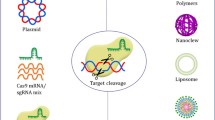
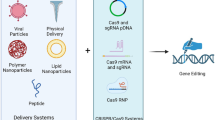
Abstract
CRISPR-Cas9 (clustered, regularly interspaced, short palindromic repeats-associated protein 9) refers to a site-specific method of gene manipulation at the genome level. CRISPR was first discovered in bacteria and archaea as small segments of DNA inserted into CRISPR arrays, which are recognized as an adaptive immune system mechanism to defend against viruses and plasmids. Since the pioneering work on CRISPR sequences in the 1980s, CRISPR-Cas9, which was adapted from the naturally occurring system, has been widely used in gene function research and genetic engineering. CRISPR-based genome editing made a huge leap forward in 2013 when studies on efficient gene manipulation in mammalian cells were reported. CRISPR-Cas9 editing is highly versatile because of its simple operation, specificity, and high efficiency in modifying target genes. Theoretically, any genome sequence can be edited by this system, which has advanced applications ranging from research in basic biology to biotechnology and medicine.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Baliou, S., Adamaki, M., Kyriakopoulos, A. M., Spandidos, D. A., Panayiotidis, M., Christodoulou, I., et al. (2018). CRISPR therapeutic tools for complex genetic disorders and cancer. International Journal of Oncology, 53(2), 443–468.
Barrangou, R., Fremaux, C., Deveau, H., Richards, M., Boyaval, P., Moineau, S., et al. (2007). CRISPR provides acquired resistance against viruses in prokaryotes. Science, 315(5819), 1709–1712.
Boussif, O., Lezoualc’h, F., Zanta, M. A., Mergny, M. D., Scherman, D., Demeneix, B., et al. (1995). A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: Polyethylenimine. Proceedings of the National Academy of Sciences, 92(16), 7297–7301.
Brouns, S. J., Jore, M. M., Lundgren, M., Westra, E. R., Slijkhuis, R. J., Snijders, A. P., et al. (2008). Small CRISPR RNAs guide antiviral defense in prokaryotes. Science, 321(5891), 960–964.
Chang, N., Sun, C., Gao, L., Zhu, D., Xu, X., Zhu, X., et al. (2013). Genome editing with RNA-guided Cas9 nuclease in zebrafish embryos. Cell Research, 23(4), 465–472.
Chen, Z., Liu, F., Chen, Y., Liu, J., Wang, X., Chen, A. T., et al. (2017). Targeted delivery of CRISPR/Cas9-mediated cancer gene therapy via liposome-templated hydrogel nanoparticles. Advanced Functional Materials, 27(46), 1703036.
Chen, G., Ma, B., Wang, Y., & Gong, S. (2018). A universal GSH-responsive nanoplatform for the delivery of DNA, mRNA, and Cas9/sgRNA ribonucleoprotein. ACS Applied Materials & Interfaces, 10(22), 18515–18523.
Chen, G., Abdeen, A. A., Wang, Y., Shahi, P. K., Robertson, S., Xie, R., et al. (2019). A biodegradable nanocapsule delivers a Cas9 ribonucleoprotein complex for in vivo genome editing. Nature Nanotechnology, 14(10), 974–980.
Cheng, W.-J., Chen, L.-C., Ho, H.-O., Lin, H.-L., & Sheu, M.-T. (2018). Stearyl polyethylenimine complexed with plasmids as the core of human serum albumin nanoparticles noncovalently bound to CRISPR/Cas9 plasmids or siRNA for disrupting or silencing PD-L1 expression for immunotherapy. International Journal of Nanomedicine, 13, 7079.
Chou, S.-J., Yang, P., Ban, Q., Yang, Y.-P., Wang, M.-L., Chien, C.-S., et al. (2020). Dual supramolecular nanoparticle vectors enable CRISPR/Cas9-mediated knockin of retinoschisin 1 gene – A potential nonviral therapeutic solution for X-linked juvenile retinoschisis. Advanced Science, 7(10), 1903432.
Cong, L., Ran, F. A., Cox, D., Lin, S., Barretto, R., Habib, N., et al. (2013). Multiplex genome engineering using CRISPR/Cas systems. Science, 339(6121), 819–823.
Deng, H., Huang, W., & Zhang, Z. (2019). Nanotechnology based CRISPR/Cas9 system delivery for genome editing: Progress and prospect. Nano Research, 12(10), 2437–2450.
Deng, S., Li, X., Liu, S., Chen, J., Li, M., Chew, S. Y., et al. (2020). Codelivery of CRISPR-Cas9 and chlorin e6 for spatially controlled tumor-specific gene editing with synergistic drug effects. Science Advances, 6(29), eabb4005.
Ding, F., Huang, X., Gao, X., Xie, M., Pan, G., Li, Q., et al. (2019). A non-cationic nucleic acid nanogel for the delivery of the CRISPR/Cas9 gene editing tool. Nanoscale, 11(37), 17211–17215.
Fu, Y., Foden, J. A., Khayter, C., Maeder, M. L., Reyon, D., Joung, J. K., et al. (2013). High-frequency off-target mutagenesis induced by CRISPR-Cas nucleases in human cells. Nature Biotechnology, 31(9), 822–826.
Gao, Y., Huang, J.-Y., O’Keeffe Ahern, J., Cutlar, L., Zhou, D., Lin, F.-H., et al. (2016). Highly branched poly(β-amino esters) for non-viral gene delivery: High transfection efficiency and low toxicity achieved by increasing molecular weight. Biomacromolecules, 17(11), 3640–3647.
Guan, L., Han, Y., Yang, C., Lu, S., Du, J., Li, H., et al. (2021). CRISPR-Cas9-mediated gene therapy in neurological disorders. Molecular Neurobiology, 59(2), 968–982.
Han, H. A., Pang, J. K. S., & Soh, B.-S. (2020). Mitigating off-target effects in CRISPR/Cas9-mediated in vivo gene editing. Journal of Molecular Medicine, 98(5), 615–632.
He, X.-Y., Liu, B.-Y., Peng, Y., Zhuo, R.-X., & Cheng, S.-X. (2018). Multifunctional vector for delivery of genome editing plasmid targeting β-catenin to remodulate cancer cell properties. ACS Applied Materials & Interfaces, 11(1), 226–237.
He, X., Long, Q., Zeng, Z., Yang, L., Tang, Y., & Feng, X. (2019). Simple and efficient targeted intracellular protein delivery with self-assembled nanovehicles for effective cancer therapy. Advanced Functional Materials, 29(50), 1906187.
Horvath, P., & Barrangou, R. (2010). CRISPR/Cas, the immune system of bacteria and archaea. Science, 327(5962), 167–170.
Hsu, P. D., Scott, D. A., Weinstein, J. A., Ran, F. A., Konermann, S., Agarwala, V., et al. (2013). DNA targeting specificity of RNA-guided Cas9 nucleases. Nature Biotechnology, 31(9), 827–832.
Hughes, T. S., Langer, S. J., Virtanen, S. I., Chavez, R. A., Watkins, L. R., Milligan, E. D., et al. (2009). Immunogenicity of intrathecal plasmid gene delivery: Cytokine release and effects on transgene expression. The Journal of Gene Medicine, 11(9), 782–790.
Iqbal, S., Yasin, M. N., & Sheardown, H. (2019). Engineering of targeted nanoparticles by using self-assembled biointegrated block copolymers. In Surface modification of nanoparticles for targeted drug delivery (pp. 451–466). Springer.
Iqbal, S., Blenner, M., Alexander-Bryant, A., & Larsen, J. (2020). Polymersomes for therapeutic delivery of protein and nucleic acid macromolecules: From design to therapeutic applications. Biomacromolecules, 21(4), 1327–1350.
Ishino, Y., Shinagawa, H., Makino, K., Amemura, M., & Nakata, A. (1987). Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. Journal of Bacteriology, 169(12), 5429–5433.
Jiang, C., Mei, M., Li, B., Zhu, X., Zu, W., Tian, Y., et al. (2017). A non-viral CRISPR/Cas9 delivery system for therapeutically targeting HBV DNA and pcsk9 in vivo. Cell Research, 27(3), 440–443.
Jinek, M., Chylinski, K., Fonfara, I., Hauer, M., Doudna, J. A., & Charpentier, E. (2012). A programmable dual-RNA–guided DNA endonuclease in adaptive bacterial immunity. Science, 337(6096), 816–821.
Jinek, M., East, A., Cheng, A., Lin, S., Ma, E., & Doudna, J. (2013). RNA-programmed genome editing in human cells. elife, 2, e00471.
Jinek, M., Jiang, F., Taylor, D. W., Sternberg, S. H., Kaya, E., Ma, E., et al. (2014). Structures of Cas9 endonucleases reveal RNA-mediated conformational activation. Science, 343, 6176.
Kang, Y. K., Kwon, K., Ryu, J. S., Lee, H. N., Park, C., & Chung, H. J. (2017). Nonviral genome editing based on a polymer-derivatized CRISPR nanocomplex for targeting bacterial pathogens and antibiotic resistance. Bioconjugate Chemistry, 28(4), 957–967.
Kantor, A., McClements, M. E., & MacLaren, R. E. (2020). CRISPR-Cas9 DNA base-editing and prime-editing. International Journal of Molecular Sciences, 21(17), 6240.
Kanduri, V., LaVigne, D., & Larsen, J. (2021). Current advances toward the encapsulation of Cas9. ACS Macro Letters, 10, 1576–1589. https://doi.org/10.1021/acsmacrolett.1c00538
Kelly, J. M., Gross, A. L., Martin, D. R., & Byrne, M. E. (2017). Polyethylene glycol-b-poly (lactic acid) polymersomes as vehicles for enzyme replacement therapy. Nanomedicine, 12(23), 2591–2606.
Kempe, K., & Nicolazzo, J. A. (2021). Biodegradable polymeric nanoparticles for brain-targeted drug delivery. In Nanomedicines for brain drug delivery (pp. 1–27). Springer.
Kim, S., Kim, D., Cho, S. W., Kim, J., & Kim, J.-S. (2014). Highly efficient RNA-guided genome editing in human cells via delivery of purified Cas9 ribonucleoproteins. Genome Research, 24(6), 1012–1019.
Kim, H. J., Ogura, S., Otabe, T., Kamegawa, R., Sato, M., Kataoka, K., et al. (2019). Fine-tuning of hydrophobicity in amphiphilic polyaspartamide derivatives for rapid and transient expression of messenger RNA directed toward genome engineering in brain. ACS Central Science, 5(11), 1866–1875.
Kleinstiver, B. P., Pattanayak, V., Prew, M. S., Tsai, S. Q., Nguyen, N. T., Zheng, Z., et al. (2016). High-fidelity CRISPR–Cas9 nucleases with no detectable genome-wide off-target effects. Nature, 529(7587), 490–495.
Kretzmann, J. A., Ho, D., Evans, C. W., Plani-Lam, J. H. C., Garcia-Bloj, B., Mohamed, A. E., et al. (2017). Synthetically controlling dendrimer flexibility improves delivery of large plasmid DNA. Chemical Science, 8(4), 2923–2930.
L’Amoreaux, N., Ali, A., Iqbal, S., & Larsen, J. (2020). Persistent prolate polymersomes for enhanced co-delivery of hydrophilic and hydrophobic drugs. Nanotechnology, 31(17), 175103.
Lao, Y.-H., Li, M., Gao, M. A., Shao, D., Chi, C.-W., Huang, D., et al. (2018). HPV oncogene manipulation using nonvirally delivered CRISPR/Cas9 or natronobacterium gregoryi argonaute. Advanced Science, 5(7), 1700540.
Lee, K., Conboy, M., Park, H. M., Jiang, F., Kim, H. J., Dewitt, M. A., et al. (2017). Nanoparticle delivery of Cas9 ribonucleoprotein and donor DNA in vivo induces homology-directed DNA repair. Nature Biomedical Engineering, 1(11), 889–901.
Lee, B., Lee, K., Panda, S., Gonzales-Rojas, R., Chong, A., Bugay, V., et al. (2018). Nanoparticle delivery of CRISPR into the brain rescues a mouse model of fragile X syndrome from exaggerated repetitive behaviours. Nature Biomedical Engineering, 2(7), 497–507.
Li, L., Song, L., Liu, X., Yang, X., Li, X., He, T., et al. (2017). Artificial virus delivers CRISPR-Cas9 system for genome editing of cells in mice. ACS Nano, 11(1), 95–111.
Li, L., Yang, Z., Zhu, S., He, L., Fan, W., Tang, W., et al. (2019). A rationally designed semiconducting polymer brush for NIR-II imaging-guided light-triggered remote control of CRISPR/Cas9 genome editing. Advanced Materials, 31(21), 1901187.
Liang, X., Potter, J., Kumar, S., Zou, Y., Quintanilla, R., Sridharan, M., et al. (2015). Rapid and highly efficient mammalian cell engineering via Cas9 protein transfection. Journal of Biotechnology, 208, 44–53.
Liang, C., Li, F., Wang, L., Zhang, Z.-K., Wang, C., He, B., et al. (2017). Tumor cell-targeted delivery of CRISPR/Cas9 by aptamer-functionalized lipopolymer for therapeutic genome editing of VEGFA in osteosarcoma. Biomaterials, 147, 68–85.
Lin, Y.-X., Wang, Y., Blake, S., Yu, M., Mei, L., Wang, H., et al. (2020). RNA nanotechnology-mediated cancer immunotherapy. Theranostics, 10(1), 281.
Lino, C. A., Harper, J. C., Carney, J. P., & Timlin, J. A. (2018). Delivering CRISPR: A review of the challenges and approaches. Drug Delivery, 25(1), 1234–1257.
Liu, B.-Y., He, X.-Y., Xu, C., Xu, L., Ai, S.-L., Cheng, S.-X., et al. (2018a). A dual-targeting delivery system for effective genome editing and in situ detecting related protein expression in edited cells. Biomacromolecules, 19(7), 2957–2968.
Liu, B.-Y., He, X.-Y., Zhuo, R.-X., & Cheng, S.-X. (2018b). Tumor targeted genome editing mediated by a multi-functional gene vector for regulating cell behaviors. Journal of Controlled Release, 291, 90–98.
Liu, Y., Cao, Z.-T., Xu, C.-F., Lu, Z.-D., Luo, Y.-L., & Wang, J. (2018c). Optimization of lipid-assisted nanoparticle for disturbing neutrophils-related inflammation. Biomaterials, 172, 92–104.
Liu, Q., Zhao, K., Wang, C., Zhang, Z., Zheng, C., Zhao, Y., et al. (2019a). Multistage delivery nanoparticle facilitates efficient CRISPR/dCas9 activation and tumor growth suppression in vivo. Advanced Science, 6(1), 1801423.
Liu, C., Wan, T., Wang, H., Zhang, S., Ping, Y., & Cheng, Y. (2019b). A boronic acid–rich dendrimer with robust and unprecedented efficiency for cytosolic protein delivery and CRISPR-Cas9 gene editing. Science Advances, 5(6), eaaw8922.
Liu, Q., Cai, J., Zheng, Y., Tan, Y., Wang, Y., Zhang, Z., et al. (2019c). NanoRNP overcomes tumor heterogeneity in cancer treatment. Nano Letters, 19(11), 7662–7672.
Luo, Y.-L., Xu, C.-F., Li, H.-J., Cao, Z.-T., Liu, J., Wang, J.-L., et al. (2018). Macrophage-specific in vivo gene editing using cationic lipid-assisted polymeric nanoparticles. ACS Nano, 12(2), 994–1005.
Lyu, Y., He, S., Li, J., Jiang, Y., Sun, H., Miao, Y., et al. (2019). A photolabile semiconducting polymer nanotransducer for near-infrared regulation of CRISPR/Cas9 gene editing. Angewandte Chemie, International Edition, 58(50), 18197–18201.
MacLaughlin, F. C., Mumper, R. J., Wang, J., Tagliaferri, J. M., Gill, I., Hinchcliffe, M., et al. (1998). Chitosan and depolymerized chitosan oligomers as condensing carriers for in vivo plasmid delivery. Journal of Controlled Release, 56(1–3), 259–272.
Makarova, K. S., Haft, D. H., Barrangou, R., Brouns, S. J., Charpentier, E., Horvath, P., et al. (2011). Evolution and classification of the CRISPR–Cas systems. Nature Reviews. Microbiology, 9(6), 467–477.
Mali, P., Yang, L., Esvelt, K. M., Aach, J., Guell, M., DiCarlo, J. E., et al. (2013). RNA-guided human genome engineering via Cas9. Science, 339(6121), 823–826.
Misra, M. K., Damotte, V., & Hollenbach, J. A. (2018). The immunogenetics of neurological disease. Immunology, 153(4), 399–414.
Monteys, A. M., Ebanks, S. A., Keiser, M. S., & Davidson, B. L. (2017). CRISPR/Cas9 editing of the mutant huntingtin allele in vitro and in vivo. Molecular Therapy, 25(1), 12–23.
Naeem, M., Majeed, S., Hoque, M. Z., & Ahmad, I. (2020). Latest developed strategies to minimize the off-target effects in CRISPR-Cas-mediated genome editing. Cell, 9(7), 1608.
Niu, Y., Shen, B., Cui, Y., Chen, Y., Wang, J., Wang, L., et al. (2014). Generation of gene-modified cynomolgus monkey via Cas9/RNA-mediated gene targeting in one-cell embryos. Cell, 156(4), 836–843.
Elzoghby, A. O., Abd-Elwakil, M. M., Abd-Elsalam, K., Elsayed, M. T., Hashem, Y., & Mohamed, O. (2016). Natural polymeric nanoparticles for brain-targeting: Implications on drug and gene delivery. Current Pharmaceutical Design, 22(22), 3305–3323.
Patel, T., Zhou, J., Piepmeier, J. M., & Saltzman, W. M. (2012). Polymeric nanoparticles for drug delivery to the central nervous system. Advanced Drug Delivery Reviews, 64(7), 701–705.
Pena, S. A., Iyengar, R., Eshraghi, R. S., Bencie, N., Mittal, J., Aljohani, A., et al. (2020). Gene therapy for neurological disorders: Challenges and recent advancements. Journal of Drug Targeting, 28(2), 111–128.
Peviani, M., Capasso Palmiero, U., Cecere, F., Milazzo, R., Moscatelli, D., & Biffi, A. (2019). Biodegradable polymeric nanoparticles administered in the cerebrospinal fluid: Brain biodistribution, preferential internalization in microglia and implications for cell-selective drug release. Biomaterials, 209, 25–40.
Qi, Y., Song, H., Xiao, H., Cheng, G., Yu, B., & Xu, F.-J. (2018). Fluorinated acid-labile branched hydroxyl-rich nanosystems for flexible and robust delivery of plasmids. Small, 14(42), 1803061.
Qiao, J., Sun, W., Lin, S., Jin, R., Ma, L., & Liu, Y. (2019). Cytosolic delivery of CRISPR/Cas9 ribonucleoproteins for genome editing using chitosan-coated red fluorescent protein. Chemical Communications, 55(32), 4707–4710.
Ran, F. A., Cong, L., Yan, W. X., Scott, D. A., Gootenberg, J. S., Kriz, A. J., et al. (2015). In vivo genome editing using Staphylococcus aureus Cas9. Nature, 520(7546), 186–191.
Rui, Y., Wilson, D. R., Sanders, K., & Green, J. J. (2019a). Reducible branched Ester-amine quadpolymers (rBEAQs) codelivering plasmid DNA and RNA oligonucleotides enable CRISPR/Cas9 genome editing. ACS Applied Materials & Interfaces, 11(11), 10472–10480.
Rui, Y., Wilson, D. R., Choi, J., Varanasi, M., Sanders, K., Karlsson, J., et al. (2019b). Carboxylated branched poly (β-amino ester) nanoparticles enable robust cytosolic protein delivery and CRISPR-Cas9 gene editing. Science Advances, 5(12), eaay3255.
Rui, Y., Wilson, D. R., & Green, J. J. (2019c). Non-viral delivery to enable genome editing. Trends in Biotechnology, 37(3), 281–293.
Rui, Y., Varanasi, M., Mendes, S., Yamagata, H. M., Wilson, D. R., & Green, J. J. (2020). Poly (Beta-Amino Ester) nanoparticles enable nonviral delivery of CRISPR-Cas9 plasmids for gene knockout and gene deletion. Molecular Therapy--Nucleic Acids, 20, 661–672.
Ryu, N., Kim, M.-A., Park, D., Lee, B., Kim, Y.-R., Kim, K.-H., et al. (2018). Effective PEI-mediated delivery of CRISPR-Cas9 complex for targeted gene therapy. Nanomedicine: Nanotechnology, Biology and Medicine, 14(7), 2095–2102.
Schumann, K., Lin, S., Boyer, E., Simeonov, D. R., Subramaniam, M., Gate, R. E., et al. (2015). Generation of knock-in primary human T cells using Cas9 ribonucleoproteins. Proceedings of the National Academy of Sciences, 112(33), 10437–10442.
Shakeri, S., Ashrafizadeh, M., Zarrabi, A., Roghanian, R., Afshar, E. G., Pardakhty, A., et al. (2020). Multifunctional polymeric nanoplatforms for brain diseases diagnosis, therapy and theranostics. Biomedicine, 8(1), 13.
Slaymaker, I. M., Gao, L., Zetsche, B., Scott, D. A., Yan, W. X., & Zhang, F. (2016). Rationally engineered Cas9 nucleases with improved specificity. Science, 351(6268), 84–88.
Sun, W., Ji, W., Hall, J. M., Hu, Q., Wang, C., Beisel, C. L., et al. (2015). Self-assembled DNA nanoclews for the efficient delivery of CRISPR–Cas9 for genome editing. Angewandte Chemie, 127(41), 12197–12201.
Sun, W., Wang, J., Hu, Q., Zhou, X., Khademhosseini, A., & Gu, Z. (2020). CRISPR-Cas12a delivery by DNA-mediated bioresponsive editing for cholesterol regulation. Science Advances, 6(21), eaba2983.
Sung, Y. K., & Kim, S. (2019). Recent advances in the development of gene delivery systems. Biomaterials Research, 23(1), 1–7.
Swiech, L., Heidenreich, M., Banerjee, A., Habib, N., Li, Y., Trombetta, J., et al. (2015). In vivo interrogation of gene function in the mammalian brain using CRISPR-Cas9. Nature Biotechnology, 33(1), 102–106.
Taharabaru, T., Yokoyama, R., Higashi, T., Mohammed, A. F. A., Inoue, M., Maeda, Y., et al. (2020). Genome editing in a wide area of the brain using dendrimer-based ternary polyplexes of Cas9 ribonucleoprotein. ACS Applied Materials & Interfaces, 12(19), 21386–21397.
Tan, Z., Jiang, Y., Ganewatta, M. S., Kumar, R., Keith, A., Twaroski, K., et al. (2019). Block polymer micelles enable CRISPR/Cas9 ribonucleoprotein delivery: Physicochemical properties affect packaging mechanisms and gene editing efficiency. Macromolecules, 52(21), 8197–8206.
Travis, J. (2015). Germline editing dominates DNA summit. American Association for the Advancement of Science.
Wan, T., Chen, Y., Pan, Q., Xu, X., Kang, Y., Gao, X., et al. (2020). Genome editing of mutant KRAS through supramolecular polymer-mediated delivery of Cas9 ribonucleoprotein for colorectal cancer therapy. Journal of Controlled Release, 322, 236–247.
Wang, M., Liu, H., Li, L., & Cheng, Y. (2014). A fluorinated dendrimer achieves excellent gene transfection efficacy at extremely low nitrogen to phosphorus ratios. Nature Communications, 5(1), 1–8.
Wang, D., Mou, H., Li, S., Li, Y., Hough, S., Tran, K., et al. (2015). Adenovirus-mediated somatic genome editing of Pten by CRISPR/Cas9 in mouse liver in spite of Cas9-specific immune responses. Human Gene Therapy, 26(7), 432–442.
Wang, H., La Russa, M., & Qi, L. S. (2016). CRISPR/Cas9 in genome editing and beyond. Annual Review of Biochemistry, 85, 227–264.
Wang, H.-X., Li, M., Lee, C. M., Chakraborty, S., Kim, H.-W., Bao, G., et al. (2017). CRISPR/Cas9-based genome editing for disease modeling and therapy: Challenges and opportunities for nonviral delivery. Chemical Reviews, 117(15), 9874–9906.
Wang, Y., Ma, B., Abdeen, A. A., Chen, G., Xie, R., Saha, K., et al. (2018). Versatile redox-responsive polyplexes for the delivery of plasmid DNA, messenger RNA, and CRISPR-Cas9 genome-editing machinery. ACS Applied Materials & Interfaces, 10(38), 31915–31927.
Wu, S.-S., Li, Q.-C., Yin, C.-Q., Xue, W., & Song, C.-Q. (2020). Advances in CRISPR/Cas-based gene therapy in human genetic diseases. Theranostics, 10(10), 4374.
Xu, C., Lu, Z., Luo, Y., Liu, Y., Cao, Z., Shen, S., et al. (2018). Targeting of NLRP3 inflammasome with gene editing for the amelioration of inflammatory diseases. Nature Communications, 9(1), 1–14.
Xu, C.-F., Chen, G.-J., Luo, Y.-L., Zhang, Y., Zhao, G., Lu, Z.-D., et al. (2021). Rational designs of in vivo CRISPR-Cas delivery systems. Advanced Drug Delivery Reviews, 168, 3–29.
Yan, M., Wen, J., Liang, M., Lu, Y., Kamata, M., & Chen, I. S. (2015). Modulation of gene expression by polymer nanocapsule delivery of DNA cassettes encoding small RNAs. PLoS One, 10(6), e0127986.
Yang, J., Zhang, Q., Chang, H., & Cheng, Y. (2015). Surface-engineered dendrimers in gene delivery. Chemical Reviews, 115(11), 5274–5300.
Yin, H., Song, C.-Q., Dorkin, J. R., Zhu, L. J., Li, Y., Wu, Q., et al. (2016). Therapeutic genome editing by combined viral and non-viral delivery of CRISPR system components in vivo. Nature Biotechnology, 34(3), 328–333.
Yue, H., Zhou, X., Cheng, M., & Xing, D. (2018). Graphene oxide-mediated Cas9/sgRNA delivery for efficient genome editing. Nanoscale, 10(3), 1063–1071.
Zhang, Z., Wan, T., Chen, Y., Chen, Y., Sun, H., Cao, T., et al. (2019a). Cationic polymer-mediated CRISPR/Cas9 plasmid delivery for genome editing. Macromolecular Rapid Communications, 40(5), 1800068.
Zhang, Z., Wang, Q., Liu, Q., Zheng, Y., Zheng, C., Yi, K., et al. (2019b). Dual-locking nanoparticles disrupt the PD-1/PD-L1 pathway for efficient cancer immunotherapy. Advanced Materials, 31(51), 1905751.
Zhang, X., Xu, C., Gao, S., Li, P., Kong, Y., Li, T., et al. (2019c). CRISPR/Cas9 delivery mediated with hydroxyl-rich nanosystems for gene editing in aorta. Advanced Science, 6(12), 1900386.
Zhang, H.-X., Zhang, Y., & Yin, H. (2019d). Genome editing with mRNA encoding ZFN, TALEN, and Cas9. Molecular Therapy, 27(4), 735–746.
Zhang, Y., Shen, S., Zhao, G., Xu, C.-F., Zhang, H.-B., Luo, Y.-L., et al. (2019e). In situ repurposing of dendritic cells with CRISPR/Cas9-based nanomedicine to induce transplant tolerance. Biomaterials, 217, 119302.
Zhang, S., Shen, J., Li, D., & Cheng, Y. (2021). Strategies in the delivery of Cas9 ribonucleoprotein for CRISPR/Cas9 genome editing. Theranostics, 11(2), 614.
Zhu, D., Shen, H., Tan, S., Hu, Z., Wang, L., Yu, L., et al. (2018). Nanoparticles based on poly (β-Amino Ester) and HPV16-targeting CRISPR/shRNA as potential drugs for HPV16-related cervical malignancy. Molecular Therapy, 26(10), 2443–2455.
Declaration of Interests
The authors declare no competing interests.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Iqbal, S., Alexander-Bryant, A., Larsen, J. (2022). Polymer-Mediated Delivery of CRISPR-Cas9 Genome-Editing Therapeutics for CNS Disease. In: Nance, E. (eds) Engineering Biomaterials for Neural Applications. Springer, Cham. https://doi.org/10.1007/978-3-031-11409-0_6
Download citation
DOI: https://doi.org/10.1007/978-3-031-11409-0_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-11408-3
Online ISBN: 978-3-031-11409-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)