat ensembles, however, with closed-space foragers able to take advantage of the higher insect densities often associated with clutter, particularly Diptera, an important taxa for many bats (Müller et al. 2012; Fuentes-Montemayor et al. 2013; see also Sect. 4.3). While the prey base of bats can probably be sustained with application of many silvicultural systems, clearcut stands regenerating as monocultures support reduced levels of moth diversity, indicating that plant species richness is important for providing adequate populations of lepidopteran prey for insectivorous bats in managed forests (Summerville and Crist 2002; Dodd et al. 2012).
5 Multi-spatial Scale Forest Management
Integrating silvicultural systems into managed forested landscapes in ways that promote habitat for forest bats must account for the fact that bats are highly mobile and exhibit considerable variability in the use of habitats both spatially and temporally (Duchamp et al. 2007). Given that resource requirements differ among species and also sex, age, and reproductive classes within species (Perry et al. 2007a; Perry and Thill 2007b; Henderson et al. 2008), designing a forested-landscape matrix with a mosaic of resources that addresses the needs of all bat species in the region will likely require the application of a mix of silvicultural methods, each implemented with different objectives in mind (Guldin et al. 2007). These would include the retention of mature forest habitat at the landscape and stand scale in the form of large reserves, narrow and large strips, streamside reserves, aggregates, and clumps (Gustafsson et al. 2012). Lindenmayer and Franklin (2002) proposed a strategic landscape-scale approach with conservation measures applied at multiple spatial scales for forests. The four main strategies identified for conservation at multiple spatial scales include: (1) establishment of large ecological reserves, (2) application of landscape-level measures in off-reserve areas, (3) application of stand-level measures in off-reserve areas, and (4) monitoring and adaptive management.
There are limited data on bats for setting overall retention thresholds at the landscape scale. Gustafsson et al. (2012) suggested a strict minimum of 5–10 % retention of old-growth forest to achieve a positive ecological response for biodiversity. However, considerably higher levels are often recommended. For example, in Tasmania, 30 % is retained in some state forests (Gustafsson et al. 2012; see also Białowieża Forest in Europe ~20 %, Ruczyński et al. 2010). This retention should be spread across the landscape to facilitate dispersal. A key question is whether there are thresholds for the retention of mature forest that can optimise the trade-off between biodiversity conservation and production.
A recent study on Tasmanian bats, using both radio-tracking and acoustic detectors, assessed the response of bats to multi-spatial scale forest management (Cawthen et al. 2013). At broader scales, maternal bat colonies selected roosts in landscapes with the highest availability of hollow-bearing trees. At more fine-scales, however, maternal colonies did not exhibit strong selection for roost trees in patches with the highest availability of hollow-bearing trees. Instead, other attributes such as hollow type were important. For overall bat activity, the extent to which bats used different types of retained forest patches varied with the composition of the surrounding landscape. Large strips and small patches of wooded habitat were used by bats to a greater extent in landscapes with less mature forest in the surrounding area (<1 km radius). For small patches, this corresponded to landscapes with <22 % mature forest in the surrounding 1 km. No thresholds in bat activity were identified for large patches (370 ha) or small corridors (3 ha). Overall, these results indicate that in the landscapes sampled, activity is low in small retained patches where mature forest is readily available nearby, though these habitat elements do provide roosts and connectivity (and probably foraging habitat) where mature forest is rare or has been lost. Thus, the type, amount, and spatial arrangement of mature forest existing in the landscape need to be considered when retaining forest habitat at finer-spatial scales.
Clearly, the extent to which forest bats respond to changes at the landscape scale remains only partially understood. Studies of bat activity at stand and landscape scales in both north-western and north-eastern forests of North America demonstrated that patterns in habitat use of bats were largely determined by habitat characteristics at the local or stand level and not at landscape scales (Erickson and West 2003; Ford et al. 2006), suggesting that silvicultural systems that create a mosaic of treatments across forested landscapes with local differences in habitat structure will support a higher overall diversity of bat species (Wigley et al. 2007). This approach has been recommended in published studies (Loeb and Waldrop 2008; Perry et al. 2008); however, other sources report both stand and landscape metrics in North America and Europe to be important in selection of activity areas of bats (Loeb and O’Keefe 2006; Yates and Muzika 2006; Fuentes-Montemayor et al. 2013), with tri-coloured bats, P. subflavus, and eastern red bats, L. borealis, most affected by local stand structure, northern long-eared bats, M. septentrionalis, negatively affected by forest edge, and Indiana bats, M. sodalis, positively affected by dead tree density and non-forested land cover. Other studies corroborate that selection of roosting sites in both bark- and cavity-roosting and foliage-roosting bat species is strongly influenced by landscape-scale metrics in both eastern and western forests of North America (Limpert et al. 2007; Perry et al. 2008; Arnett and Hayes 2009; Lacki et al. 2010).
6 Summary and Future Possibilities
This review of the effects of silvicultural systems on forest bats demonstrated that almost all treatments evaluated were compatible with some use by forest bats, depending on the suite of species considered: closed-space species feed in intact forests, but respond to creation of small canopy gaps and less to reduced tree densities and open-edge interfaces; edge-space species exploit edge habitat along tracks, coupe edges, and other linear features such as creeks, but fare poorly within dense regrowth that often dominates soon after harvest; and, open-space foragers benefit temporarily from silvicultural treatments that significantly reduces cluttered air space and provides edge interfaces for roosting. These patterns were largely consistent across three different continents.
To sustain high levels of bat diversity in managed forests at the landscape scale, a balance of needs for these three groupings of bats is desirable and will likely require a mix of silvicultural treatments and exclusion areas staggered across the landscape, regardless of forest type or geographic region. Use of edge habitats, exclusion areas/set-asides, and riparian corridors for roosting and foraging by bats was a consistent theme in the literature reviewed, and these habitat elements need to be considered in forest planning. These landscape features accompany forest fragmentation, however, and it remains unclear to what extent increasing loss of the unharvested forest matrix will lead to declines in population numbers of forest bats. Unfortunately, data on densities of occupied roosts and, thus, potential for landscape-scale population estimates of bats are few (Clement and Castleberry 2013; Fleming et al. 2013). Regardless, population studies could integrate the potential benefits of multiple prescriptions at a scale over which bats themselves sample the landscape. Population studies are likely to provide the ultimate test of the effectiveness of a silvicultural regime, especially when such studies take a long-term perspective. Long-term studies on forest bats are notably lacking in the published literature.
Application of silvicultural treatments in regenerating forests to reduce tree densities and open gaps in the forest canopy shows promise for creating forested landscapes that support diverse and sustainable populations of bats. Forests with reduced tree density and vegetative clutter permit higher levels of light penetration, with this increased exposure hypothesised to enhance the suitability of live and dead trees for roosting by bark- and cavity-roosting bats in temperate climates (Boyles and Aubrey 2006). Further, LiDAR studies demonstrate that reduced clutter in the mid- and understory layers of forests is correlated with higher levels of activity by low-frequency (≤34 kHz) open-space bats (Britzke et al. 2011; Dodd et al. 2013). However, closed-space bat species that glean insects from vegetation and manoeuvre well within clutter benefit from a relatively dense understorey and higher tree densities, which can act as sources of insect prey (Fuentes-Montemayor et al. 2013). Therefore, management that encourages habitat heterogeneity to fulfil the requirements of different species is needed. Bat activity is also vertically stratified, but there is a paucity of information on the effects of high canopy forest structure on bat activity (Adams et al. 2009; Müller et al. 2013), and research to address this gap would be valuable.
The quality and density of old trees in exclusion areas must not be overlooked. Roost abundance stands out as a key variable in our conceptual model (Fig. 5.1). The posited relationship is for increasing bat populations with increasing numbers of roosts, though with a threshold at the upper end of roost abundance rather than at low roost abundance. Densities of hollow trees sufficient to support populations of roosting bats are unknown and remain a major knowledge gap (Law 1996), but will likely be species contingent and based on roost switching behaviours and social dynamics within colonies (Johnson et al. 2013) and the density of other hollow-dependent fauna. Even small colonies of bats can require a large number of roosts over the active season. For example, Russo et al. (2005a, b) estimated that over a period of a month a colony of 12 female barbastelle bats, B. barbastellus, would require approximately 18 different trees for roosting. Although the retention and sustained recruitment of large mature trees at various stages of decay is essential in harvested forests for the future long-term maintenance of bat roosts and other hollow-dependent fauna, this might best be achieved through regular harvest exclusion areas (unharvested buffers, old-growth forest, etc.) that can maintain high local densities of potential roosts. There remains little guidance on how much undisturbed forest should be retained at a landscape scale.
Paradigm shifts in forest management away from even-aged to retention systems (Puettmann et al. 2009) are already in place in Pacific coast forests of North America and Australian eucalypt forests and are being encouraged for use in management of forests globally (Gustafsson et al. 2012; Lindenmayer et al. 2012). These systems allow for maintenance in post-harvest forests of tree species compositions, canopy structures, and ecosystem functions typical of preharvest conditions. We conclude from our review that the use of multi-scale retention systems may be a compatible approach for sustaining habitats of bats in forests. These silvicultural systems are designed to provide spatial variation in retained tree densities and distribution of residual patches of uncut forest, both of which lead to habitat complexity within stands and across landscapes. These systems intentionally mimic natural disturbance regimes and have broad biodiversity benefits across multiple taxa (Long 2009). Retention of old forest patches is likely to be most important where harvest intensity is high, such as in clearcut or heavy selection practices, or where retention of critical habitat components is low. Stand-level (site-scale) retention should be greater where old-growth forest in the surrounding landscape is scarce and where logging practices are more intense. The effectiveness of this multi-scale approach will require testing through monitoring and research tailored for different environments, multiple taxa and silvicultural practices. Monitoring the effectiveness of these strategies is an essential part of adaptive management and a fundamental part of ecological sustainable forestry and the ‘social license to operate’ that is increasingly required by forest certification schemes (Lindenmayer and Franklin 2002).
References
Adams M, Law B (2011) The impact of forest thinning on bat activity: towards improved clutter-based hypotheses. In: Law B, Eby P, Lunney D, Lumsden L (eds) Biology and conservation of australasian bats. Royal Zoological Society of New South Wales, pp. 363–379
Adams MD, Law BS, French KO (2009) Vegetation structure influences the vertical stratification of open- and edge-space aerial-foraging bats in harvested forests. For Ecol Manage 258:2090–2100
Aihartza JR, Garin I, Goiti U et al (2003) Spring habitat selection by the mediterranean horseshoe bat (Rhinolophus euryale) in the Urdaibai biosphere reserve (Basque country). Mammalia 67:25–32
Altringham JD (1996) Bats: biology and behaviour. Oxford University Press, Oxford
Arlettaz R (1999) Habitat selection as a major resource partitioning mechanism between the two sympatric sibling bat species Myotis myotis and Myotis blythii. J Anim Ecol 68:460–471
Arnett EB, Hayes JP (2009) Use of conifer snags as roosts by female bats in western forests. J Wildl Manage 73:214–225
Aubry KB, Halpern CB, Peterson CE (2009) Variable-retention harvests in the Pacific Northwest: a review of short-term findings from the DEMO study. For Ecol Manage 258:398–408
Baker MD, Lacki MJ (2006) Day roosting habitat of female long-legged myotis in ponderosa pine forests. J Wildl Manage 70:207–215
Baker SC, Read SM (2011) Variable retention silviculture in Tasmania’s wet forests: rationale, adaptive management and synthesis of biodiversity benefits. Aust For 74:218–232
Bernardos DA, Chambers CL, Rabe ML (2004) Selection of Gambel oak roosts by southwestern myotis in ponderosa pine-dominated forests, northern Arizona. J Wildl Manage 68:595–601
Betts BJ (1998) Roosts used by maternity colonies of silver-haired bats in northeastern Oregon. J Mammal 79:643–650
Boerner REJ, Hart SC, McIver JD (2008) The national fire and fire surrogate study: ecological consequences of alternative fuel reduction methods in seasonally dry forests. For Ecol Manage 255:3075–3080
Borkin K, Parsons S (2011a) Home range and habitat selection by a threatened bat in exotic plantation forest. For Ecol Manage 262:845–852
Borkin K, Parsons S (2011b) Sex-specific roost selection by bats in clearfell harvested plantation forest: improved knowledge advises management. Acta Chiropter 13:373–383
Borkin K, O’Donnell CFJ, Parsons S (2011) Bat colony size reduction coincides with clear-fell harvest operations and high rates of roost loss in plantation forest. Biodivers Cons 20:3537–3548
Boyles JG, Aubrey DP (2006) Managing forests with prescribed fire: implications for a cavity-dwelling bat species. For Ecol Manage 222:108–115
Brigham RM (2007) Bats in forests: what we know and what we need to learn. In: Lacki MJ, Hayes JP, Kurta A (eds) Bats in forests: conservation and management. The Johns Hopkins University Press, Baltimore, pp 1–15
Brigham RMR, Vonhof MJ, Barclay RM et al (1997) Roosting behaviour and roost-site preferences of forest-dwelling California bats (Myotis californicus). J Mammal 78:1231–1239
Britzke ER, Duchamp JE, Murray KL et al (2011) Acoustic identification of bats in the eastern United States: a comparison of parametric and nonparametric methods. J Wildl Manage 75:660–667
Broders HG, Forbes GJ (2004) Interspecific and intersexual variation in roost-site selection of northern long-eared and little brown bats in the Greater Fundy National Park ecosystem. J Wildl Manage 68:602–610
Brown GW, Nelson JL, Cherry KA (1997) The influence of habitat structure on insectivorous bat activity in montane ash forests of the central highlands, Victoria. Aust Forestry 60:138–146
Burford LS, Lacki MJ, Covell CV (1999) Occurrence of moths among habitats in a mixed mesophytic forest: implications for management of forest bats. For Sci 45:323–332
Carter TC, Feldhamer GA (2005) Roost tree use by maternity colonies of Indiana bats and northern long-eared bats in southern Illinois. For Ecol Manage 219:259–268
Cawthen L, Nicol S, Law B et al (2013) Effectiveness of the multi-spatial scale approach to forest management—a Tasmanian case study on bats. Abstract for the international bat conference, Costa Rica
Clement MJ, Castleberry SB (2013) Estimating density of a forest-dwelling bat: a predictive model for Rafinesque’s big-eared bat. Pop Ecol 55:205–215
Crampton LH, Barclay RMR (1998) Selection of roosting and foraging habitat by bats in different-aged aspen mixedwood stands. Conserv Biol 12:1347–1358
Crome FHJ, Richards GC (1988) Bats and gaps: microchiropteran community structure in a Queensland rainforest. Ecology 69:1960–1969
Cryan PM, Bogan MA, Yanega GM (2001) Roosting habits of four bat species in the black hills of South Dakota. Acta Chiropter 3:43–52
Curtin RA, Squire RH, Mackowski CM (1991) Management of native hardwood forests of north coast New South Wales. In: McKinnell FH, Hopkins ER, Fox JED (eds) Forest management in Australia. Surrey Beatty & Sons, NSW, pp 77–107
de Oliveira MC, Smith GC, Hogan LD (1999) Current limitations in the use of bat detectors to assess the impacts of logging—a pilot study in south-east Queensland. Aust Zool 31:110–117
Di Salvo I, Russo D, Sarà M (2009) Habitat preferences of bats in a rural area of Sicily determined by acoustic surveys. It J Mamm 20:137–146
Dodd LE, Lacki MJ (2007) Prey consumed by Corynorhinus townsendii ingens in the Ozark mountain region. Acta Chiropter 9:451–461
Dodd LE, Lacki MJ, Rieske LK (2008) Variation in moth occurrence and implications for foraging habitat of Ozark big-eared bats. For Ecol Manage 255:3866–3872
Dodd LE, Lacki MJ, Britzke ER et al (2012) Forest structure affects trophic linkages: how silvicultural disturbance impacts bats and their insect prey. For Ecol Manage 267:262–270
Dodd LE, Skowronski NS, Dickinson MB et al (2013) Using LiDAR to link forest canopy structure with bat activity and insect occurrence: preliminary findings. Mammoth cave national park’s 10th research symposium: celebrating the diversity of research in the mammoth cave region. Mammoth Cave, Kentucky, pp 50–57
Downs NC, Racey PA (2006) The use by bats of habitat features in mixed farmland in Scotland. Acta Chiropter 8:169–185
Duchamp JE, Arnett EB, Larson MA et al (2007) Ecological considerations for landscape-level management of bats. In: Lacki MJ, Hayes JP, Kurta A (eds) Bats in forests: conservation and management. The Johns Hopkins University Press, Baltimore, pp 237–261
Duerr DA, Mistretta PA (2013) Invasive pests—insects and diseases. In: Wear DN, Greis JG (eds) The southern forest futures project. USDA Forest Service, Southern Research Station, Gen. Tech. Rep. SRS-178, Asheville, North Carolina, pp 457–491
Elmore LW, Miller DA, Vilella FJ (2004) Selection of diurnal roosts by red bats (Lasiurus borealis) in an intensively managed pine forest in Mississippi. For Ecol Manage 199:11–20
Entwhistle AC, Racey PA, Speakman JR (1996) Habitat exploitation by a gleaning bat, Plecotus auritus. Phil Trans Roy Soc Lon Ser B 351:921–931
Erickson JL, West SD (1996) Managed forests in the western Cascades: the effects of seral stage on bat habitat use patterns. In: Barclay RMR, Brigham RM (eds) Bats and forests symposium, October 19–21, 1995, Victoria, British Columbia. Canadian Research Branch, BC Ministry of Forests, Victoria, BC. Working Paper 23/1996, pp 215–227
Erickson JL, West SD (2003) Associations of bats with local structure and landscape features of forested stands in western Oregon and Washington. Biol Conserv 109:95–102
European Environment Agency (2006) European forest types: categories and types for sustainable forest management reporting and policy. EEA Technical report No 9/2006. Available at: http://www.eea.europa.eu/publications/technical_report_2006_9
Fisher JT, Wilkinson L (2005) The response of mammals to forest fire and timber harvest in the North American boreal forest. Mammal Rev 35:51–81
Fleming HL, Jones JC, Belant JL et al (2013) Probability of detection and visual count error for Rafinesque’s big-eared bat (Corynorhinus rafinesquii) and southeastern myotis (Myotis austroriparius) in tree cavities. Amer Midl Nat 169:56–65
Florence RG (1996) Ecology and silviculture of eucalypt forests. CSIRO Publishing, Victoria
Food and Agricultural Organisation of the United Nations (2012). State of the World’s Forests, Rome. Available at: http://www.fao.org/docrep/016/i3010e/i3010e00.htm
Ford WM, Menzel MA, Rodrigue JL et al (2005) Relating bat species presence to simple habitat measures in a central Appalachian forest. Biol Conserv 126:528–539
Ford WM, Menzel JM, Menzel MA et al (2006) Presence and absence of bats across habitat scales in the upper coastal plain of South Carolina. J. Wildl Manage 70:1200–1209
Forestry Commission (2005) Woodland management for bats. Forestry Commission Publications, Wetherby UK. Available at: http://www.forestry.gov.uk/pdf/woodland-management-for-bats.pdf/$FILE/woodland-management-for-bats.pdf
Foster R, Kurta A (1999) Roosting ecology of the northern bat (Myotis septentrionalis) and comparisons with the endangered Indiana bat (Myotis sodalis). J Mammal 80:659–672
Fries C, Johansson O, Pettersson B et al (1997) Silvicultural models to maintain and restore natural stand structures in Swedish boreal forests. For Ecol Manage 94:89–103
Fuentes-Montemayor E, Goulson D, Cavin L et al (2012) Factors influencing moth assemblages in woodland fragments on farmland: implications for woodland creation and management schemes. Biol Conserv 153:265–275
Fuentes-Montemayor E, Goulson D, Cavin L et al (2013) Fragmented woodlands in agricultural landscapes: the influence of woodland character and landscape context on bats and their insect prey. Agri Ecosyst Environ 172:6–15
Greenaway F, Hill DA (2004) Woodland management advice for Bechstein’s and barbastelle bat. English Nature Research Reports no 658
Grindal SD (1996) Habitat use by bats in fragmented forests. In: Barclay RMR, Brigham RM (eds) Bats and forests symposium, October 19–21, 1995, Victoria, British Columbia. Canadian Research Branch, BC Ministry of Forests, Victoria, BC. Working Paper 23/1996, pp 260–272
Grindal SD (1999) Habitat use by bats, Myotis spp., in western Newfoundland. Can Field-Nat 113:258–263
Grindal SD, Brigham RM (1998) Short-term effects of small-scale habitat disturbance on activity by insectivorous bats. J Wildl Manage 62:996–1003
Grindal SD, Brigham RM (1999) Impacts of forest harvesting on habitat use by foraging insectivorous bats at different spatial scales. Ecoscience 6:25–34
Grindal SD, Morissette JL, Brigham RM (1999) Concentration of bat activity in riparian habitats over an elevation gradient. Can J Zool 77:972–977
Guldin JM, Emmingham WH, Carter SA et al (2007) Silvicultural practices and management of habitat for bats. In: Lacki MJ, Hayes JP, Kurta A (eds) Bats in forests: conservation and management. The Johns Hopkins University Press, Baltimore, pp 177–205
Gustafsson L, Baker SC, Bauhus J et al (2012) Retention forestry to maintain multifunctional forests: a world perspective. Bioscience 62:633–645
Hansen MC, Stehman SV, Potapov PV (2010) Quantification of global gross forest cover loss. Proc US Nat Acad Sci 107:8650–8655
Hanspach J, Fischer J, Stagoll K et al (2012) Using trait-based filtering as a predictive framework for conservation: a case study of bats on farms in southeastern Australia. J Appl Ecol 49:842–850
Harrod RJ, Peterson DW, Povak NA et al (2009) Thinning and prescribed fire effects on overstory tree and snag structure in dry coniferous forests of the interior Pacific Northwest. For Ecol Manage 258:712–721
Hart JA, Kirkland GL Jr, Grossman SC (1993) Relative abundance and habitat use by tree bats, Lasiurus spp., in southcentral Pennsylvania. Can Field-Nat 107:208–212
Hayes JP (2009) Post-fire salvage logging in central Oregon: short-term response in bats, birds and small mammals. Fire Sci Brief 1–6
Hayes JP, Adam MD (1996) The influence of logging riparian areas on habitat utilization by bats in western Oregon. In: Barclay RMR, Brigham RM (eds) Bats and forests symposium, October 19–21, 1995, Victoria, British Columbia. Canadian Research Branch, BC Ministry of Forests, Victoria, BC. Working Paper 23/1996, pp 228–237
Hayes JP, Loeb SC (2007) The influences of forest management on bats in North America. In: Lacki, MJ, Hayes, JP, Kurta (eds) Bats in forests: conservation and management. The Johns Hopkins University Press, Baltimore, pp 207–235
Hein CD, Castleberry SB, Miller KV (2008a) Male Seminole bat winter roost-site selection in a managed forest. J Wildl Manage 72:1756–1764
Hein CD, Castleberry SB, Miller KV (2008b) Sex-specific summer roost-site selection by Seminole bats in response to landscape-level forest management. J Mammal 89:964–972
Hein CD, Castleberry SB, Miller KV (2009a) Site-occupancy of bats in relation to forested corridors. For Ecol Manage 257:1200–1207
Hein CD, Miller KV, Castleberry SB (2009b) Evening bat summer roost-site selection on a managed pine landscape. J Wildl Manage 73:511–517
Henderson LE, Farrow LJ, Broders HG (2008) Intra-specific effects of forest loss on the distribution of the forest-dependent northern long-eared bat (Myotis septentrionalis). Biol Conserv 141:1819–1828
Hessburg PF, Povak NA, Salter RB (2010) Thinning and prescribed fire effects on snag abundance and spatial pattern in an eastern Cascade range dry forest, Washington, USA. For Sci 56:74–87
Hill DA, Greenaway F (2008) Conservation of bats in British woodland. Brit Wild 161:161–169
Hillen J, Kiefer A, Veith M (2010) Interannual fidelity to roosting habitat and flight paths by female western barbastelle bats. Acta Chiropter 12:187–195
Hogberg LK, Patriquin KJ, Barclay RMR (2002) Use by bats of patches of residual trees in logged areas of the boreal forest. Amer Midl Nat 148:282–288
Humes ML, Hayes JP, Collopy MW (1999) Bat activity in thinned, unthinned, and old-growth forests in western Oregon. J Wildl Manage 63:553–561
Humphrey JW (2005) Benefits to biodiversity from developing old-growth conditions in British upland spruce plantations: a review and recommendations. Forestry 78:33–53
Humphrey JW, Bailey S (2012) Managing deadwood in forests and woodlands. Forestry Commission Practice Guide. Forestry Commission, Edinburgh. i–iv+ 1–24 pp
Hutchinson JT, Lacki MJ (2000) Selection of day roosts by red bats in mixed mesophytic forests. J Wildl Manage 64:87–94
Jantzen MK, Fenton MB (2013) The depth of edge influence among insectivorous bats at forest-field interfaces. Can J Zool 91:287–292
Johnson JB, Ford WM, Edwards JW (2012a) Roost networks of northern myotis (Myotis septentrionalis) in a managed landscape. For Ecol Manage 266:223–231
Johnson JS, Kropczynski JN, Lacki MJ et al (2012b) Social networks of Rafinesque’s big-eared bats (Corynorhinus rafinesquii) in bottomland hardwood forests. J Mammal 93:1545–1558
Johnson JS, Kropczynski JN, Lacki MJ (2013) Social network analysis and the study of sociality in bats. Acta Chiropter 15:1–17
Jung K, Kaiser S, Böhm S et al (2012) Moving in three dimensions: effects of structural complexity on occurrence and activity of insectivorous bats in managed forest stands. J Appl Ecol 49:523–531
Kalcounis MC, Hobson KA, Brigham RM et al (1999) Bat activity in the boreal forest: importance of stand type and vertical strata. J Mammal 80:673–682
Kalcounis-Rüppell MC, Pysllakis JM, Brigham RM (2005) Tree roost selection by bats: an empirical synthesis using meta-analysis. Wildl Soc Bull 33:1123–1132
Kavanagh RP, Webb G (1998) Effects of variable-intensity logging on mammals, reptiles and amphibians at Waratah creek, Southeastern New South Wales. Pac Cons Biol 4:326–347
Kerth G, König B (1999) Fission, fusion and nonrandom associations in female Bechstein’s bats (Myotis bechsteinii). Behaviour 136:1187–1202
Klug BJ, Goldsmith DA, Barclay RMR (2012) Roost selection by the solitary, foliage-roosting hoary bat (Lasiurus cinereus) during lactation. Can J Zool 90:329–336
Kroll AJ, Lacki MJ, Arnett EB (2012) Research needs to support management and conservation of cavity-dependent birds and bats on forested landscapes in the Pacific Northwest. Western J Appl Forest 27:128–136
Krusic RA, Yamasaki M, Neefus CD et al (1996) Bat habitat use in white Mountain National Forest. J Wildl Manage 60:625–631
Lacki MJ, Schwierjohann JH (2001) Day-roost characteristics of northern bats in mixed mesophytic forest. J Wildl Manage 65:482–488
Lacki MJ, Amelon SK, Baker MD (2007) Foraging ecology of bats in forests. In: Lacki MJ, Hayes JP, Kurta A (eds) Bats in forests: conservation and management. Johns Hopkins University Press, Baltimore, pp 83–127
Lacki MJ, Cox DR, Dickinson MB (2009) Meta-analysis of summer roosting characteristics of two species of Myotis bats. Amer Midl Nat 161:321–329
Lacki MJ, Baker MD, Johnson JS (2010) Geographic variation in roost-site selection of long-legged myotis in the Pacific Northwest. J Wildl Manage 74:1218–1228
Lacki MJ, Baker MD, Johnson JS (2012) Temporal dynamics of roost snags of long-legged myotis in the Pacific Northwest, USA. J Wildl Manage 76:1310–1316
Lähde E, Laiho O, Norokorpi Y (1999) Diversity-orientated silviculture in the Boreal zone of Europe. For Ecol Manage 118:223–243
Law BS (1996) The ecology of bats in south-east Australian forests and potential impacts of forestry practices: a review. Pac Cons Biol 2:363–374
Law BS (2004) Challenges for the management of bats in state forests of NSW. In: Lunney D (ed) The conservation of Australia’s forest fauna, 2nd edn. Royal Zoological Society of NSW, Sydney, pp 748–760
Law BS, Anderson J (2000) Roost preferences and foraging ranges of the eastern forest bat Vespadelus pumilus under two disturbance histories on the mid-north coast of New South Wales. Aust Ecol 25:352–367
Law BS, Chidel M (2001) Bat activity 22 years after first-round intensive logging of alternate coupes near Eden, NSW. Aust Forestry 64:242–247
Law BS, Chidel M (2002) Tracks and riparian zones facilitate the use of Australian regrowth forest by insectivorous bats. J Appl Ecol 39:605–617
Law B, Chidel M (2004) Roosting and foraging ecology of the golden-tipped bat Kerivoula papuensis on the south coast of New South Wales. Wildl Res 31:73–82
Law B, Chidel M (2006) Eucalypt plantings on farms: use by insectivorous bats. Biol Cons 133:236–249
Law BS, Law PR (2011) Early responses of bats to alternative silvicultural treatments in wet eucalypt forests of Tasmania. Pac Cons Biol 17:36–47
Law BS, Chidel M, Penman T (2011) Do young eucalypt plantations benefit bats in an intensive agricultural landscape? Wildl Res 38:173–187
Lee P, Smyth C, Boutin S (2004) Quantitative review of riparian width buffer guidelines from Canada and the United States. J Environ Manage 70:165–180
Limpert DL, Birch DL, Scott MS et al (2007) Tree selection and landscape analysis of eastern red bat day roosts. J Wildl Manage 71:478–486
Lindenmayer DB, Franklin JF (2002) Conserving forest biodiversity: a comprehensive multiscaled approach. Island Press, Washington
Lindenmayer DB, Noss RF (2006) Salvage logging, ecosystem processes, and biodiversity conservation. Conserv Biol 20:949–958
Lindenmayer DB, Franklin JF, Lõhmus A et al (2012) A major shift to the retention approach for forestry can help resolve some global forestry sustainability issues. Conserv Letters 5:421–431
Lintott P, Minderman J, Fuentes-Montemayor E et al (2014) Moth species richness, abundance and diversity in fragmented urban woodlands: implications for conservation and management strategies. Biodiv Conserv 23:2875–2901
Lloyd A, Law B, Goldingay R (2006) Bat activity on riparian zones and upper slopes in Australian timber production forests and the effectiveness of riparian buffers. Biol Cons 129:207–220
Loeb SC, O’Keefe JM (2006) Habitat use by forest bats in South Carolina in relation to local, stand, and landscape characteristics. J Wildl Manage 70:1210–1218
Loeb SC, Waldrop TA (2008) Bat activity in relation to fire and fire surrogate treatments in southern pine stands. For Ecol Manage 255:3185–3192
Long JN (2009) Emulating natural disturbance regimes as a basis for forest management: a North American view. For Ecol Manage 257:1868–1873
Luck GW, Smallbone L, Threlfall C et al (2013) Patterns in bat functional guilds across multiple urban centres in south-eastern Australia. Landscape Ecol 28:455–469
Lumsden LF, Bennett AF, Silins JE (2002) Location of roosts of the lesser long-eared bat Nyctophilus geoffroyi and Gould’s wattled bat Chalinolobus gouldii in a fragmented landscape in south-eastern Australia. Biol Cons 106:237–249
Lunde RE, Harestad AS (1986) Activity of little brown bats in coastal forests. Northwest Sci 60:206–209
Lunney D, Barker J, Priddel D et al (1988) Roost selection by Gould’s long-eared bat, Nyctophilus gouldi Thomas (Chiroptera: Vespertilionidae), in logged forest on the south coast of New South Wales. Aust Wildl Res 15:375–384
Maron M, Lill A, Watson DM et al (2005) Temporal variation in bird assemblages: how representative is a one-year snapshot? Aust Ecology 30:383–394
Mason WL, Quine CP (1995) Silvicultural possibilities for increasing structural diversity in British spruce forests: the case of Kielder. For Ecol Manage 79:13–28
Mason B, Kerrand G, Simpson J (1999) What is continuous cover forestry? Forestry commission information note 29. Forestry Commission, Edinburgh. Available at: http://www.forestry.gov.uk/pdf/fcin29.pdf/$FILE/fcin29.pdf
Mattson TA, Buskirk SW, Stanton NL (1996) Roost sites of the silver-haired bat (Lasionycteris noctivagans) in the Black Hills, South Dakota. Great Basin Nat 56:247–253
Mazurek MJ, Zielinski WJ (2004) Individual legacy trees influence vertebrate wildlife diversity in commercial forests. For Ecol Manage 193:321–334
Mehr M, Brandl R, Kneib T et al (2012) The effect of bark beetle infestation and salvage logging on bat activity in a national park. Biodivers Conserv 21:2775–2786
Menzel MA, Carter TC, Menzel JM et al (2002) Effects of group selection silviculture in bottomland hardwoods on the spatial activity patterns of bats. For Ecol Manage 162:209–218
Meyer CFJ, Matthew JS, Willig MR (2016) Responses of tropical bats to habitat fragmentation, logging, and deforestation. In: Voigt CC, Kingston T (eds) Bats in the Anthropocene: conservation of bats in a changing world. Springer International AG, Cham, pp. 105–141
Meyer CFJ, Fründ J, Lizano WP et al (2008) Ecological correlates of vulnerability to fragmentation in Neotropical bats. J Appl Ecol 45:381–391
Miles AC, Castleberry SB, Miller DA et al (2006) Multi-scale roost-site selection by evening bats on pine-dominated landscapes in southwestern Georgia. J Wildl Manage 70:1191–1199
Miller DA (2003) Species diversity, reproduction, and sex ratios of bats in managed pine forest landscapes of Mississippi. Southeast Nat 2:59–72
Miller DA, Stihler CW, Sasse DB et al. (2011) Conservation and management of eastern big-eared bats (Corynorhinus spp.). In: Loeb SC, Lacki MJ, Miller DA (eds) Conservation and management of eastern big-eared bats: a symposium. USDA, Forest Service, Southern Research Station, GTR SRS-145. Asheville, North Carolina, pp 53–61
Mississippi Museum of Natural Science (2005) Mississippi’s comprehensive wildlife conservation strategy. Mississippi Department of Wildlife, Fisheries, and Parks, Jackson, Mississippi
Mitchell RJ, Palik BJ, Hunter ML Jr (2002) Natural disturbance as a guide to silviculture. For Ecol Manage 155:315–317
Montreal Process Implementation Group for Australia (2008) Australia’s state of the forests report 2008. Bureau of Rural Sciences, Canberra
Moore PT, DeRose RJ, Long JN et al (2012) Using silviculture to influence carbon sequestration in southern Appalachian spruce-fir forests. Forests 3:300–316
Mormann BM, Robbins LW (2007) Winter roosting ecology of eastern red bats in southwest Missouri. J Wildl Manage 71:213–217
Mortimer G (2006) Foraging, roosting and survival of Natterer’s bats, Myotis nattereri, in a commercial coniferous plantation. PhD thesis, St. Andrews University, UK
Müller J, Mehr M, Bässler C et al (2012) Aggregative response in bats: prey abundance versus habitat. Oecologia 169:673–684
Müller J, Brandl R, Buchner J et al (2013) From ground to above canopy—bat activity in mature forests is driven by vegetation density and height. For Ecol and Manage 360:179–184
Murphy SE, Greenaway F, Hill DA (2012) Patterns of habitat use by female brown long-eared bats presage negative impacts of woodland conservation management. J Zool (Lond) 288:177–183
Murray SW, Kurta A (2004) Nocturnal activity of the endangered Indiana bat (Myotis sodalis). J Zool (Lond) 262:197–206
Nicholson E (1999) Winds of change for silvicultural practice in NSW native forests. Aust For 62:223–235
O’Hara KL (2002) The historical development of uneven-aged silviculture in North America. Forestry 75:339–346
O’Hara KL (2009) Multiaged silviculture in North America. J For Sci 55:432–436
O’Keefe JM, Loeb SC, Lanham JD et al (2009) Macrohabitat factors affect day roost selection by eastern red bats and eastern pipistrelles in the southern Appalachian Mountains, USA. For Ecol Manage 257:1757–1763
Ober HK, Hayes JP (2008) Influence of vegetation on bat use of riparian areas at multiple spatial scales. J Wildl Manage 72:396–404
Owen SF, Menzel MA, Ford WM et al (2003) Home-range size and habitat used by the northern myotis (Myotis septentrionalis). Amer Midl Nat 150:352–359
Owen SF, Menzel MA, Edwards JW et al (2004) Bat activity in harvested and intact forest stands in the Allegheny Mountains. Northern J Appl Forest 21:154–159
Park KJ, Masters E, Altringham JD (1998) Social structure of three sympatric bat species (Vespertilionidae). J Zool (Lond) 244:379–389
Parker DI, Cook JA, Lewis SW (1996) Effects of timber harvest on bat activity in southeastern Alaska’s temperate rainforests. In: Barclay RMR, Brigham RM (eds) Bats and forests symposium, October 19–21, 1995, Victoria, British Columbia. Canadian Research Branch, BC Ministry of Forests, Victoria, BC. Working Paper 23/1996, pp 277–292
Parks CG, Bernier P (2010) Adaptation of forests and forest management to changing climate with emphasis on forest health: a review of science, policies and practices. For Ecol Manage 259:657–659
Patriquin KJ, Barclay RMR (2003) Foraging by bats in cleared, thinned and unharvested boreal forest. J Appl Ecol 40:646–657
Perdue M, Steventon JD (1996) Partial cutting and bats: a pilot study. In: Barclay RMR, Brigham RM (eds) Bats and forests symposium, October 19–21, 1995, Victoria, British Columbia. Canadian Research Branch, BC Ministry of Forests, Victoria, BC. Working Paper 23/1996, pp 273–276
Perry RW, Thill RE (2007a) Roost characteristics of hoary bats in Arkansas. Amer Midl Nat 158:132–138
Perry RW, Thill RE (2007b) Roost selection by male and female northern long-eared bats in a pine dominated landscape. For Ecol Manage 247:220–226
Perry RW, Thill RE (2008) Diurnal roosts of male evening bats (Nycticeius humeralis) in diversely managed pine-hardwood forests. Amer Midl Nat 160:374–385
Perry RW, Thill RE, Carter SA (2007a) Sex-specific roost selection by adult red bats in a diverse forested landscape. For Ecol Manage 253:48–55
Perry RW, Thill RE, Leslie DM Jr (2007b) Selection of roosting habitat by forest bats in a diverse forest landscape. For Ecol Manage 238:156–166
Perry RW, Thill RE, Leslie DM Jr (2008) Scale-dependent effects of landscape structure and composition on diurnal roost selection by forest bats. J Wildl Manage 72:913–925
Pettit TW, Wilkins KT (2012) Canopy and edge activity of bats in a quaking aspen (Populus tremuloides) forest. Can J Zool 90:798–807
Puettmann KJ, Coates KD, Messier C (2009) A critique of silviculture: managing for complexity. Island Press, Washington, DC
Rabe MJ, Morrell TE, Green H et al (1998) Characterisitcs of ponderosa pine snag roosts used by reproductive bats in northern Arizona. J Wildl Manage 62:612–621
Recher HF, Gowing G, Kavanagh R et al (1983) Birds, resources and time in a tablelands forest. Proc Ecol Soc Aust 12:101–123
Regnery B, Couvet D, Kubarek L et al (2013a) Tree microhabitats as indicators of bird and bat communities in Mediterranean forests. Ecol Indic 34:221–230
Regnery B, Paillet Y, Couvet D et al (2013b) Which factors influence the occurrence and density of tree microhabitats in Meditteranean Oak forests? For Ecol Manage 295:118–125
Rhodes M, Wardell-Johnson GW, Rhodes MP et al (2006) Applying network analysis to the conservation of habitat trees in urban environments: a case study from Brisbane, Australia. Cons Biol 20:861–870
Ruczyński I, Bogdanowicz W (2008) Summer roost selection by tree-dwelling bats Nyctalus noctula and N. leisleri: a multiscale analysis. J Mammal 89:942–951
Ruczyński I, Nicholls B, MacLeod CD et al (2010) Selection of roosting habitats by Nyctalus noctula and Nyctalus leisleri in Białowieża Forest—adaptive response to forest management? For Ecol Manage 259:1633–1641
Russo D, Cistrone L, Jones G et al (2004) Roost selection in barbastelle bats (Barbastella barbastellus, Chiroptera: Vespertilionidae) in beech woodlands of central Italy: consequences for conservation. Biol Cons 117:73–81
Russo D, Almenar D, Aihartza J, Goiti U, Salsamendi E, Garin I (2005a) Habitat selection in sympatric Rhinolophus mehelyi and R. euryale (Mammalia: Chiroptera). J Zool Lond 266:327–332
Russo D, Cistrone L, Jones G (2005b) Spatial and temporal patterns of roost use by tree-dwelling barbastelle bats, Barbastellus barbastellus. Ecography 28:769–776
Russo D, Cistrone L, Jones G (2007) Emergence time in forest bats: the influence of canopy closure. Acta Oecolgica 31:119–126
Russo D, Cistrone L, Garonna AP et al (2010) Reconsidering the importance of harvested forests for the conservation of tree-dwelling bats. Biodivers Conserv 19:2501–2515
Sasse DB, Pekins PJ (1996) Summer roosting ecology of northern long-eared bats (Myotis septentrionalis) in the White Mountain National Forest. In: Barclay RMR, Brigham RM (eds) Bats and forests symposium, October 19–21, 1995, Victoria, British Columbia. Canadian Research Branch, BC Ministry of Forests, Victoria, BC. Working Paper 23/1996, pp 91–101
Schnitzler H-U, Moss CF, Denzinger A (2003) From spatial orientation to food acquisition in echolocating bats. Trends Ecol Evol 18:386–394
Schulz M (2000) Roosts used by the golden-tipped bat Kerivoula papuensis (Chiroptera: Vespertilionidae). J Zool (Lond) 250:467–478
Sedgeley JA, O’Donnell CFJ (1999) Roost selection by the long-tailed bat, Chalinolobus tuberculatus, in temperate New Zealand rainforest and its implications for the conservation of bats in managed forests. Biol Cons 88:261–276
Senior P, Butlin RK, Altringham JD (2005). Sex and segregation in temperate bats. Proc Roy Soc B—Biol Sci 272:2467–2473
Society of American Foresters (2010) Forest types of North America. Available at: http://www.encyclopediaofforestry.org/index.php?title=BioEco12
Stringer JW, Perkins C (2001) Kentucky forest practice guidelines for water quality management. University of Kentucky, College of Agriculture, Cooperative Extension Service, FOR-67, Lexington, Kentucky
Summerville KS, Crist TO (2002) Effects of timber harvest on forest Lepidoptera: community, guild, and species responses. Ecol Applica 12:820–835
Swystun MB, Psyllakis JM, Brigham RM (2001) The influence of residual tree patch isolation on habitat use by bats in central British Columbia. Acta Chiropter 3:197–201
Taylor RJ, Savva NM (1988) Use of roost sites by four species of bats in state forest in south-eastern Tasmania. Aust Wildl Res 15:637–645
Thomas DW (1988) The distribution of bats in different ages of Douglas-fir forests. J Wildl Manage 52:619–626
Tibbels AE, Kurta A (2003) Bat activity is low in thinned and unthinned stands of red pine. Can J For Res 33:2436–2442
Titchenell MA, Williams RA, Gehrt SD (2011) Bat responses to shelterwood harvests and forest structure in oak-hickory forests. For Ecol Manage 262:980–988
U.S. Department of Agriculture Forest Service (2009) U.S. forest resource facts and historic trends: forest facts 1952–2007 US metric revised rev072411. FS-801 M. Available at: http://fia.fs.fed.us
U.S. Department of Agriculture and U.S. Department of the Interior (1994) Record of decision for amendments to Forest Service and Bureau of Land Management planning documents within the range of the northern spotted owl. U.S. Forest Service, Portland, Oregon
U.S. Fish and Wildlife Service (2009) Range-wide Indiana bat protection and enhancement plan guidelines. Available at: http://www.fws.gov/frankfort/pdf/inbatpepguidelines
U.S. Fish and Wildlife Service (2012) North American bat death toll exceeds 5.5 million from white-nose syndrome. News Release, U.S. Fish and Wildlife Service, Arlington, Virginia
Vonhof MJ, Barclay RMR (1997) Use of tree stumps as roosts by the western long-eared bat. J Wildl Manage 61:674–684
Waldien DL, Hayes JP, Arnett EB (2000) Day-roosts of female long-eared myotis in western Oregon. J Wildl Manage 64:785–796
Walsh AL, Harris S (1996) Factors determining the abundance of vespertilionid bats in Britain: geographical, land class and local habitat relationships. J Appl Ecol 33:519–529
Watrous KS, Donovan TM, Mickey RM et al (2006) Predicting minimum habitat characteristics for the Indiana bat in the Champlain Valley. J Wildl Manage 70:1228–1237
Wear DN, Greis JG (2013) The southern forests futures project. USDA Forest Service, Southern Research Station, Gen. Tech. Rep. SRS-178, Asheville, North Carolina
Webala PW, Craig MD, Law BS et al (2010) Roost site selection by southern forest bat Vespadelus regulus and Gould’s long-eared bat Nyctophilus gouldi in logged jarrah forests, south-western Australia. For Ecol Manage 260:1780–1790
Webala PW, Craig MD, Law BS et al (2011) Bat habitat use in logged jarrah eucalypt forests of south-western Australia. J Appl Ecol 48:398–406
Wigley TB, Miller DA, Yarrow GK (2007) Planning for bats on forest industry lands in North America. In: Lacki MJ, Hayes JP, Kurta A (eds) Bats in forests: conservation and management. Johns Hopkins University Press, Baltimore, pp 293–318
Willis CKR, Brigham RM (2004) Roost switching, roost sharing and social cohesion: forest-dwelling big brown bats, Eptesicus fuscus, conform to the fission-fusion model. Anim Behav 68:495–505
Willis CKR, Brigham RM (2005) Physiological and ecological aspects of roost selection by reproductive female hoary bats (Lasiurus cinereus). J Mammal 86:85–94
World Resources Institute (2014) Southern forests for the future. Available at: http://www.seesouthernforests.org
Wunder L, Carey AB (1996) Use of the forest canopy by bats. Northwest Sci 70:79–85
Yates MD, Muzika RM (2006) Effect of forest structure and fragmentation on site occupancy of bat species in Missouri Ozark forests. J Wildl Manage 70:1238–1248
Young RA, Giese RL (eds) (2003) Introduction to forest ecosystem science and management, 3rd edn. John Wiley and Sons, New York
Zahn A, Haselbach H, Güttinger R (2004) Foraging activity of central European Myotis myotis in a landscape dominated by spruce monocultures. Mamm Biol 70:265–270
Zimmerman GS, Glanz WE (2000) Habitat use by bats in eastern Maine. J Wildl Manage 64:1032–1040
Acknowledgements
Thanks to L. Cawthen, L. Lumsden, T. Kingston, J. Müller, D. Russo, and J. Williams for helpful comments on a draft.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Glossary
- Clearcut/Clear-fell Harvest
-
Also referred to as uniform selection and heavy group selection, it removes all trees from a large management area and allows natural regeneration to take place, resulting in even-aged regrowth with high stem density. The aim is to mimic natural stand replacing events such as wildfire or large storms
- Coupe/Cutblocks
-
A defined area of forest, which may vary in size, in which harvesting takes place usually over one year
- Deferment Harvests
-
Sometimes also referred to as a shelterwood or clearcut with reserves. A deferment harvest retains a limited number of canopy trees (reserve trees) while allowing regeneration in the understory. These two tree levels are then allowed to develop together until the end of the next rotation, whereupon other trees are retained for canopy cover
- Forest Zoning
-
Where management for multiple objectives in a forest incorporates broad exclusion areas such that logging is excluded from patches of forest deemed to be environmentally sensitive or where patches of forest are specified to allow different silvicultural practices (Florence 1996)
- Gap Release
-
Creation of canopy gaps typically <0.1 ha to allow the growth of younger, often suppressed trees
- Green Tree
-
The retention of live trees on an otherwise harvested area as part of a variable retention harvest
- Group Selection Harvest
-
Removes all trees from small patches, with the aim of using disturbance to stimulate regeneration of new trees, but simultaneously maintaining a well-connected mosaic of patches of varying size, containing varying numbers of residual mature trees
- LiDAR
-
A remote sensing technology that measures distance by illuminating a target with a laser and analyses the reflected light
- Patch Cuts
-
An area of felling smaller than a clearcut but removing a larger number of trees than a group selection harvest
- Prescriptions
-
Targeted retention that aims to mitigate the effects of logging on environmental features. Hollow tree retention and riparian exclusion zones are two common prescriptions, but can also include exclusion zones surrounding significant bat roosts
- SeedTree Harvest
-
The retention of a few residual trees in a harvested area to provide seeds for the forest to regenerate
- Self-thinning
-
Density-dependent mortality within an even-aged stand of trees as they grow in size, leading to reduced tree density
- Shelterwood Harvest
-
See deferment harvest.
- Shelterwood Systems
-
Removal of canopy trees in a series of selective harvests leaving sufficient trees for regeneration and shelter. New seedlings are left to establish before mature trees are removed
- Silviculture
-
The art and science of manipulating a stand of trees by controlling the supplies of water, nutrients, and solar radiation by altering forest structure, towards a desired future condition (Guldin et al. 2007), typically for timber production but also for biodiversity conservation goals
- Single Tree Selection
-
Removes a scattering of high value individual trees from management areas, with repeat cuts taking place at regular intervals over time. However, intensity can vary. Cumulative effects can result in reduced hollow tree density unless there is a specific retention of old trees
- Stand
-
A group of forest trees sufficiently uniform in species composition or age to be considered a management unit
- Thinning
-
Felling to decrease tree stem density within young regrowth forests to reduce competition for resources among trees and promote the growth of the stand (Florence 1996)
- Variable Retention Harvests
-
Creation of multi-aged stands in clearcut zones by retaining clumps, patches, or aggregates of old trees within the clearcut
Rights and permissions
Open Access This chapter is distributed under the terms of the Creative Commons Attribution Noncommercial License, which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Copyright information
© 2016 The Author(s)
About this chapter
Cite this chapter
Law, B., Park, K.J., Lacki, M.J. (2016). Insectivorous Bats and Silviculture: Balancing Timber Production and Bat Conservation. In: Voigt, C., Kingston, T. (eds) Bats in the Anthropocene: Conservation of Bats in a Changing World. Springer, Cham. https://doi.org/10.1007/978-3-319-25220-9_5
Download citation
DOI: https://doi.org/10.1007/978-3-319-25220-9_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-25218-6
Online ISBN: 978-3-319-25220-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)