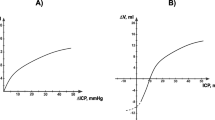
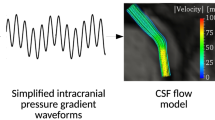
Abstract
This is a review of the physiology of cerebral spinal fluid (CSF), including its purpose and content and the details of CSF production, circulation, and resorption. An introduction to the pathology of the skull base and how it impacts CSF physiology is presented. Also explored are the specifics of intracranial pressure (ICP) and the relationship to skull base pathology. Lastly is a review of approaches to ICP control via modulation of the intracranial constituents of the brain, blood, and CSF.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Rink C, Khanna S. Significance of brain tissue oxygenation and the arachidonic acid cascade in stroke. Antioxid Redox Signal. 2011;14(10):1889–903. https://doi.org/10.1089/ars.2010.3474.
Hofman FM, Chen TC. Chapter 3—Choroid plexus: structure and function, the choroid plexus and cerebrospinal fluid. Cambridge: Academic Press; 2016. p. 29–40, ISBN 9780128017401
Sakka L, Coll G, Chazal J. Anatomy and physiology of cerebrospinal fluid. Eur Ann Otorhinolaryngol Head Neck Dis. 2011;128(6):309–16. https://doi.org/10.1016/j.anorl.2011.03.002. Epub 2011 Nov 18
Tumani H, Huss A, Bachhuber F. The cerebrospinal fluid and barriers—anatomic and physiologic considerations. Handb Clin Neurol. 2017;146:21–32. https://doi.org/10.1016/B978-0-12-804279-3.00002-2.
Oldendorf WH, Hyman S, Braun L, Oldendorf SZ. Blood-brain barrier: penetration of morphine, codeine, heroin, and methadone after carotid injection. Science. 1972;178(4064):984–6. https://doi.org/10.1126/science.178.4064.984.
Nau R, Sörgel F, Prange HW. Lipophilicity at pH 7.4 and molecular size govern the entry of the free serum fraction of drugs into the cerebrospinal fluid in humans with uninflamed meninges. J Neurol Sci. 1994;122(1):61–5. https://doi.org/10.1016/0022-510x(94)90052-3.
Harary M, Dolmans RGF, Gormley WB. Intracranial pressure monitoring-review and avenues for development. Sensors (Basel). 2018;18(2):465. https://doi.org/10.3390/s18020465.
Xu Q, Yu SB, Zheng N, Yuan XY, Chi YY, Liu C, Wang XM, Lin XT, Sui HJ. Head movement, an important contributor to human cerebrospinal fluid circulation. Sci Rep. 2016;19(6):31787. https://doi.org/10.1038/srep31787.
Miller AD, Leslie RA. The area postrema and vomiting. Front Neuroendocrinol. 1994;15(4):301–20. https://doi.org/10.1006/frne.1994.1012.
Sabancı PA, Batay F, Civelek E, Al Mefty O, Husain M, Abdulrauf SI, Karasu A. Meckel’s cave. World Neurosurg. 2011;76(3–4):335–41; discussion 266-7. https://doi.org/10.1016/j.wneu.2011.03.037.
Ahn JH, Cho H, Kim JH, Kim SH, Ham JS, Park I, Suh SH, Hong SP, Song JH, Hong YK, Jeong Y, Park SH, Koh GY. Meningeal lymphatic vessels at the skull base drain cerebrospinal fluid. Nature. 2019;572(7767):62–6. https://doi.org/10.1038/s41586-019-1419-5. Epub 2019 Jul 24
Brierly JB, Field EJ, Yoffey JM. Passage of indian ink particles from the cranial subarachnoid space. J Anat. 1949;83(Pt 1):77.
Silver I, Li B, Szalai J, Johnston M. Relationship between intracranial pressure and cervical lymphatic pressure and flow rates in sheep. Am J Phys. 1999;277(6):R1712–7. https://doi.org/10.1152/ajpregu.1999.277.6.R1712.
Mollanji R, Bozanovic-Sosic R, Zakharov A, Makarian L, Johnston MG. Blocking cerebrospinal fluid absorption through the cribriform plate increases resting intracranial pressure. Am J Physiol Regul Integr Comp Physiol. 2002;282(6):R1593–9. https://doi.org/10.1152/ajpregu.00695.2001.
Földi M, Csillik B, Zoltán OT. Lymphatic drainage of the brain. Experientia. 1968;24(12):1283–7. https://doi.org/10.1007/BF02146675.
Bothwell SW, Janigro D, Patabendige A. Cerebrospinal fluid dynamics and intracranial pressure elevation in neurological diseases. Fluids Barriers CNS. 2019;16(1):9. https://doi.org/10.1186/s12987-019-0129-6.
Xie L, Kang H, Xu Q, Chen MJ, Liao Y, Thiyagarajan M, O'Donnell J, Christensen DJ, Nicholson C, Iliff JJ, Takano T, Deane R, Nedergaard M. Sleep drives metabolite clearance from the adult brain. Science. 2013;342(6156):373–7. https://doi.org/10.1126/science.1241224.
Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA, Benveniste H, Vates GE, Deane R, Goldman SA, Nagelhus EA, Nedergaard M. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid β. Sci Transl Med. 2012;4(147):147ra111. https://doi.org/10.1126/scitranslmed.3003748.
Schwake M, Schipmann S, Müther M, Stögbauer L, Hanning U, Sporns PB, Ewelt C, Dziewas R, Minnerup J, Holling M, Stummer W. Second-look strokectomy of cerebral infarction areas in patients with severe herniation. J Neurosurg. 2019;132(1):1–9. https://doi.org/10.3171/2018.8.JNS18692.
Wu EM, El Ahmadieh TY, Kafka B, Caruso J, Aoun SG, Plitt AR, Neeley O, Olson DM, Ruchinskas RA, Cullum M, Batjer H, White JA. Ventriculoperitoneal shunt outcomes of Normal pressure hydrocephalus: a case series of 116 patients. Cureus. 2019;11(3):e4170. https://doi.org/10.7759/cureus.4170.
Warf BC. Comparison of endoscopic third ventriculostomy alone and combined with choroid plexus cauterization in infants younger than 1 year of age: a prospective study in 550 African children. J Neurosurg. 2005;103(6 Suppl):475–81. https://doi.org/10.3171/ped.2005.103.6.0475.
Golanov EV, Bovshik EI, Wong KK, Pautler RG, Foster CH, Federley RG, Zhang JY, Mancuso J, Wong ST, Britz GW. Subarachnoid hemorrhage - induced block of cerebrospinal fluid flow: role of brain coagulation factor III (tissue factor). J Cereb Blood Flow Metab. 2018;38(5):793–808. https://doi.org/10.1177/0271678X17701157. Epub 2017 Mar 28
Germanwala AV, Huang J, Tamargo RJ. Hydrocephalus after aneurysmal subarachnoid hemorrhage. Neurosurg Clin N Am. 2010;21(2):263–70. https://doi.org/10.1016/j.nec.2009.10.013.
Custodio G, Taques GR, Figueiredo BC, Gugelmin ES, Oliveira Figueiredo MM, Watanabe F, Pontarolo R, Lalli E, Torres LF. Increased incidence of choroid plexus carcinoma due to the germline TP53 R337H mutation in southern Brazil. PLoS One. 2011;6(3):e18015. https://doi.org/10.1371/journal.pone.0018015.
Grygotis LA, Chew FS. Choroid plexus carcinoma of the lateral ventricle. AJR Am J Roentgenol. 1997;169(5):1400. https://doi.org/10.2214/ajr.169.5.9353468.
Kornienko VN. Diagnostic Neuroradiology. In: Igor Nikolaevich Pronin. New York: Springer. ISBN:3540756523.
Kerleroux B, Cottier JP, Janot K, Listrat A, Sirinelli D, Morel B. Posterior fossa tumors in children: radiological tips & tricks in the age of genomic tumor classification and advance MR technology. J Neuroradiol. 2020;47(1):46–53. https://doi.org/10.1016/j.neurad.2019.08.002. Epub 2019 Sep 18
West KW, Turner MK, Vane DW, Boaz J, Kalsbeck J, Grosfeld JL. Ventricular gallbladder shunts: an alternative procedure in hydrocephalus. J Pediatr Surg. 1987;22(7):609–12. https://doi.org/10.1016/s0022-3468(87)80110-0.
Morosanu CO, Filip GA, Nicolae L, Florian IS. From the heart to the bladder-particularities of ventricular shunt topography and the current status of cerebrospinal fluid diversion sites. Neurosurg Rev. 2020;43(3):847–60. https://doi.org/10.1007/s10143-018-1033-2. Epub 2018 Oct 18
Tao C, Fan C, Hu X, Ma J, Ma L, Li H, Liu Y, Sun H, He M, You C. The effect of fenestration of the lamina terminalis on the incidence of shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage (FISH): study protocol for a randomized controlled trial. Medicine (Baltimore). 2016;95(52):e5727. https://doi.org/10.1097/MD.0000000000005727.
Mao J, Zhu Q, Ma Y, Lan Q, Cheng Y, Liu G. Fenestration of lamina terminalis during anterior circulation aneurysm clipping on occurrence of shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage: meta-analysis. World Neurosurg. 2019;129:e1–5. https://doi.org/10.1016/j.wneu.2019.01.270. Epub 2019 Feb 16
Illes S. More than a drainage fluid: the role of CSF in signaling in the brain and other effects on brain tissue. Handb Clin Neurol. 2017;146:33–46. https://doi.org/10.1016/B978-0-12-804279-3.00003-4.
Kato K, Ujiie H, Higa T, Hayashi M, Kubo O, Okada Y, Hori T. Clinical presentation of intracranial epidermoids: a surgical series of 20 initial and four recurred cases. Asian. J Neurosurg. 2010;5(1):32–40.
Kralick F, Oh J, Medina T, Noh HM. Micro-fabricated shunt to mimic arachnoid granulations for the treatment of communicating hydrocephalus. Acta Neurochir Suppl. 2012;114:239–42. https://doi.org/10.1007/978-3-7091-0956-4_47.
Heilman CB, Basil GW, Beneduce BM, Malek AM. Anatomical characterization of the inferior petrosal sinus and adjacent cerebellopontine angle cistern for development of an endovascular transdural cerebrospinal fluid shunt. J Neurointerv Surg. 2019;11(6):598–602. https://doi.org/10.1136/neurintsurg-2018-014445. Epub 2019 Jan 9
Malek A, Heilman C. E-020 design of an endovascular transdural cerebrospinal fluid shunt implant to MIMIC arachnoid granulation function. J NeuroInterventional Surgery. 2018;10:A57.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Davies, J., Paff, M., Chen, J.W., Golshani, K., Hsu, F.P.K. (2023). CSF Physiology and Intracranial Pressure. In: Kuan, E.C., Tajudeen, B.A., Djalilian, H.R., Lin, H.W. (eds) Skull Base Reconstruction . Springer, Cham. https://doi.org/10.1007/978-3-031-27937-9_3
Download citation
DOI: https://doi.org/10.1007/978-3-031-27937-9_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-27936-2
Online ISBN: 978-3-031-27937-9
eBook Packages: MedicineMedicine (R0)