Abstract
Unresectable locally recurrent breast cancers (LRBCs) lead to life-threatening situations and a tremendous loss of quality of life. Most patients have received radiotherapy (RT) during primary treatment and re-irradiation (re-RT) using standard doses might be denied due to the risk of unacceptable cumulative toxicity. Therefore, an effective tumor control is often abandoned.
Mild hyperthermia (39–43 °C), allows for significant reductions in re-RT dose and effective tumor responses with acceptable toxicity even after heavy pretreatment. More than 280 patients have been treated with superficial hyperthermia by water-filtered infrared A (wIRA-HT), which can cover even large lesions. Hyperthermia was immediately followed by hypofractionated re-RT of 5x4 Gy, once a week.
Evaluating 201 patients, overall response rate was 94%. 170 patients with macroscopic LRBC were stratified into 4 size classes (rClasses), and detailed response rates are presented. Accordingly, response rates of 31 patients with microscopic disease after resection of LRBC are communicated.
RT-related acute and late toxicity was limited to grades 1 and 2. Due to this low toxicity, re-recurrences after CR and new local progression after PR could be re-treated using the same treatment schedule. Real-time thermographic and visual controls have allowed to almost exclude the risk of thermal skin damage.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
- Recurrent breast cancer
- Thermotherapy
- wIRA-hyperthermia
- Lymphangitis carcinomatosa
- Carcinoma en cuirasse
- Superficial hyperthermia
- Contact-free hyperthermia
- Re-irradiation
- Hypofractionated X-irradiation
1 Introduction
In 75% of all cases of primary breast cancer, tumor tissue is removed by breast-conserving surgery (BCS) to avoid mastectomy. In standard procedure, BCS is followed by adjuvant radiotherapy (RT) using a total dose of 50–60 Gy in order to eradicate remaining cancer cells and decrease the risk of locoregional recurrence. Improved approaches for the early detection of breast cancer and surgical techniques and RT protocols have decreased the median incidence rate of locoregional recurrence in high-income countries to 0.6% per year. However, the increasing number of longtime breast cancer survivors increases the number of patients at risk of developing a local recurrence later, thereby leading to a median cumulative local recurrence rate of 6.2% [1]. In subgroups of patients, such as triple-negative breast cancer in premenopausal patients, cancer recurrence occurs considerably more often and after a shorter time interval. The first manifestation of locally recurrent breast cancer (LRBC) after BCS is generally treated by mastectomy, in some cases even by a second BCS. The option of a second adjuvant radiotherapy of 50–60 Gy must be weighed against the risk of severe side effects due to cumulative radiotoxicity.
If the recurrent tumor cannot completely be removed by mastectomy with an adequate safety margin or if the pathologist finds cancer cells in the margin of the removed tissue, then the so-called “microscopic disease” carries a high risk of macroscopic recurrence. Furthermore, locoregional recurrences at the chest wall in the pre-irradiated area are often irresectable and/or resection would require complicated surgical procedures that would be associated with major mutilations. Another RT at a standard dosage, which is required for effective tumor control, must be denied due to expected unacceptable toxicity for heart, lung, and rib bones. An inflammatory subgroup of locoregional recurrences, lymphangiosis carcinomatosa, even tends to recur repeatedly after short time gaps. In these final stages of unresectable locally recurrent disease in pre-irradiated sites, the ambition for tumor control is often abandoned, and patients suffer from a tremendous loss of quality of life caused by uncontrolled local tumor growth with symptoms such as pain, constriction, bleeding, ulceration, and infection. These symptoms also often lead to social isolation.
Mild hyperthermia (HT) as a potent radiosensitizer (Thomsen et al., Chap. 5, this book) enables a reduced RT dose and toxicity. In combined HT/RT schedules, good tumor control can be achieved using total re-irradiation doses of 20–39 Gy with very different fractionation schedules. A systematic review and meta-analysis have demonstrated the efficacy of combined HT/RT in the treatment of LRBC, with an especially high benefit for pre-irradiated patients [2]. In most of the published studies, HT was applied by microwave (MW) devices consisting of a microwave antenna and a water bolus, which is directly applied to the target region. Superficial HT using MW can achieve effective hyperthermia temperatures >39 °C up to a depth of approx. 30–40 mm. However, the MW technique has considerable limitations as the treatment of large-sized diffuse tumor spread cannot be covered by one applicator, especially in the case of heterogeneous body contours. With ulcerating lesions, the applicator contact may be uncomfortable and painful. In addition, the incidence of thermal skin damage is high [3].
2 Patients and Treatments
So far, tumor response and local control have been documented in 280 patients presenting with LRBC in pre-irradiated sites and treated with thermography-controlled, superficial wIRA-HT. In 2020, an intermediate analysis of the outcome data of 201 patients was published [4]. Since tumor size has been described as being the most significant prognostic factor [5], the evaluation has been stratified into 4 size classes of macroscopic disease, ranging from tumors <10 cm (rClass I), up to carcinoma en cuirasse with extensions to the back (rClass IV, as shown in Fig. 6.1). In addition, the microscopic disease is classified as rClass 0.
Size classification for locally recurrent breast cancer, as suggested by Notter et al. (modified from [4])
2.1 Basic Characteristics of the Patients
In the intermediate analysis, 170 patients presented with macroscopic disease and 31 patients with microscopic disease. Most patients were heavily pretreated and sent to thermography-controlled wIRA-hyperthermia since no other therapeutic option remained. All patients had previously received radiotherapy, with 116 patients having received more than 2 systemic chemotherapies. Of the patients with macroscopic disease, 141 (83%) had large-sized tumors (rClass II–IV), 49 of whom had 2 or more regions to be treated. Poor prognostic factors were prominent in these patients: 50 patients (25%) presented with triple-negative breast cancer, 115 patients (57%) presented with inflammatory LRBC (lymphangiosis carcinomatosa), and 70 patients (35%) with ulcerating lesions. More than half of the patients with macroscopic disease presented with additional distant metastasis, which is the decisive factor for limited life expectancy. However, effective control of local tumor growth is crucial, even for patients with a life expectancy of just a few months, in order to achieve a satisfying palliative effect, and to improve the quality of life during the remaining lifetime.
2.2 Treatment Schedule
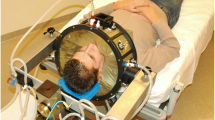
The basic treatment protocol was thermography-controlled, superficial wIRA-hyperthermia directly followed by hypofractionated re-irradiation of 5 × 4 Gy, once a week, as introduced by Notter et al. [6]. The total re-irradiation dose of 20 Gy is the lowest RT dose ever applied in a protocol aiming for effective tumor control. Since wIRA-HT works with a contact-free energy input, the treatment allows the patient to stay in a comfortable position (see Fig. 6.2a–c). Automatic control of the maximum surface temperature between 42 °C and 43 °C achieves effective hyperthermic temperatures >39 °C up to a depth of approximately 25 mm (Thomsen et al.; see Chap. 5) and overheating leading to thermal skin damage can be avoided. The time interval between HT and RT is <5 min for the transport of the patient from HT to the RT room and RT delivery.
3 Results
3.1 Tumor Response and Toxicity
The assessment of a new treatment method is based on the ratio of efficacy and toxicity. High toxicity is especially unacceptable in a therapy with palliative intent. Tumor response rates are shown in Table 6.1. As expected, rates of complete response (CR) decrease with increasing tumor size. Correspondingly, rates of partial response (PR) develop in opposite directions. In patients with large-sized LRBC and distant metastasis, the purely palliative treatment was often deliberately confined to regions where the tumor caused debilitating symptoms. With an overall clinical response (CR + PR) of 95% only very few patients experienced no benefit from the treatment.
As shown in Table 6.2, toxicity is extremely low. All grade I acute side effects did not require any medical intervention and had almost no impact on quality of life. Radiation dermatitis grade II and scurfs only occurred in 14 patients. Thermal skin damage (a rather common side effect in microwave hyperthermia [3]) occurred in only 1 patient. Hyperpigmentation had only a cosmetic impact and was not relevant for these patients.
Figure 6.3 (a–d) show an example of a patient with CR (rClass 2), and Figs. 6.4 and 6.5 (a, b) demonstrate patients with PR (rClasses III and IV)
3.2 Local Control and Re-Recurrence
In the majority of patients, the local tumor remained controlled during lifetime. Some patients with a formerly fatal prognosis of local tumor growth, but without metastasis could even be cured. However, the progressed stage of disease and especially the characteristics of inflammatory lymphangiosis carcinomatosa are associated with a high risk of repeat recurrences and new local progression, respectively, distant metastasis. Local control rates after CR and locally progression-free rates after PR are shown in Tables 6.3 and 6.4.
The extremely low toxicity of combined thermography-controlled wIRA-HT and re-RT with a total dose of 20 Gy allows for repeat re-irradiation(s) in case of repeat recurrence(s). Of the 21 patients presenting with re-recurrences after CR, 19 were treated with the same schedule again. Among these 19 patients, 13 achieved another CR, and 6 patients had a PR. Repeat re-irradiation was also applied in 24 of 40 patients presenting with new local progression after PR. Two patients achieved a CR, and 16 patients achieved a PR. In some patients, a formerly desperate situation could be turned into a sustainable management of tumor control throughout several subsequent re-recurrences with good quality of life for several years. An example of this scenario is shown in Fig. 6.6.
Patient with periodically recurring lymphangiosis carcinomatosa. Before treatment (left column) and after treatment (right column). Schedule of some treatments was interrupted after 2 or 3 therapeutic sessions due to an independently occurring urinary bladder infection and unavailability of the patient
The Kaplan–Meier estimates are commonly used for comparing a new therapeutic method with an existing standard therapy in comparative clinical trials. Unfortunately, the characteristics of unresectable, large-sized LRBC in pre-irradiated regions do not allow for the design of comparative studies since there is no standard therapy available. Therefore, the comparison of a promising therapeutic intervention with a non-treatment-responsive best supportive care strategy cannot be justified ethically [7, 8]. Nevertheless, the Kaplan–Meier estimates can be used to compare overall survival with respect to tumor size and to the presence of metastasis (Fig. 6.7). As expected, overall survival progressively reduces with increasing tumor size (Fig. 6.7a). The high mortality in the first 18 months is primarily caused by death due to metastatic disease. In our analysis, the presence of metastasis has the most significant impact on overall survival. The upper curve in Fig. 6.7b (including all patients) and in Fig. 6.7c (including patients with macroscopic disease) shows a relevant proportion of long-term survivors. By using combined HT/re-RT in the treatment of inoperable LRBC in pre-irradiated areas, a curative intent should not be excluded. Figure 6.7d shows survival of patients with initially microscopic disease, only very few of whom had distant metastasis.
4 Conclusion and Outlook
Combining HT and re-RT at considerably reduced RT doses has shown an excellent efficacy/toxicity ratio in the treatment of unresectable LRBC in pre-irradiated sites and offers a new option for patients in formerly desperate situations of uncontrollable tumor growth. Thermography-controlled superficial wIRA-HT is mainly indicated in the treatment of large-sized lesions, lymphangiosis carcinomatosa, and ulcerated lesions up to a tissue depth of approximately 2.5 cm (Thomsen et al., Chap. 5 in this book). Clinical experiences have shown a comparable efficacy in the treatment of other superficial tumors (e.g., malignant melanoma, skin carcinomas, radiation-induced angiosarcoma, and Merkel cell carcinoma).
In addition to the described combination with radiotherapy, combined treatment schedules including chemotherapy and immunotherapy should be considered in the future.
References
Spronk I, Schellevis FG, Burgers JS, de Bock GH, Korevaar JC. Incidence of isolated local breast cancer recurrence and contralateral breast cancer: a systematic review. Breast. 2018;39:P70–9.
Datta NR, Puric E, Klingbiel D, Gomez S, Bodis S. Hyperthermia and radiation therapy in locoregional recurrent breast cancers: a systematic review and meta-analysis. Int J Radiat Oncol Biol Phys. 2016;94:1073–87.
Bakker A, van der Zee J, van Tienhoven G, Kok HP, Rasch CRN, Crezee H. Temperature and thermal dose during radiotherapy and hyperthermia for recurrent breast cancer are related to clinical outcome and thermal toxicity: a systematic review. Int J Hyperth. 2019;36(1):1024–39.
Notter M, Thomsen AR, Nitsche M, Hermann RM, Wolff H, Habl G, Münch K, Grosu AL, Vaupel P. Combined wIRA-hyperthermia and hypofractionated re-irradiation in the treatment of locally recurrent breast cancer: evaluation of therapeutic outcome based on a novel size classification. Cancers. 2020;12, 606. https://doi.org/10.3390/cancers12030606.
Oldenborg S, Griesdoorn V, Os RV, Kusumanto YH, Oei BS, Venselaar JL, Paul J, Vörding ZVS, Heymans MW, Kolff MW. Reirradiation and hyperthermia for irresectable locoregional recurrent breast cancer in previously irradiated area: size matters. Radiother Oncol. 2015;117:223–8.
Notter M, Piazena H, Vaupel P. Hypofractionated re-irradiation of large-sized recurrent breast cancer with thermography-controlled, contact-free water-filtered infra-red-A hyperthermia: a retrospective study of 73 patients. Int J Hyperth. 2017;33:227–36.
Thomsen AR, Vaupel P, Grosu AL, Notter M. Hyperthermia plus re-irradiation in the management of unresectable locoregional recurrence of breast cancer in previously irradiated sites. J Clin Oncol Sep. 2020;8:JCO2001857. https://doi.org/10.1200/JCO.20.01857.
Buchholz TA, Ali S, Hunt KK. Reply to Thomsen A (2020). J Clin Oncol. 2020;8:JCO2002247. https://doi.org/10.1200/JCO.20.02247.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2022 The Author(s)
About this chapter
Cite this chapter
Notter, M., Thomsen, A.R., Grosu, A.L., Münch, K., Vaupel, P. (2022). Thermography-Controlled, Contact-Free wIRA-Hyperthermia Combined with Hypofractionated Radiotherapy for Large-Sized Lesions of Unresectable, Locally Recurrent Breast Cancer. In: Vaupel, P. (eds) Water-filtered Infrared A (wIRA) Irradiation. Springer, Cham. https://doi.org/10.1007/978-3-030-92880-3_6
Download citation
DOI: https://doi.org/10.1007/978-3-030-92880-3_6
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-92879-7
Online ISBN: 978-3-030-92880-3
eBook Packages: MedicineMedicine (R0)