Abstract
CAR-T cell treatment comes with significant side effects that challenge the structure and capacity of haematology wards and will regularly necessitate intermittent patient transfer to the ICU. Life-threatening adverse events include cytokine release syndrome and immune effector cell-associated neurotoxicity syndrome, which can occur within hours or days after administration. Sepsis might also require ICU admission within the days that follow CAR-T infusion in these high-risk immunocompromised patients.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
CAR-T cell treatment comes with significant side effects that challenge the structure and capacity of haematology wards and will regularly necessitate intermittent patient transfer to the ICU. Life-threatening adverse events include cytokine release syndrome and immune effector cell-associated neurotoxicity syndrome, which can occur within hours or days after administration. Sepsis might also require ICU admission within the days that follow CAR-T infusion in these high-risk immunocompromised patients.
Critical care and ICU specialists play an important role in the management of patients receiving CAR-T therapies. A substantial number of patients require an ICU bed, and CRS is the leading reason for ICU admission (Fitzgerald et al. 2017; Gutierrez et al. 2020). Prompt and appropriate ICU management relies on a fine-tuned dialogue between haematologists and ICU specialists and on an appropriate definition of the threshold moment to target ICU admission. Hence, less than half of patients require high-dose vasopressors, mechanical ventilation, or renal replacement therapy (Azoulay et al. 2020). However, critical care also benefits those in whom appropriate antibiotics, source control of sepsis, echo-guided fluid expansion, prevention of acute kidney injury, and an optimal oxygenation strategy are provided.
In some patients with comorbidities, the role of ICU specialists starts at the time of patient eligibility for CAR-T cell therapy. Evaluation of patient frailty and risk for developing organ dysfunction and sepsis helps define the optimal timing of ICU admission. When patients are starting lymphodepletion, the ICU specialists at least receive a transmission. Of course, when patients have persistent stage 1 or stage 2 CRS, again, the ICU specialist is alerted. Overall, these careful strategies have allowed a reduction in the need for ICU admission, with the numbers balanced with widespread use of cell therapy and immunotherapy worldwide, which has been helpful in a setting of scarce ICU beds.
To optimally manage CAR-T recipients, haematologists, oncologists, and intensivists need to acquire the necessary knowledge and skills. Transdisciplinary meetings ease harmonization of patient management, keeping all participants aware of the advances in each specialty. Until recently, the ICU has primarily been used as a bridge to cure patients with cancer (Azoulay et al. 2017; Gray et al. 2021). However, CAR-T cell therapy challenges these concepts, producing a time-limited trial that is offered to every CAR-T cell recipient, despite the underlying refractory malignancy, and significant hopes are put towards complete remission or bridging to another promising therapy. However, not all patients respond to treatment with CAR-T cells, and many patients ultimately relapse. Thus, we will need to adapt the approach to admission and discharge from the ICU in a context of uncertainty and with hope for recovery.
The key points below emphasize the role of the ICU specialist throughout the CAR-T cell recipient journey and proposes the importance of maintaining tight collaboration across the involved specialties.
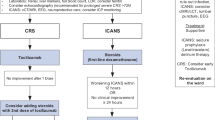
CAR-T cell therapy: Framework to emphasize multidisciplinary collaboration.
Adapted from Azoulay E et al., Am J Respir Crit Care Med. 2019
-
Consultation with an ICU specialist to assess eligibility for CAR-T cell therapy and anticipate post-infusion risks; consultation at the time of lymphodepletion and once any sign of toxicity or sepsis occurs.
-
Apply a common information network to share important information.
-
Reach agreement on the goals of care.
-
Time-limited trials should be considered for all CAR-T cell recipients.
-
CRS and ICANS must be assessed clinically several times per day for at least 7 days.
-
Elicit prompt ICU admission once CRS reaches grade II.
-
Leverage the latest advances.
-
Liaise with all stakeholders to facilitate research.
-
Share experiences with other specialists.
References
Azoulay E, Darmon M, Valade S. Acute life-threatening toxicity from CAR-T cell therapy. Intensive Care Med. 2020;46(9):1723–6. https://doi.org/10.1007/s00134-020-06193-1.
Azoulay E, Schellongowski P, Darmon M, et al. The intensive care medicine research agenda on critically ill oncology and hematology patients. Intensive Care Med. 2017;19 https://doi.org/10.1007/s00134-017-4884-z.
Azoulay E, Shimabukuro-Vornhagen A, Darmon M, von Bergwelt-Baildon M. Critical Care Management of Chimeric Antigen Receptor T Cell-related Toxicity. Be aware and prepared. Am J Respir Crit Care Med. 2019;200(1):20–3. https://doi.org/10.1164/rccm.201810-1945ED.
Fitzgerald JC, Weiss SL, Maude SL, et al. Cytokine release syndrome after chimeric antigen receptor T cell therapy for acute lymphoblastic leukemia. Crit Care Med. 2017;45(2):e124–31. https://doi.org/10.1097/CCM.0000000000002053.
Gray TF, Temel JS, El-Jawahri A. Illness and prognostic understanding in patients with hematologic malignancies. Blood Rev. 2021;45:100692. https://doi.org/10.1016/j.blre.2020.100692.
Gutierrez C, Brown ART, Herr MM, et al. The chimeric antigen receptor-intensive care unit (CAR-ICU) initiative: surveying intensive care unit practices in the management of CAR-T cell associated toxicities. J Crit Care. 2020;58:58–64. https://doi.org/10.1016/j.jcrc.2020.04.008.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2022 The Author(s)
About this chapter
Cite this chapter
Holtick, U., Azoulay, E. (2022). ICU. In: Kröger, N., Gribben, J., Chabannon, C., Yakoub-Agha, I., Einsele, H. (eds) The EBMT/EHA CAR-T Cell Handbook. Springer, Cham. https://doi.org/10.1007/978-3-030-94353-0_31
Download citation
DOI: https://doi.org/10.1007/978-3-030-94353-0_31
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-94352-3
Online ISBN: 978-3-030-94353-0
eBook Packages: MedicineMedicine (R0)