Abstract
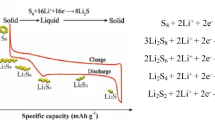
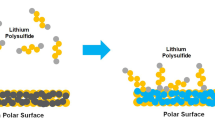
Alkali metal-sulfur batteries have developed profound interest among researchers due to their high theoretical energy density (>2500 Wh kg−1). However, their commercial exploitation potential is hindered due to the lower electronic conductivity of sulfur and other discharging products, deteriorating polysulfide shuttling, and substantial volume variations during discharging. Pseudocapacitive materials can address these issues. Their superior intrinsic properties, such as higher electronic conductivity, better ionic conduction, and abundant functional groups that, trap polysulfides and make them suitable candidates for batteries. Pseudocapacitive materials such as layered transition metal oxides (TMOs), phosphides (TMPs), chalcogenides (TMCs), MXenes, etc. have the metal center acting as Lewis’s entity to attract polysulfide anions. In contrast, the anionic species, such as oxides/phosphides and chalcogens, react with metal ions (Li, Na, and K) to provide the synergetic effect. This chapter discusses the benefits of using these materials for metal-sulfur batteries and associated challenges in a detailed manner.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
X. Wang, H.M. Kim, Y. Xiao, Y.K. Sun, Nanostructured metal phosphide-based materials for electrochemical energy storage. J. Mater. Chem. A 4, 14915–14931 (2016). https://doi.org/10.1039/c6ta06705k
K. Kisu, S. Kim, R. Yoshida, H. Oguchi, N. Toyama, S. Orimo, Microstructural analyses of all-solid-state Li–S batteries using LiBH4-based solid electrolyte for prolonged cycle performance. J. Energy Chem. 50, 424–429 (2020). https://doi.org/10.1016/j.jechem.2020.03.069
H. Fan, W. Luo, S. Dou, Z. Zheng, Advanced two-dimensional materials toward polysulfides regulation of metal–sulfur batteries. SmartMat 1–30 (2023). https://doi.org/10.1002/smm2.1186
M. Salama, A.R. Rosy, R. Yemini, Y. Gofer, D. Aurbach, M. Noked, Metal-sulfur batteries: overview and research methods. ACS Energy Lett. 4, 436–446 (2019). https://doi.org/10.1021/acsenergylett.8b02212
K. Fu, Y. Gong, G.T. Hitz, D.W. McOwen, Y. Li, S. Xu, Y. Wen, L. Zhang, C. Wang, G. Pastel, J. Dai, B. Liu, H. Xie, Y. Yao, E.D. Wachsman, L. Hu, Three-dimensional bilayer garnet solid electrolyte based high energy density lithium metal-sulfur batteries. Energy Environ. Sci. 10, 1568–1575 (2017). https://doi.org/10.1039/c7ee01004d
D. Sui, L. Si, C. Li, Y. Yang, Y. Zhang, W. Yan, A comprehensive review of graphene-based anode materials for lithium-ion capacitors. Chemistry (Easton) 3, 1215–1246 (2021). https://doi.org/10.3390/chemistry3040089
Q. Liu, X. Han, Q. Dou, P. Xiong, Y. Kang, S.-W. Kang, B.-K. Kim, H.S. Park, Multiphase and multicomponent nickel-iron oxide heterostructure as an efficient separator modification layer for advanced lithium sulfur batteries. Batter. Supercaps 4, 1843–1849 (2021). https://doi.org/10.1002/batt.202100156
R. Vishnoi, K. Sharma, Y.S. Yogita, R. Singhal, Investigation of sequential thermal annealing effect on Cu-C70 nanocomposite thin film. Thin Solid Films 680, 75–80 (2019). https://doi.org/10.1016/j.tsf.2019.04.004
V. Marangon, E. Scaduti, V.F. Vinci, J. Hassoun, Scalable composites benefiting from transition-metal oxides as cathode materials for efficient lithium-sulfur batteries. ChemElectroChem 9, e202200374 (2022). https://doi.org/10.1002/celc.202200374
J. Xie, X. Luo, L. Chen, X. Gong, L. Zhang, J. Tian, ZIF-8 derived boron, nitrogen co-doped porous carbon as metal-free peroxymonosulfate activator for tetracycline hydrochloride degradation: performance, mechanism and biotoxicity. Chem. Eng. J. 440, 135760 (2022). https://doi.org/10.1016/j.cej.2022.135760
X. Kang, Z. Jin, H. Peng, Z. Cheng, L. Liu, X. Li, L. Xie, J. Zhang, Y. Dong, The role of selenium vacancies functionalized mediator of bimetal (Co, Fe) selenide for high-energy–density lithium-sulfur batteries. J. Colloid Interface Sci. 637, 161–172 (2023). https://doi.org/10.1016/j.jcis.2023.01.090
P. Han, S.H. Chung, A. Manthiram, Designing a high-loading sulfur cathode with a mixed ionic-electronic conducting polymer for electrochemically stable lithium-sulfur batteries. Energy Storage Mater. 17, 317–324 (2019). https://doi.org/10.1016/j.ensm.2018.11.002
J. Wen, L. Huang, Y. Huang, W. Luo, H. Huo, Z. Wang, X. Zheng, Z. Wen, Y. Huang, A lithium-MXene composite anode with high specific capacity and low interfacial resistance for solid-state batteries. Energy Storage Mater. 45, 934–940 (2022). https://doi.org/10.1016/j.ensm.2021.12.033
Y. Dahiya, M. Hariram, M. Kumar, A. Jain, D. Sarkar, Modified transition metal chalcogenides for high performance supercapacitors: current trends and emerging opportunities. Coord. Chem. Rev. 451, 214265 (2022). https://doi.org/10.1016/j.ccr.2021.214265
Y.H. Liu, C.Y. Wang, S.L. Yang, F.F. Cao, H. Ye, 3D MXene architectures as sulfur hosts for high-performance lithium-sulfur batteries. J. Energy Chem. 66, 429–439 (2022). https://doi.org/10.1016/j.jechem.2021.08.040
Y. Xu, L. Wang, Q. Xu, L. Liu, X. Fang, C. Shi, B. Ye, L. Chen, W. Peng, Z. Liu, W. Chen, 3D hybrids based on WS2/N, S co-doped reduced graphene oxide: facile fabrication and superior performance in supercapacitors. Appl. Surf. Sci. 480, 1126–1135 (2019). https://doi.org/10.1016/j.apsusc.2019.02.217
C. Ye, J. Shan, H. Li, C.-C. Kao, Q. Gu, S.-Z. Qiao, Reducing overpotential of solid-state sulfide conversion in potassium-sulfur batteries. Angew Chem. Int. Ed. 62, e202301681 (2023). https://doi.org/10.1002/anie.202301681
R. Gao, Z. Wang, S. Liu, G. Shao, X. Gao, Metal phosphides and borides as the catalytic host of sulfur cathode for lithium–sulfur batteries. Int. J. Miner. Metall. Mater. 29, 990–1002 (2022). https://doi.org/10.1007/s12613-022-2451-2
Z. Yang, C. Xu, M. Xia, X. Zhang, H. Yan, H. Yu, T. Sun, L. Zhang, F. Hu, J. Shu, Thermodynamic analysis and perspective of aqueous metal-sulfur batteries. Mater. Today 49, 184–200 (2021). https://doi.org/10.1016/j.mattod.2021.01.012
G. Liu, Q. Sun, Q. Li, J. Zhang, J. Ming, Electrolyte issues in lithium-sulfur batteries: development, prospect, and challenges. Energy Fuels 35, 10405–10427 (2021). https://doi.org/10.1021/acs.energyfuels.1c00990
M.K. Aslam, I.D. Seymour, N. Katyal, S. Li, T. Yang, S. Bao, G. Henkelman, M. Xu, Metal chalcogenide hollow polar bipyramid prisms as efficient sulfur hosts for Na-S batteries. Nat. Commun. 1–11. https://doi.org/10.1038/s41467-020-19078-0
H. Yang, B. Zhang, Y.X. Wang, K. Konstantinov, H.K. Liu, S.X. Dou, Alkali-metal sulfide as cathodes toward safe and high-capacity metal (M = Li, Na, K) sulfur batteries. Adv. Energy Mater. 10, 1–24 (2020). https://doi.org/10.1002/aenm.202001764
S. Risse, S. Angioletti-Uberti, J. Dzubiella, M. Ballauff, Capacity fading in lithium/sulfur batteries: a linear four-state model. J. Power. Sources 267, 648–654 (2014). https://doi.org/10.1016/j.jpowsour.2014.05.076
S. Bai, X. Liu, K. Zhu, S. Wu, H. Zhou, Metal–organic framework-based separator for lithium–sulfur batteries. Nat. Energy 1, 16094 (2016). https://doi.org/10.1038/nenergy.2016.94
M.J. Klein, A. Dolocan, C. Zu, A. Manthiram, An effective lithium sulfide encapsulation strategy for stable lithium-sulfur batteries. Adv. Energy Mater. 7, 1–9 (2017). https://doi.org/10.1002/aenm.201701122
H. Chu, H. Noh, Y.J. Kim, S. Yuk, J.H. Lee, J. Lee, H. Kwack, Y.K. Kim, D.K. Yang, H.T. Kim, Achieving three-dimensional lithium sulfide growth in lithium-sulfur batteries using high-donor-number anions. Nat. Commun. 10, 1–12 (2019). https://doi.org/10.1038/s41467-018-07975-4
M. Zhu, S. Li, J. Liu, B. Li, Applied Surface Science Promoting polysulfide conversion by V2O3 hollow sphere for enhanced lithium-sulfur battery. Appl. Surf. Sci. 473, 1002–1008 (2019). https://doi.org/10.1016/j.apsusc.2018.12.189
Z. Xu, M. Wu, Z. Chen, C. Chen, J. Yang, T. Feng, E. Paek, D. Mitlin, Direct structure-performance comparison of all-carbon potassium and sodium ion capacitors. Adv. Sci. 6, 1802272 (2019). https://doi.org/10.1002/advs.201802272
P. Rajkumar, K. Diwakar, G. Radhika, K. Krishnaveni, R. Subadevi, M. Sivakumar, Effect of silicon dioxide in sulfur/carbon black composite as a cathode material for lithium sulfur batteries. Vacuum 161, 37–48 (2019). https://doi.org/10.1016/j.vacuum.2018.12.016
Y. Zhang, X. Ge, Q. Kang, Z. Kong, Y. Wang, L. Zhan, Vanadium oxide nanorods embed in porous graphene aerogel as high-efficiency polysulfide-trapping-conversion mediator for high performance lithium-sulfur batteries. Chem. Eng. J. 393, 124570 (2020). https://doi.org/10.1016/j.cej.2020.124570
Y. Li, B. Su, Manganese dioxide nanosheet functionalized sulfur@ PEDOT core–shell nanospheres for advanced lithium–sulfur batteries. J. Mater. Chem. A 4, 9403–9412 (2016). https://doi.org/10.1039/c6ta03211g
Y. Wang, R. Zhang, J. Chen, H. Wu, S. Lu, K. Wang, H. Li, C.J. Harris, K. Xi, R.V. Kumar, S. Ding, Enhancing catalytic activity of titanium oxide in lithium–sulfur batteries by band engineering. Adv. Energy Mater. 9, 1900953 (2019). https://doi.org/10.1002/aenm.201900953
X. Song, T. Gao, S. Wang, Y. Bao, G. Chen, L.-X. Ding, H. Wang, Free-standing sulfur host based on titanium-dioxide-modified porous-carbon nanofibers for lithium-sulfur batteries. J. Power Sources 356, 172–180 (2017). https://doi.org/10.1016/j.jpowsour.2017.04.093
X. Lang, X. Wang, Y. Liu, K. Cai, L. Li, Q. Zhang, Cobalt-based metal organic framework (Co-MOFs)/graphene oxide composites as high-performance anode active materials for lithium-ion batteries. Int. J. Energy Res. 45, 4811–4820 (2021). https://doi.org/10.1002/er.6080
Q. Sun, B. Xi, J.Y. Li, H. Mao, X. Ma, J. Liang, J. Feng, S. Xiong, Nitrogen-doped graphene-supported mixed transition-metal oxide porous particles to confine polysulfides for lithium-sulfur batteries. Adv. Energy Mater. 8, 1–10 (2018). https://doi.org/10.1002/aenm.201800595
X. Jia, B. Liu, J. Liu, S. Zhang, Z. Sun, X. He, H. Li, G. Wang, H. Chang, Fabrication of NiO-carbon nanotube/sulfur composites for lithium-sulfur battery application. RSC Adv. 11, 10753–10759 (2021). https://doi.org/10.1039/d1ra00216c
X.Y. Yu, X.W. (David) Lou, Mixed metal sulfides for electrochemical energy storage and conversion. Adv. Energy Mater. 8, 1–37 (2018). https://doi.org/10.1002/aenm.201701592
Z. Ali, T. Zhang, M. Asif, L. Zhao, Y. Yu, Y. Hou, Transition metal chalcogenide anodes for sodium storage. Mater. Today 35, 131–167 (2020). https://doi.org/10.1016/j.mattod.2019.11.008
C. Wang, L. Sun, K. Li, Z. Wu, F. Zhang, L. Wang, Unravel the catalytic effect of two-dimensional metal sulfides on polysulfide conversions for lithium-sulfur batteries. ACS Appl. Mater. Interfaces 12, 43560–43567 (2020). https://doi.org/10.1021/acsami.0c09567
P. Wang, B. Xi, M. Huang, W. Chen, J. Feng, S. Xiong, Emerging catalysts to promote kinetics of lithium–sulfur batteries. Adv. Energy Mater. 11, 2002893 (2021). https://doi.org/10.1002/aenm.202002893
B. Qin, Q. Wang, W. Yao, Y. Cai, Y. Chen, P. Wang, Y. Zou, X. Zheng, J. Cao, J. Qi, W. Cai, Heterostructured Mn3O4-MnS multi-shelled hollow spheres for enhanced polysulfide regulation in lithium-sulfur batteries. Energy Environ. Mater. 1–9 (2022). https://doi.org/10.1002/eem2.12475
G. Liu, Q. Zeng, X. Sui, S. Tian, X. Li, Q. Wu, X. Wang, K. Tao, E. Xie, Z. Zhang, Modulating the d-p orbital coupling of manganese chalcogenides for efficient polysulfides conversion in lithium–sulfur batteries. J. Power. Sources 552, 232244 (2022). https://doi.org/10.1016/j.jpowsour.2022.232244
C. Yang, Y. Li, W. Peng, F. Zhang, X. Fan, In situ N-doped CoS2 anchored on MXene toward an efficient bifunctional catalyst for enhanced lithium-sulfur batteries. Chem. Eng. J. 427, 131792 (2022). https://doi.org/10.1016/j.cej.2021.131792
H. Xiang, N. Deng, H. Zhao, X. Wang, L. Wei, M. Wang, B. Cheng, W. Kang, A review on electronically conducting polymers for lithium-sulfur battery and lithium-selenium battery: progress and prospects. J. Energy Chem. 58, 523–556 (2021). https://doi.org/10.1016/j.jechem.2020.10.029
B. He, W.C. Li, Z.Y. Chen, L. Shi, Y. Zhang, J.L. Xia, A.H. Lu, Multilevel structured carbon film as cathode host for Li-S batteries with superhigh-areal-capacity. Nano Res. 14, 1273–1279 (2021). https://doi.org/10.1007/s12274-020-3102-4
Y. Liu, X. Que, X. Wu, Q. Yuan, H. Wang, J. Wu, Y. Gui, W. Gan, ZIF-67 derived carbon wrapped discontinuous CoxP nanotube as anode material in high-performance Li-ion battery. Mater. Today Chem. 17, 100284 (2020). https://doi.org/10.1016/j.mtchem.2020.100284
R. Reinhold, U. Stoeck, H.J. Grafe, D. Mikhailova, T. Jaumann, S. Oswald, S. Kaskel, L. Giebeler, Surface and electrochemical studies on silicon diphosphide as easy-to-handle anode material for lithium-based batteries—the phosphorus path. ACS Appl. Mater. Interfaces 10, 7096–7106 (2018). https://doi.org/10.1021/acsami.7b18697
Z. Li, P. Li, X. Meng, Z. Lin, R. Wang, The interfacial electronic engineering in binary sulfiphilic cobalt boride heterostructure nanosheets for upgrading energy density and longevity of lithium-sulfur batteries. Adv. Mater. 33, 2102338 (2021). https://doi.org/10.1002/adma.202102338
J. Cheng, D. Zhao, L. Fan, X. Wu, M. Wang, N. Zhang, K. Sun, Ultra-high rate Li-S batteries based on a novel conductive Ni2P yolk-shell material as the host for the S cathode. J. Mater. Chem. A 5, 14519–14524 (2017). https://doi.org/10.1039/c7ta03236f
Y. Chen, W. Zhang, D. Zhou, H. Tian, D. Su, C. Wang, D. Stockdale, F. Kang, B. Li, G. Wang, Co-Fe mixed metal phosphide nanocubes with highly interconnected-pore architecture as an efficient polysulfide mediator for lithium-sulfur batteries. ACS Nano 13, 4731–4741 (2019). https://doi.org/10.1021/acsnano.9b01079
Q. Pang, C.Y. Kwok, D. Kundu, X. Liang, L.F. Nazar, Lightweight metallic MgB2 mediates polysulfide redox and promises high-energy-density lithium-sulfur batteries. Joule 3, 136–148 (2019). https://doi.org/10.1016/j.joule.2018.09.024
Y. Gu, L.Q. Fan, J.L. Huang, C.L. Geng, J.M. Lin, M.L. Huang, Y.F. Huang, J.H. Wu, N-doped reduced graphene oxide decorated NiSe2 nanoparticles for high-performance asymmetric supercapacitors. J. Power. Sources 425, 60–68 (2019). https://doi.org/10.1016/j.jpowsour.2019.03.123
J. Shen, Y. Feng, P. Wang, G. Qiu, L. Zhang, L. Lu, H. Wang, R. Wang, V. Linkov, S. Ji, Conductive sulfur-rich copolymer composites as lithium-sulfur battery electrodes with fast kinetics and a high cycle stability. ACS Sustain. Chem. Eng. 8, 10389–10401 (2020). https://doi.org/10.1021/acssuschemeng.0c01791
Y. Xie, H. Zhao, H. Cheng, C. Hu, W. Fang, J. Fang, J. Xu, Z. Chen, Facile large-scale synthesis of core–shell structured sulfur@polypyrrole composite and its application in lithium–sulfur batteries with high energy density. Appl. Energy 175, 522–528 (2016). https://doi.org/10.1016/j.apenergy.2016.03.085
X. Wang, S. Zhang, H. Zhang, S. Gao, S. Han, Q. Xu, J. Xu, W. Lu, X. Wu, L. Chen, 3D porous spherical sulfur/carbon cathode materials with in situ vapor-phase polymerized polypyrrole coating layer for high-performance lithium-sulfur batteries. ACS Sustain. Chem. Eng. 7, 17491–17499 (2019). https://doi.org/10.1021/acssuschemeng.9b04805
F. Wu, J. Chen, R. Chen, S. Wu, L. Li, S. Chen, T. Zhao, Sulfur/polythiophene with a core/shell structure: synthesis and electrochemical properties of the cathode for rechargeable lithium batteries. J. Phys. Chem. C 115, 6057–6063 (2011). https://doi.org/10.1021/jp1114724
W. Wei, J. Li, Q. Wang, D. Liu, J. Niu, P. Liu, Hierarchically porous SnO2 nanoparticle-anchored polypyrrole nanotubes as a high-efficient sulfur/polysulfide trap for high-performance lithium-sulfur batteries. ACS Appl. Mater. Interfaces 12, 6362–6370 (2020). https://doi.org/10.1021/acsami.9b18426
T. Wang, D. Luo, Y. Zhang, Z. Zhang, J. Wang, G. Cui, X. Wang, A. Yu, Z. Chen, Hierarchically porous Ti3C2 MXene with tunable active edges and unsaturated coordination bonds for superior lithium-sulfur batteries. ACS Nano 15, 19457–19467 (2021). https://doi.org/10.1021/acsnano.1c06213
N. Li, Z. Xu, P. Wang, Z. Zhang, B. Hong, J. Li, Y. Lai, High-rate lithium-sulfur batteries enabled via vanadium nitride nanoparticle/3D porous graphene through regulating the polysulfides transformation. 398, 1–8 (2020). https://doi.org/10.1016/j.cej.2020.125432
Y. Zhang, W. Tang, R. Zhan, H. Liu, H. Chen, J. Yang, M. Xu, An N-doped porous carbon/MXene composite as a sulfur host for lithium-sulfur batteries. Inorg. Chem. Front. 6, 2894–2899 (2019). https://doi.org/10.1039/c9qi00723g
X. Liang, Y. Rangom, C.Y. Kwok, Q. Pang, L.F. Nazar, Interwoven MXene nanosheet/carbon-nanotube composites as Li–S cathode hosts. Adv. Mater. 29, 1–7 (2017). https://doi.org/10.1002/adma.201603040
H. Liu, H. Guo, N. Wu, W. Yao, R. Xue, M. Wang, W. Yang, Rational design of nickel-cobalt selenides derived from multivariate bimetal metal-organic frameworks for high-performance asymmetric supercapacitor. J. Alloys Compd. 156535 (2020). https://doi.org/10.1016/j.jallcom.2020.156535
J. Zhou, R. Li, X. Fan, Y. Chen, R. Han, W. Li, J. Zheng, B. Wang, X. Li, Environmental Science for sulfur storage in fast, long-cycle Li–S batteries. Energy Environ. Sci. 7, 2715–2724 (2014). https://doi.org/10.1039/c4ee01382d
S. Li, Z. Zhao, S. Wang, R. Liang, Z. Li, G. Chen, Graphene-wrapped chromium-MOF (MIL-101)/sulfur composite for performance improvement of high-rate rechargeable Li–S batteries. J. Mater. Chem. A 2, 13509–13512 (2014). https://doi.org/10.1039/c4ta01241k
K. Xi, S. Cao, X. Peng, C. Ducati, R.V. Kumar, A.K. Cheetham, Metal–organic frameworks for lithium sulphur batteries. 49, 2192–94 (2013). https://doi.org/10.1039/c3cc38009b
A. Wei, L. Wang, Z. Li, Metal-organic framework derived binary-metal oxide/MXene composite as sulfur host for high-performance lithium-sulfur batteries. J. Alloys Compd. 899, 163369 (2022)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Dahiya, Y., Agarwal, S., Kumar, M., Sarkar, D., Jain, A. (2024). Pseudocapacitive Materials for Metal-Sulfur Batteries. In: Gupta, R.K. (eds) Pseudocapacitors. Engineering Materials. Springer, Cham. https://doi.org/10.1007/978-3-031-45430-1_18
Download citation
DOI: https://doi.org/10.1007/978-3-031-45430-1_18
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-45429-5
Online ISBN: 978-3-031-45430-1
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)