Abstract
Phylogenetic diversity has become invaluable for conservation biology in the last decades, reflecting its link to option values and to evolutionary potential. We argue that its use will continue to grow rapidly in the next decades because of the transformation of systematics with new molecular techniques and especially metagenomics. In a near future, phylogenetic diversity typically will be the very first result at hand, and the great challenge of biodiversity sciences will be to preserve its link with natural history and the remainder of biological knowledge through species vouchers and names. The phylogeny availability and the very wide sampling allowed will facilitate obtaining detailed biodiversity information at local scale and considering the transition across scales – a fundamental need well highlighted in international conservation guidelines, and historically so difficult to achieve. All this suggests that phylogenetic diversity might be at the center of more explicit identification of conservation priorities and options. For concluding, we explore an emerging local-to-regional-to-global challenge: the possibility of defining “planetary boundaries” for biodiversity on the basis of phylogenetic diversity.
Given the rate at which sequence data in the public domain are accumulating, with initiatives to sequence the entire biota … on the horizon, it seems likely that within a decade or two, phylogenetic data will cease to be the limiting factor: It could even be that an organism’s place in the Tree of Life often will be one of the few things we know about it. Mace et al. (2003)
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
Phylogenetic diversity is now a core part of conservation biology, reflecting its link to option values and to evolutionary potential . Further, there is good overlap with related issues in broader ecology. These include community ecology’s interest in productivity (e.g. Cadotte et al. 2012), resilience (e.g. Pugliesi and Rapini 2015) and the functioning of evosystems (e.g. Srivastava et al. 2012) and microbial ecology’s use of PD as a cornerstone for exploring diversity patterns at multiple scales (Lozupone and Knight 2005, 2008; Faith et al. 2009). As the chapters in this book demonstrate, the development of new methods and their applications are very much tuned into human impacts and sustainability issues. Thus, red list ings, drivers of extinction, and changes in spatial and temporal distribution of phylogenetic diversity are common elements of these studies. All this promotes the incorporation of phylogenetic diversity in the international conservation agenda.
These prospects are magnified by the remarkable facilities for obtaining entire or large parts of genomes or other molecular sequences of any kind of organisms, and by the sheer magnitude of biological (gene sequences, trait databases, species occurrences, red list s) and environmental data (climate layers for past, present and future interpolated to very fine spatial scale s; land-use layers, spatial data indicating particular important risks such as fires, floods, and so on) now available in the public domain. All these allow for rapid estimation of the phylogenetic relationships for a large number of organisms in association with potential distribution and threats for species and lineages. In addition, under the stimulus of modern phylogenetic and molecular methods, systematics is going through a significant transformation that will certainly influence biodiversity conservation (Mace et al. 2003; Pons et al. 2006; Vogler and Monaghan 2006; Faith et al. 2010; Yahara et al. 2010). For closing this book, we will briefly describe this transformation of systematics and then discuss some impacts of these changes in biological conservation. We finish by exploring the possibility of defining “planetary boundaries ” for biodiversity on the basis of phylogenetic diversity , and its important role in linking biodiversity into broader societal perspectives and needs.
In Phase with Modern Systematics and NGS Methods: The Tree First, Then the Species
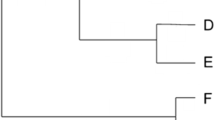
Conventionally, species are first characterized, then described with morphological or molecular data, and only then analyzed for building a phylogenetic tree (Fig. 1). As the entire operation demanded a long time and effort of specialists, the extent to which the later stage of the process – calculations of phylogenetic diversity – provided additional information “worth waiting for” was a recurrent and important question. Stopping at the first step and using species richness was accepted as a good proxy of biodiversity and sometimes justified, as when phylogenetic trees were expected to be balanced, or when the species with higher values of phylogenetic diversity were widespread , so not bringing important additional information (Rodrigues et al. 2005; Hartmann and André 2013). This rationale involved an unfortunate circular reasoning in that it required the knowledge about the phylogeny of the group to be able to discard it.
The traditional data processing in systematics, beginning with the sampling of specimens and the characterization and description of species. Specimens were then specifically sampled for phylogenetic characters, allowing to build phylogenetic trees and to compute phylogenetic diversity . In parallel, other biological knowledge was obtained separately. Note that in this framework, the number of species is obtained at the early step of species characterization (the so-called “morphospecies” may be obtained before description if necessary) before the phylogenetic analysis (Drawings by Agathe Haevermans)
This process now has been turned upside down. In the new paradigm, systematics proceeds in an all-in-one operation i.e. the result of the data processing makes that the species position on the tree is part of species delimitation and characterization (Fig. 2). A global sample of characters (e.g. DNA ) x individuals can be partitioned into clusters – potentially species – through a tree-like guidance. The new rationale is simple: to define species, we need first to recognize and delimit different groups of individuals, by contrasting their characters (Goldstein et al. 2000; Pons et al. 2006; Vogler and Monaghan 2006; O’Meara 2010; Pante et al. 2015; but see DeSalle et al. 2005). This phylogenetic perspective is still certainly new for many, although it is inexorably implemented in the most recent molecular methods used for biodiversity exploration and characterization, such as molecular species delimitation or metagenomics.
The new and upcoming data processing in systematics, beginning with molecular characterization or even metagenomics, jointly allowing phylogenetic analysis and species characterization, and therefore computation of phylogenetic diversity . Note also that some assessments of phylogenetic diversity may proceed without the species characterization. The species description and name attribution is the last, but not the least, step to keep molecular data connected with biological knowledge. Note that in the same line, proteomics could provide – to some extent – the species functional characterization, with molecular analysis on its own (Drawings by Agathe Haevermans)
Metagenomics recently went one step further by considering global amounts of DNA from environmental samples. In this approach there is no need to assemble the matrix ‘individuals x characters’ that is already all in the test tube. This technique is also remarkable by capturing all DNA at the same time and carrying out a very wide sampling including microbes and all organisms usually ignored by traditional taxonomic screening (Tringe and Rubin 2005; Yahara et al. 2010). Combined with proteomics, it can even provide functional information at the same time, by obtaining both DNA species and protein synthesis. At the point we are now, systematics is therefore able to offer a comprehensive picture of diversity , linking species, their relationships and their characters to conservation biology (Funk et al. 2002; Wilson 2003; Faith et al. 2010; Lean and MacLaurin chapter “The Value of Phylogenetic Diversity ”).
Obviously, this new framework enhanced by molecular biology and metagenomics will maintain biological significance and usefulness as long as molecular proxies will remain related to species concepts, taxon names and classifications linking to the wider biological knowledge (Mace 2004; German National Academy of Sciences Leopoldina 2014). The peril to invest only in an isolated and blind molecular database was already keenly emphasized by many taxonomists at the occasion of the rise of the barcoding initiative (e.g., Will et al. 2005). Building the network between names, biological knowledge and molecular data is from far the biggest challenge of present-day systematics and other sciences of diversity , much beyond the molecular technical tour de force (Grandcolas et al. 2013). We must keep in mind that this challenge takes place in a difficult moment when discovery rates of species new to science do not decline (Tancoigne and Dubois 2013) but in a context of rising rates of extinction (Régnier et al. 2015).
Impacts on Biodiversity Conservation
The first impact of these methodological breakthroughs in biodiversity conservation is the growing availability of phylogenies with adequate taxon and character sampling at fine scale . As a consequence, it will increase not only the possibility of identifying taxa and areas whose conservation will maximize phylogenetic diversity (Forest et al. 2007; Buerki et al. 2015; Soulebeau et al. chapter “ Conservation of Phylogenetic Diversity in Madagascar ’s Largest Endemic Plant Family, Sarcolaenaceae ”) or whose loss would contribute to major losses of our evolutionary heritage (Faith and Richards 2012; Faith 2015). It will also facilitate the transition across scales, a fundamental need well highlighted in international conservation guidelines (e.g. the Convention on Biological Diversity , “CBD ”), and historically so difficult to achieve. For example, some targets can be established at a global scale based on a general phylogeny (for example, a phylogeny with samples of all genera or families), and a more detailed phylogeny with the regional diversification of the group (including for example a large sample of the species occurring in this region) will allow for establishing the areas to be protected for attainment of the broader target. This, associated with modern methods of Systematic Conservation Planning (Moilanen and Arponen 2011; Kukkala and Moilanen 2013; Faith chapter “Using Phylogenetic Dissimilarities Among Sites for Biodiversity Assessments and Conservation”) in which biological variables, including phylogenetic diversity can be considered along with costs, risks and return to investment, will certainly contribute to more explicit identification of conservation priorities and options (Pollock et al. 2015; Arponen and Zupan chapter “Representing Hotspots of Evolutionary History in Systematic Conservation Planning for European Mammals ”; Silvano et al. chapter “Priorities for Conservation of the Evolutionary History of Amphibians in the Cerrado ”). With these developments in mind, we will close this book by exploring an emerging local-to-regional-to-global challenge: the possibility of defining “planetary boundaries ” for biodiversity on the basis of phylogenetic diversity.
Phylogenetic Diversity as a Basis for Defining “Planetary Boundaries” for Biodiversity
The idea that we are approaching a state of shift in the planet’s environment, due to various human activities within the “Anthropocene” (Barnosky et al. 2012) is attracting attention in the scientific community . The definition and quantification of “planetary boundaries ” is one approach to respond to this. “Planetary boundaries” (see Rockström et al. 2009; Steffen et al. 2015) refer to the idea of a “safe operating space” for humanity. The planetary boundaries framework considers processes relating to climate change , biodiversity loss, land-system change, biogeochemical flows, stratospheric ozone depletion, ocean acidification, freshwater use, atmospheric aerosol loading, and chemical pollution. The rationale is that exceeding the identified boundaries means that thresholds and undesirable changes threaten human well-being.
There has been much debate about how to define a meaningful boundary related to biodiversity . The current rate of extinctions and the corresponding biodiversity crisis suggest a possible focus on global extinction rates. However, recent work has focused more on phylogenetic and functional diversity (Faith et al. 2010; Mace et al. 2014; Steffen et al. 2015). These aspects may have a good regional -to-global scope, and appealing links to current and future well-being. These two key aspects for a biodiversity boundary are now being investigated through a global change international program called “Future Earth”. The PD calculus may provide ways to describe boundaries related to phylogenetic diversity “tipping points ” (Faith et al. 2010). Such phylogenetic tipping points correspond to the irreversible loss of deep branches of the tree of life, following successive losses over time of descendent taxa. The tipping points, and corresponding boundaries, then link naturally to concerns about the loss of evolutionary or evosystem services, including option values (unanticipated future benefits for humans) and evolutionary potential . Such option values of biodiversity typically reflect global-scale benefits for future generations, and so they are a natural consideration for planetary boundaries . At the same time, phylogenetic diversity has local importance (e.g. for resilience and delivery of evosystem services) and may be part of regional-scale planning. Early warnings with respect to a phylogenetic planetary boundary may focus on the changing status of Phylogenetic Key Biodiversity Areas – those places on the planet that are outstanding in their current contribution to retaining global phylogenetic diversity (Brooks et al. 2015; Faith chapter “The Value of Phylogenetic Diversity ”).
The interest in Planetary Boundaries also reminds us that there are “boundaries” in the utility of phylogeny for conservation. The PD measure (Faith 1992; Faith chapter “The Value of Phylogenetic Diversity ”) is useful, but does not tell us all we need to know about functional traits – one of the other possible foci for a biodiversity boundary. Functional traits , by their nature, are not always well accounted for by the PD assumption that shared ancestry explains shared features. This assumption could be especially hard to justify if these traits are defined too intrinsically and are therefore not heritable (Grandcolas et al. 2010; Weiher et al. 2011). Therefore, an alternative model assuming that shared habitat explains shared traits may be useful. Such companion models to phylogenetic diversity are in development (Faith chapter “Using Phylogenetic Dissimilarities Among Sites for Biodiversity Assessments and Conservation ”). At the global scale , such approaches could provide, for multiple taxonomic groups, a running report card on risk of loss of functional trait diversity. This would nicely complement the emerging use of a PD report card to assess risks associated with resilience-loss, tipping points and planetary boundaries .
These issues highlight the broader need to integrate phylogenetic diversity – and its associated option values – into the broader perspectives on sustainability and multiple needs of society. This book demonstrates that effective development of the measures – the toolbox – enables phylogenetic diversity to be “on the table” in these policy contexts.
References
Barnosky AD, Hadly EA, Bascompte J et al (2012) Approaching a state shift in Earth’s biosphere. Nature 486(7401):52–58. doi:10.1038/Nature11018
Brooks TM, Cuttelod A, Faith DP et al (2015) Why and how might genetic and phylogenetic diversity be reflected in the identification of key biodiversity areas? Phil Trans R Soc Lond B 370(1662). Unsp 20140019 doi:10.1098/Rstb.2014.0019
Buerki S, Callmander MW, Bachman S et al (2015) Incorporating evolutionary history into conservation planning in biodiversity hotspots. Phil Trans R Soc B 370(1662). Artn 20140014 doi:10.1098/Rstb.2014.0014
Cadotte MW, Dinnage R, Tilman D (2012) Phylogenetic diversity promotes evosystem stability. Ecology 93(8):S223–S233
DeSalle R, Egan MG, Siddall M (2005) The unholy trinity: taxonomy, species delimitation and DNA barcoding. Phil Trans R Soc Lond B 360(1462):1905–1916
Faith DP (1992) Conservation evaluation and phylogenetic diversity. Biol Conserv 61:1–10
Faith DP (2015) Phylogenetic diversity and extinction: avoiding tipping points and worst-case losses from the tree of life. Phil Trans R Soc Lond B 370:20140011. doi:10.1098/rstb.2014.0011
Faith DP, Lozupone C, Nipperess D, Knight R (2009) The cladistic basis for the phylogenetic diversity (PD) measure links evolutionary features to environmental gradients and supports broad applications of microbial ecology’s “phylogenetic beta diversity” framework. Int J Mol Sci 10(11):4723–4741
Faith DP, Magallón S, Hendry AP et al (2010) Evosystem services: an evolutionary perspective on the links between biodiversity and human well-being. Curr Opin Environ Sustain 2:66–74
Faith DP, Richards Z (2012) Climate change impacts on the tree of life: changes in phylogenetic diversity illustrated for Acropora corals. Biology 1:906–932
Forest F, Grenyer R, Rouget M et al (2007) Preserving the evolutionary potential of floras in biodiversity hotspots. Nature 455:757–760
Funk VA, Sakai AK, Richardson K (2002) Biodiversity: the interface between systematics and conservation. Syst Biol 51(2):235–237
German National Academy of Sciences Leopoldina (2014) Challenges and opportunities of integrative taxonomy for research and society – taxonomic research in the era of OMICS technologies. Deutsche Akademie der Naturforscher Leopoldina e.V. Nationale Akademie der Wissenschaften (German National Academy of Sciences), Halle, 54 pages
Goldstein PZ, Desalle R, Amato G, Vogler AP (2000) Conservation genetics at the species boundary. Conserv Biol 14(1):120–131. doi:10.1046/j.1523-1739.2000.98122.x
Grandcolas P, Nattier R, Legendre F, Pellens R (2010) Mapping extrinsic traits such as extinction risks or modelled bioclimatic niches on phylogenies: does it make sense at all? Cladistics 26:1–5
Grandcolas P, Daubin V, Chave J et al (2013) Systématique, Phylogénie. In: Thiébault S, Hadi H (eds) Prospective de l’Institut Ecologie Environnement du CNRS. Compte-Rendu des Journées des 24 et 25 Octobre 2012, Avignon. Les Cahiers Prospectives. Hors série. CNRS, Paris, pp 75–78 [http://www.cnrs.fr/inee/outils/docs/prospectiveavignon2012.pdf]
Hartmann K, André J (2013) Should evolutionary history guide conservation? Biodivers Conserv 22(2):449–458. doi:10.1007/S10531-012-0422-Z
Kukkala AS, Moilanen A (2013) Core concepts of spatial prioritisation in systematic conservation planning. Biol Rev 88(2):443–464. doi:10.1111/Brv.12008
Lozupone C, Knight R (2005) UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 71(12):8228–8235
Lozupone C, Knight R (2008) Species divergence and the measurement of microbial diversity. FEMS Microbiol Rev 32(4):557–578
Mace GM (2004) The role of taxonomy in species conservation. Phil Trans R Soc Lond B 359:711–719
Mace GM, Gittleman JL, Purvis A (2003) Preserving the tree of life. Science 300:1707–1709
Mace GM, Reyers B, Alkemade R et al (2014) Approaches to defining a planetary boundary for biodiversity. Glob Environ Chang 28:289–297. doi:10.1016/J.Gloenvcha.2014.07.009
Moilanen A, Arponen A (2011) Setting conservation targets under budgetary constraints. Biol Conserv 144(1):650–653. doi:10.1016/J.Biocon.2010.09.006
O’Meara BC (2010) New heuristic methods for joint species delimitation and species tree inference. Syst Biol 59:59–73
Pante E, Schoelinck C, Puillandre N (2015) From integrative taxonomy to species description: one step beyond. Syst Biol 64(1):152–160. doi:10.1093/Sysbio/Syu083
Pollock LJ, Rosauer DF, Thornhill AH et al (2015) Phylogenetic diversity meets conservation policy: small areas are key to preserving eucalypt lineages. Phil Trans R Soc B 370(1662). Unsp 20140007 doi:10.1098/Rstb.2014.0007
Pons J, Barraclough TG, Gomez-Zurita J, Cardoso A, Duran DP, Hazell S, Kamoun S, Sumlin WD, Vogler AP (2006) Sequence-based species delimitation for the DNA taxonomy of undescribed insects. Syst Biol 55(4):595–609. doi:10.1080/10635150600852011
Pugliesi L, Rapini A (2015) Tropical refuges with exceptionally high phylogenetic diversity reveal contrasting phylogenetic structures. Int J Biodivers. http://dx.doi.org/10.1155/2015/758019
Régnier C, Achaz G, Lambert A, Cowie RH, Bouchet P, Fontaine B (2015) Mass extinction in poorly known taxa. Proc Natl Acad Sci USA 112(25):7761–7766. doi:10.1073/pnas.1502350112
Rockström J, Steffen W, Noone K, Persson A, Chapin FS, Lambin EF, Lenton TM, Scheffer M, Folke C, Schellnhuber HJ, Nykvist B, de Wit CA, Hughes T, van der Leeuw S, Rodhe H, Sorlin S, Snyder PK, Costanza R, Svedin U, Falkenmark M, Karlberg L, Corell RW, Fabry VJ, Hansen J, Walker B, Liverman D, Richardson K, Crutzen P, Foley JA (2009) A safe operating space for humanity. Nature 461(7263):472–475. doi:10.1038/461472a
Rodrigues AS, Brooks TM, Gaston KJ (2005) Integrating phylogenetic diversity in the selection of priority areas for conservation: does it make a difference? In: Purvis A, Gittleman JL, Brooks T (eds) Phylogeny and conservation. Cambridge University Press, Cambridge, pp 101–119
Srivastava DS, Cadotte MW, MacDonald AAM, Marushia RG, Mirotchnick N (2012) Phylogenetic diversity and the functioning of evosystems. Ecol Lett 15(7):637–648. doi:10.1111/J.1461-0248.2012.01795.X
Steffen W, Richardson K, Rockström J et al (2015) Planetary boundaries: guiding human development on a changing planet. Science 347(6223):736. doi:10.1126/science.1259855
Tancoigne E, Dubois A (2013) Taxonomy: no decline, but inertia. Cladistics 29(5):567–570. doi:10.1111/cla.12019
Tringe SG, Rubin EM (2005) Metagenomics: DNA sequencing of environmental samples. Nat Rev Genet 6:805–814
Vogler AP, Monaghan MT (2006) Recent advances in DNA taxonomy. J Zool Syst Evol Res 45(1):1–10. doi:10.1111/j.1439-0469.2006.00384.x
Weiher E, Freund D, Bunton T et al (2011) Advances, challenges and a developing synthesis of ecological community assembly theory. Phil Trans R Soc B 366:2403–2413
Will KW, Mishler BD, Wheeler QD (2005) The perils of DNA barcoding and the need for integrative taxonomy. Syst Biol 54(5):844–851. doi:10.1080/10635150500354878
Wilson EO (2003) The encyclopedia of life. Trends Ecol Evol 18(2):77–80. doi:10.1016/s0169-5347(02)00040-x
Yahara T, Donoghue M, Zardoya R et al (2010) Genetic diversity assessments in the century of genome science. Curr Opin Environ Sustain 2:43–49, http://dx.doi.org/10.1016/j.cosust.2010.03.008
Acknowledgements
We thank Agathe Haevermans for kindly providing the drawings for the two figures of this chapter.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is distributed under the terms of the Creative Commons Attribution-Noncommercial 2.5 License (http://creativecommons.org/licenses/by-nc/2.5/) which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
The images or other third party material in this chapter are included in the work’s Creative Commons license, unless indicated otherwise in the credit line; if such material is not included in the work’s Creative Commons license and the respective action is not permitted by statutory regulation, users will need to obtain permission from the license holder to duplicate, adapt or reproduce the material.
Copyright information
© 2016 The Author(s)
About this chapter
Cite this chapter
Pellens, R., Faith, D.P., Grandcolas, P. (2016). The Future of Phylogenetic Systematics in Conservation Biology: Linking Biodiversity and Society. In: Pellens, R., Grandcolas, P. (eds) Biodiversity Conservation and Phylogenetic Systematics. Topics in Biodiversity and Conservation, vol 14. Springer, Cham. https://doi.org/10.1007/978-3-319-22461-9_19
Download citation
DOI: https://doi.org/10.1007/978-3-319-22461-9_19
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-22460-2
Online ISBN: 978-3-319-22461-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)