Abstract
In recent years, the focus of microplastic research has begun to observe a shift from the marine towards terrestrial and freshwater environments. This is in response to a greater awareness of the predominance of land-based sources in marine microplastic contamination. In this regard, terrestrial and freshwater environments are often perceived as conduits for microplastic particles to the oceans, but this overlooks substantial and important complexities associated with these systems, as well as the need to protect these ecosystems in their own right. This chapter focuses on several critical sources and pathways deemed to be highly important for the release of microplastics to the environment. These include road-associated microplastic particles (RAMP) and emissions related to agriculture that are, thus far, under-researched. Transfers and accumulations of particles within terrestrial and freshwater systems are also reviewed, including the state of knowledge on the occurrence of microplastics in different environmental compartments (air, water, sediments, biota). Methodological constraints are addressed, with particular focus on the need for greater harmonisation along all stages of sampling, analysis, and data handling. Finally, the chapter discusses the ultimate fate of particles released to terrestrial and freshwater environments and highlights critical research gaps that should be addressed to evolve our understanding of microplastic contamination in complex and dynamic environmental systems.
You have full access to this open access chapter, Download chapter PDF
Similar content being viewed by others
Keywords
4.1 Introduction
Recent research has begun to document widespread and pervasive contamination of terrestrial and freshwater environmental systems by microplastic particles. Several papers have now pointed out a dichotomy that exists: all plastic is produced on land – and the majority of plastic is consumed and disposed of on land – and yet the primary focus for microplastics research still concentrates predominately on the marine environment (e.g. Blettler et al. 2018; Dris et al. 2015; Horton et al. 2017a, b; Lambert and Wagner 2018; Mai et al. 2018). This is where microplastic contamination was first observed (Carpenter et al. 1972; Carpenter and Smith 1972; Shiber 1979) and is highlighted as the eventual recipient for microplastic particles in the environment. Evidence has shown that microplastics are distributed widely across the global ocean and may have negative impacts on the marine ecosystem , particularly in remote and sensitive regions (Avio et al. 2015). Despite this, a focus on marine microplastics misses several important characteristics of their release and geographical distribution that are integral to efforts to reduce environmental contamination. First and foremost, the majority of microplastic particles are released through land-based sources (Rochman 2018). A thorough assessment of these sources is therefore essential to identify actions to effectively reduce microplastic emissions. This is frequently referred to through the ‘turning off the tap’ analogy (Boucher and Friot 2017; Evans-Pughe 2017); however, this touches upon a second important detail. Many land-based processes, such as fluvial and atmospheric transport, are described as transfers of plastic from land to sea. They should not, however, be considered as pipelines of plastics to the sea: the transport of microplastic particles from their source to the marine environment is expected to be highly complex. Particles released on land likely encounter a range of dynamic environments which can transform particles and may also retain them across a range of timescales, thus acting as a sink of microplastic pollution, and with similar potential impacts as reported for the marine systems. A thorough understanding of these processes is still lacking.
One of the main barriers to establishing this understanding is the paucity of high-quality and comparable data. This is underpinned by methodological complexities associated with the analysis of microplastic contamination in a range of environmental compartments and matrices. Here, a marine-freshwater skew also exists: the majority of harmonisation and standardisation efforts by international bodies and working groups primarily focus on the marine environment (e.g. Frias et al. 2018; GESAMP 2015, 2016, 2019; Isobe et al. 2019). It is important that methods established for the marine environment are not uncritically transferred to terrestrial and freshwater samples. Sampling for microplastics should be closely tied to the specific research questions at hand. There exists a wealth of methodological approaches outside of the plastic research field that may be tailored to include the capture of microplastic particles and which would generate samples that also correspond to a range of relevant hydrological, geomorphological, and aeolian processes. Moreover, in many cases, differences in the type, quantity, and ratios of non-plastic organic and inorganic sample constituents complicate analytical methods and may require the development of new approaches to prepare samples. These new methods require validation to ensure the production of comparable datasets. Recent efforts to crystallise the quality requirements for data reporting have included non-marine sample types (e.g. Koelmans et al. 2019). These describe several good practices which should be applied to all assessments of microplastic contamination: ensuring that sampling is representative, including both blanks and relevant recovery tests, verifying particles as microplastic through chemical analyses, and considering the sources of error in the data.
This chapter will draw together existing research from terrestrial and freshwater environments to address the current state of knowledge and identify important gaps in our understanding of sources and processes related to microplastic contamination across a range of spatial and temporal scales. This will include a review of available data on the occurrence of microplastic particles in selected focused environments: (1) agricultural systems; (2) urban environments, with a particular focus on road-derived microplastics; (3) river systems; (4) lakes; and (5) the atmosphere.
4.2 Microplastics in Terrestrial Environments
The majority of all plastics ever produced – approximately 60% or 4900 Mt. – have been discarded and are now present in either landfills or the environment (Geyer et al. 2017). Establishing the proportions that have been directly (e.g. littering, spills, discharges) or indirectly (e.g. leaching) released to the environment, released to land or the ocean , or released across different spatial and temporal scales is difficult. Plastics used in marine industries (e.g. aquaculture), lost in spills at sea, or directly discarded to the ocean (e.g. littering from ships or at the coast) are likely to represent a small proportion of the total plastics entering the ocean each year. Estimates currently place this at around 5–20%, indicating that the majority of marine plastic waste comes from land-based sources (e.g. Mehlhart and Blepp 2012; Zhou et al. 2011). This chapter addresses microplastic contamination, which is typically associated with more issues due to methodological difficulties, greater heterogeneity, and a lack of clear definitions. The proportion of plastic waste that is released to the environment in the micro-size range is essentially unknown. It is also expected that many plastic items may fragment into micro- or nano-sized particles when exposed to different environmental conditions, but this has not been demonstrated experimentally for many plastic polymer or product types or in a range of relevant environmental settings. Hence, the rates of particle release and associated particle size distributions are not well-understood. The upshot of this is that sources and pathways of microplastic to the terrestrial environment are typically poorly defined.
In recent years, more research has begun to focus on terrestrial environments in regard to microplastic contamination, although the total number of publications remains far below that for the marine environment. This section focuses on agricultural and urban environments, as settings that are likely to be important for the environmental release or impact of microplastic particles. Figure 4.1 presents some of the sources and release pathways of microplastics, as well as key processes associated with their fate and transport. In this chapter, sources refer to direct releases of microplastic to the environment – for example, the production of tyre wear particles during vehicle use – whilst release pathways describe processes or practices that release microplastic to the environment but are not the primary source. Land application of sewage sludge is one example of this, where microplastics in sludge are derived from the culmination of a diverse set of sources that occurs prior to environmental release.
4.2.1 Agriculture
Agricultural environments have recently been identified as recipients of considerable plastic debris, typically concentrated into micro- and nanoplastic size fractions (ECHA 2019). This results from the culmination of a wide range of different sources and release pathways of small plastic particles to farmed soil . These include (1) the application of sewage sludge-derived biosolids on land as a soil conditioner and fertiliser; (2) the release of small plastic fragments from an array of plastic products used in agriculture, termed plasticulture; (3) the use of synthetic polymers in microencapsulation technologies for agrochemicals and seed coatings; (4) the breakdown of plastic litter from roadsides or the farm environment; and (5) the use of water that contains microplastics for irrigation.
Many studies have reported the enrichment of microplastic particles in sewage sludge (e.g. Li et al. 2018; Lusher et al. 2018; Mahon et al. 2017; Xu et al. 2020). Wastewater treatment plants (WWTPs) receive small plastic particles from a diverse range of sources including households, industry, and stormwater. Many of the treatment processes employed to purify the water are also effective at trapping many of these small particles; reported removal efficiencies range between 64.4 (Liu et al. 2019a, b, c) and 99.9% (Magnusson and Norén 2014; Vollertsen and Hansen 2017). Much of the material that is retained in the WWTP is transferred to the solid sludge phase. One technique to handle the generation of this solid by-product from the wastewater treatment process is the application of treated sludge (biosolids) to land to amend soil properties such as pH, soil texture, and nutrient content. This is particularly relevant for agricultural land – for example, 76% of land application of biosolids in Norway is to farmed soils (Lusher et al. 2018). However, this results in the release of microplastic particles to agricultural environments. Estimates indicate that this is responsible for the annual emission of 63,000–430,000 and 44,000–300,000 tonnes of microplastics to European and North American farmlands, respectively (Nizzetto et al. 2016).
Plasticulture is likely to represent an important source of microplastics to agricultural soils. One of their main applications is mulching: thin films are placed above or below the ground to amend soil conditions, improve water use efficiency, and reduce pests and weeds with the aim of increasing crops yield. Further uses of thin films include greenhouse and tunnel systems and as wrappings for hay bales. Additionally, plastic netting may be used in plasticulture systems to protect crops from pests. Microplastic debris may be formed during and after use as a result of environmental conditions and agricultural practices that promote degradation or mechanical fragmentation. Typically, it is difficult to remove 100% of these plastic products from the fields following use (Steinmetz et al. 2016). Geographic regions where high-intensity plasticulture converges with stronger environmental degradative forcing (e.g. higher solar insolation, increased variability between day and night temperature, higher humidity) are likely to represent hotspots for microplastic contamination from this source, such as in the Mediterranean agricultural belt and China (Espí et al. 2006; Liu et al. 2014; Scarascia-Mugnozza et al. 2012). Estimates associated with the release of microplastics from plasticulture are largely missing but may be as high as 24% of the total mass of the product when it is removed from the land (Dong et al. 2013). Future projections for Chinese farmlands estimate that mulching-derived plastic contamination in the soil may reach as high as 2000 kg hm−2 after 141 years of repeated application (Dong et al. 2013).
Advancements in agricultural technologies include the incorporation of synthetic polymers in the encapsulation of seeds and agrochemicals. This utilises polymerisation, coacervation, coating, and micro- and nano-encapsulation technologies to build a polymer matrix or thin coating, which may include non-biodegradable and insoluble plastic polymers such as polyethylene, polystyrene, polyurethane, or polyesters (França et al. 2019). Through this approach, fertiliser products are coated or encapsulated in a polymer shell which regulates the release of the active ingredient over a period of several months through the process of diffusion. Once the fertiliser is entirely released, the shell remains, representing a direct source of small plastic particles to the environment (Sinha et al. 2019). Seeds may also be coated in a polymeric film that incorporates germination-enhancing products such as fungicides or insecticides (Accinelli et al. 2019). The inclusion of these components into a film reduces the dispersion of agrochemicals that may otherwise be applied in powder form during sowing. The use of these technologies is expected to input between 5400 and 39,700 tonnes of microplastic to agricultural environments in the EU each year (ECHA 2019). Despite this, no published study has observed this release under field conditions.
Additional sources and release pathways for microplastic to agricultural environments include potential inputs derived from plastic litter, irrigation systems, and atmospheric deposition. Plastic litter within farms and from the surrounding environment may fragment due to environmental degradation, leading to the release of particles to soils. Atmospheric deposition, through windthrow of particles from adjacent systems or transport from further distances, may also introduce microplastic to farm environments. This is likely to be particularly relevant for low-density polymer types (Rezaei et al. 2019). Finally, irrigation has been proposed as a potential release pathway including the spreading of microplastic contaminated waters or through degradation of plastic pipe systems (Zhang and Liu 2018). Estimates for the release of microplastics from these sources are entirely unknown.
Only a small number of studies have thus far investigated microplastic loadings in agricultural environments. Table 4.1 presents the plastic exposures and reported concentrations of these studies. The highest values are associated with fields undergoing multiple plastic exposure routes, greenhouse systems, sewage, irrigation, plastic litter, and proximity to roads, and are located in China (Chen et al. 2020; Zhang and Liu 2018). Fields that have undergone multiple applications of sewage sludge also present high soil microplastic concentrations (Corradini et al. 2019). Fields undergoing mulching with plastic films exhibit variable concentrations across two orders of magnitude (Huang et al. 2020; Liu et al. 2018; Lv et al. 2019a; Zhou et al. 2019), but this range may be linked to both the intensity and temporal frame of the plastic-cropping systems (Huang et al. 2020). Piehl et al. (2018) studied a farm in Germany that had no history of plasticulture or sludge application to soils. Despite this, low levels of microplastic contamination were observed, potentially derived from atmospheric deposition. The concentrations reported by Piehl et al. (2018) were several orders of magnitude below those reported by other studies. This suggests that agricultural practices involving plastics can significantly increase microplastic contamination in soils; however, several methodological disparities also exist between studies which may explain some of the observed variance. Differences in agricultural practices resulting from regional, seasonal, or crop-type variability may also result in large variations in soil microplastic concentrations. For example, different crops and environmental settings benefit from different forms of plasticulture and different countries implement a range of restrictions on the use of sewage sludge-derived biosolids.
Soil represents a complex matrix from which to isolate microplastic particles. Methods for analysing small plastic particle sizes are costly and time-consuming and require additional processing steps to clean up soil samples. For this reason, few studies examine the smallest microplastic size fractions, and, therefore, current assessments may represent an underestimate. Methods for analysing environmental nanoplastic contamination are largely non-existent, and complex soil matrices likely present an additional analytical challenge. Hence, there are no studies reporting nanoplastic contamination in soil environments, and so estimations of the contributions from agricultural products that contain or are expected to generate nanoplastics have not yet been possible. This is despite the possibility that nanoplastic particles could negatively influence soil functioning (Benckiser 2019).
The distribution of microplastic particles within agricultural environments is expected to be driven by a complex range of processes. Agricultural practices are likely to be highly relevant; in particular, the intensity and spatial scales associated with the use of plastic and plastic-containing products, the efficiency of plasticulture removal and waste handling, and the extent of ploughing or tilling of the land. This will govern the initial spread of particles across land and within soil profiles. Beyond this, processes related to wind erosion (Rezaei et al. 2019), bioturbation (Huerta Lwanga et al. 2017; Maaß et al. 2017; Yu et al. 2019), and water-mediated transport (Keller et al. 2020; O’Connor et al. 2019) are expected to transfer particles both within and from agricultural environments. Further research is necessary to document and quantify these processes under relevant field conditions.
4.2.2 Urban Environments
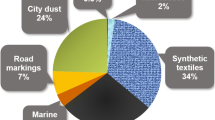
Urban environments can be expected to represent important domains for the release and cycling of plastic debris, based on the concentration of plastic production, consumption, and waste generation activities in these areas. Urban zones are characterised by higher population densities and may also comprise industrial areas that are involved in the production of plastics or manufacturing of plastic products. Releases of microplastics may include emissions from industry via air or water, the breakdown of larger plastic items (such as litter) due to environmental degradation or mechanical stress, and shedding from textiles such as clothing and home furnishings. Despite this, very few studies have thus far reported microplastic concentrations in samples from urban terrestrial environments. Fuller and Gautam (2016) identified poly(vinyl) chloride, polyethylene, and polystyrene microplastic in soils from an industrial area in Sydney, Australia, as part of a method development case study. Plastic debris was also noted in urban soil profiles from Stuttgart, Germany, but the size (macro-, meso-, micro-) of these particles was not described in detail (Lorenz and Kandeler 2005). Three studies have documented microplastics in urban dust samples from sites across Iran (Abbasi et al. 2017, 2019; Dehghani et al. 2017). Microplastic fibres and fragments were also observed at concentrations of approximately 2.9–166 particles g−1, and spatial patterns of microplastic abundance were correlated with factors such as the location of commercial or industrial districts, population density, and traffic load. Particles exhibited a range of colours and morphologies, representing a heterogeneous mix of potential sources. Importantly, particles potentially derived from tyre rubber and other road sources dominated the samples. Several additional studies have pointed towards roads as important sources of microplastics (e.g. Kole et al. 2017; Sommer et al. 2018). Hence, this section will focus on road environments as a critical component of urban systems regarding microplastic contamination and releases.
4.2.2.1 Roads
Roads represent complex anthropogenic environments comprising artificial ground, a high degree of mechanical abrasion from vehicle tyres on the road surface, and emissions of a range of contaminants from exhaust, tyres, the road surface, and other debris. Runoff from road environments is typically characterised by high levels of particulates and may be contaminated by a range of heavy metals (e.g. zinc, copper, cadmium, nickel) and organic pollutants (e.g. polycyclic aromatic hydrocarbons, organophosphates, octylphenols, phthalates) (Grung et al. 2017; Hallberg et al. 2014; Meland et al. 2010a, b; Meland and Rødland 2018). Road runoff has received renewed interest in recent years due to the presence of particles with polymer components; it has been identified as one of the largest sources of microplastic particles in the environment (Baensch-Baltruschat et al. 2020; Kole et al. 2017; Wagner et al. 2018). In particular, particles created by the wear and tear of car tyres are estimated to be the single largest source of microplastics in several countries, such as Norway, Sweden, and Denmark (Lassen et al. 2015; Magnusson et al. 2016; Sundt et al. 2014; Sundt et al. 2016; Vogelsang et al. 2019); although, these estimations are based on emission factors and need to be supported by peer-reviewed experimental or environmental evidence. Similar estimations using emission factors have been conducted in China, estimating that close to 55% of all primary microplastic emissions are derived from tyres (Wang et al. 2019a, b). The authors also compared their emissions to Norway and Denmark and calculated that the release in China is 85 times higher than in Norway and 400 times higher compared to Denmark. For this review we use the term road-associated microplastic particles (RAMP), first introduced in Vogelsang et al. (2019). RAMP comprises several categories of particle types: tyre-wear particles (TWP), road-wear particles from polymer-modified bitumen (RWPPMB), and road-wear particles from road marking (RWPRM) (Vogelsang et al. 2019). The RAMP terminology differs from the tire and road wear particle (TRWP) terminology, used in several other studies (Baensch-Baltruschat et al. 2020; Klöckner et al. 2019), by only including particles with plastic components whilst TRWP may also include particles without plastic components.
In general, very little research has thus far been conducted on RAMP. It is expected that large particles from road runoff will accumulate at the roadside or be captured in gully-pots, whilst smaller particles have the potential to spread further and be transported with the runoff (Vogelsang et al. 2019). However, these assumptions are based on the behaviour of road runoff particles in general, and further research on the emission of RAMP from road environments is needed.
A small number of studies have attempted to measure concentrations of TWP in the terrestrial environment. Fig. 4.2 presents the range of concentrations that have thus far been reported (Baensch-Baltruschat et al. 2020; Bye and Johnsen 2019; Rødland et al. 2020; Wik and Dave 2009). Tyres are composed of a complex mix of ingredients including natural and synthetic rubbers, mineral oils, fillers, antioxidants, and antiozonants (Wik and Dave 2009). Hence, they are difficult to quantify using chemical analyses. Nearly all the current studies have used tracers to estimate the concentration of TWP, such as different benzothiazoles and zinc that are present in tyres, whilst some others have instead measured concentrations of tyre-related polymers. The most commonly studied matrices include road dust from road surfaces, roadside soils, sediments from sedimentation basins, and river sediments. The studies have a wide geographic spread and represent different traffic volumes. Moreover, they employ a wide range of different analytical approaches, so comparisons between studies should be approached with caution (Rødland 2019).
Reported concentrations of TWP in terrestrial and freshwater environments. Each data entry is a mean value and red dots represent outliers. The figure summarizes a number of different studies from 1997 to 2020. (These are further described in Baensch-Baltruschat et al. 2020; Bye and Johnsen 2019; Rødland et al. 2020; Wik and Dave 2009)
Concentrations of TWP in the environment vary by several orders of magnitude (Fig. 4.2). This variability has a spatial component, related to proximity to the road environment and different environmental matrices. For example, concentrations of TWP in road dust differed between road surfaces outside tunnels (700–124, 000 mg kg−1) and inside tunnels (2700–210,000 mg kg−1) (Wik and Dave 2009). Moreover, Bye and Johnsen (2019) measured TWP in tunnel wash water from Smestad tunnel in Oslo (annual average daily traffic (AADT): 66,322) and found 4038 mg kg−1 of TWP. This corresponded to the accumulation of TWP since the previous tunnel wash – a period of 60 days – and a production of nearly 3 kg of TWP per day. Concentrations for road runoff material also ranged between an order of magnitude: 12–179 mg kg−1 (Wik and Dave 2009). In roadside soil, the highest concentrations were found closest to the road, ranging between 400 and 158,000 mg kg−1 at 0 m, with considerably lower concentrations from 1 to 30 m from the road (0–900 mg kg−1) (Baensch-Baltruschat et al. 2020; Wik and Dave 2009). One study also demonstrated the accumulation of TWP in roadside snow (563 mg L−1; Bauman and Ismeyer 1998). In road sedimentation basins, the highest concentrations were found in the sediments, 350–130,000 mg kg−1 (Klöckner et al. 2019; Wik and Dave 2009), and lower concentrations were found in water, 2.3 mg L−1 (Wik and Dave 2009). One study has looked at the retention of TWP in a soil retention filters and reported a concentration of 150,000 mg kg−1 (Klöckner et al. 2019). A study of microplastic particles in road de-icing salt (Rødland et al. 2020), used in areas with cold winter climate to ensure traffic safety, also found TWP in the salt, coming from the production sites and/or roads nearby the salt collection sites. However, the concentrations are very low compared to the contribution from tire wear itself, only 0.77 mg kg −1 salt.
These data include some discrepancies in the analytical approach. For example, Eisentraut et al. (2018) used thermal extraction desorption gas chromatography-mass spectrometry (TED-GC-MS) to measure the amount of styrene-butadiene rubber (SBR), a synthetic polymer, from sediments in a road runoff treatment. They recorded values of between 3.9 and 9.3 mg g−1 SBR in their samples. The approximate concentration of SBR in common tyres is 11.3% (Eisentraut et al. 2018). This probably varies a lot between different tyre brands and types of tyres (e.g. summer and winter tyres, studded and non-studded tyres); however, it can be used to calculate the concentration of TWP. This gives a result of 34.5–82.0 mg kg−1 for runoff sediments.
In addition to these studies, rubber particles potentially derived from tyre wear have been reported for snow samples from several sites in the Swiss Alps, Bremen, and Svalbard and from ice floes in the Fram Strait (Bergmann et al. 2019). This implies that TWP may be mobilised by atmospheric transport processes. However, the methods used in the study could not provide confirmation of TWP occurrence, and suspected particles were reported based upon their morphology.
Other components of RAMP are wholly under-researched. A single peer-reviewed publication reported RWPRM in sediments from the River Thames, UK (Horton et al. 2017a). No studies have recorded their occurrence in terrestrial samples. Furthermore, no studies have thus far measured the concentrations of RWPPMB alone in the environment. More research on the occurrence of RAMP in the terrestrial environment is needed to establish the relative contributions from different components and identify the scale of emissions from road environments.
4.2.3 Occurrence of Microplastics in Terrestrial Organisms
Very few studies have thus far reported the occurrence of microplastic particles in terrestrial organisms. Entanglement and incorporation of plastic into nests have been reported for both urban and agricultural crows in California, USA (Townsend and Barker 2014). Anthropogenic material, most commonly composed of plastic , was observed in 85.2% of nests, but this was typically in the size range of meso- or macro-plastic. Ingestion of microplastics by terrestrial birds in Shanghai, China, has been reported by Zhao et al. (2016), where plastic fibres and fragments accounted for 62.6% of litter items identified in digestive tracts. Carlin et al. (2020) observed an average of 6.22 microplastic particles present in the gastrointestinal tracts of birds of prey from central Florida, although many of these were identified to be rayon, which is sometimes excluded from microplastic counts as it is not a true synthetic polymer. It has been noted that microplastics now appear to be ubiquitous in the gut contents of bird species (Holland et al. 2016). Only a single study has documented the occurrence of microplastics in a terrestrial macroinvertebrate under field conditions. Panebianco et al. (2019) observed concentrations of 0.07 ± 0.01 particles g−1 tissue in three species of edible snails (H. aperta, H. aspersa, and H. pomatia) from Italy. Despite the current paucity of data on the uptake of microplastics by terrestrial organisms, numerous laboratory studies have demonstrated ingestion of microplastics by a range of species and have investigated related impacts. This is addressed in more detail in Chap. 8: Ecotoxicology of Plastic Pollution.
4.3 Pathways to Freshwater Environments
Most inputs of microplastics to freshwater systems can be characterised as release pathways – emissions are typically not direct sources and have instead travelled through other systems first. There are a small number of sources of microplastic to freshwater environments, such as the in situ fragmentation of plastic litter, point discharges from plastic industries, and the generation of micro-sized particles of polymeric paint or plastics from boats or other aquatic infrastructures. This section describes five key pathways to freshwater environments: spread from agricultural environments, releases from littering and landfill leachate, discharges from urban drainage systems, road runoff, and WWTP effluents.
4.3.1 Transfers from Agricultural Environments
Agricultural soils have been highlighted as potentially highly significant reservoirs of microplastics, which may actually exceed loadings currently observed in the global ocean (Nizzetto et al. 2016). The potential for agricultural microplastic contamination to propagate across wider spatial scales is, therefore, of particular interest in terms of global microplastic patterns and cycling. Processes such as windthrow, surface runoff, throughflow, and leaching are likely to be relevant for the transfer of plastic particles from soil systems, dependent upon factors such as particle size, morphology, and surface charge (Hurley and Nizzetto 2018). A small number of studies have demonstrated some of these processes experimentally within soil profiles (e.g. Keller et al. 2020; O’Connor et al. 2019), but, thus far, no published, peer-reviewed study has quantified the release of microplastics from soils and, especially, agricultural environments. A report from Ranneklev et al. (2019) presented preliminary data of microplastics in water discharged from a field amended with sewage sludge into a sedimentation pond connected to a stream in Norway. Approximately 2 particles L−1 were observed in the discharge water; however, the flow of discharge water from the field was not quantified. Nevertheless, this indicates that agricultural soils represent a release pathway for microplastic to freshwater systems. Based on the potential scale of microplastic contamination associated with agriculture, further research is urgently required to quantify transfers to the wider environment.
4.3.2 Transfers from Urban Environments
4.3.2.1 Littering and Leaching of Plastic Waste
Estimates for the mismanagement of plastic waste have been used as a means of assessing emissions of plastic to the marine environment (e.g. Jambeck et al. 2015). The transport pathways connecting this land-based release of plastic to the oceans are described as inland waterways, wastewater outflows, and wind action. Hence, the transfer of litter from populated or industrial areas to freshwater systems is often assumed. The generation of litter can be from littering practices or accidental releases during stages of waste handling, such as municipal waste management (Kum et al. 2005; Muñoz-Cadena et al. 2012). This litter may already be in the size range of microplastic particles or may act as a source of microplastics through the breakdown of larger plastics into micro-sized fragments. This fragmentation can be caused by weathering processes that chemically alter and weaken plastic polymers or through mechanical abrasion. Movement via water is likely to be an important process for the transport of litter from urban terrestrial environments to nearby freshwater systems and is addressed in more detail in Sect. 4.3.2.2. Wind action has also been identified as an important process distributing microplastics around the environment and is described in Sect. 4.5.
An additional release pathway associated with this form of (micro)plastic contamination is landfill leachate. Thus far, two published, peer-reviewed studies have reported concentrations of between 0.42 and 24.6 particles L−1 in leachate from municipal solid waste landfills in China (He et al. 2019; Su et al. 2019). Fewer particles were detected in older landfill systems, which has been linked to increased consumption and disposal of plastic in recent years (Su et al. 2019). Leachate discharges may emit microplastics to nearby soils or to freshwater systems.
4.3.2.2 Urban Drainage
Urban drainage systems designed to handle surface water runoff during precipitation events represent a key pathway linking urban and freshwater environments. Larger plastic items, typically litter, are often captured by drainage systems, and a body of research exists around documenting this process and engineering solutions to reduce blockages or prevent release into waterways (Armitage 2007; Armitage et al. 2001; Armitage and Rooseboom 2000; Marais et al. 2001, 2004). As described above, urban environments are expected to represent hotspots for microplastic contamination. Urban drainage is likely to act as a conduit for these particles to enter freshwater systems.
Several studies identify combined sewer overflows (CSOs) as a potentially important source of microplastics to freshwater systems (Ballent et al. 2016; Eriksen et al. 2013; Hurley et al. 2018), but very little work has attempted to quantify the scale of release or investigate the composition of microplastics. UNEP (2009) specifically identify sewage treatment and CSOs as one of the eight key land-based sources of marine litter, highlighting the important role that they are expected to play as a pathway for particles from urban environments. CSOs are a feature in many urban drainage systems; they allow for the direct release of untreated wastewater during periods of increased precipitation to prevent the system from backing up. Dris et al. (2018) sampled three CSOs in Paris, France, during a storm event. Very high levels of synthetic fibres (up to 190,000 fibres L−1) and fragments (up to 3100 fragments L−1) were reported. These results were higher than those observed for wastewater and stormwater alone, and it was suggested that this could be due to the accumulation of particles within the system during dry weather periods, which may then be resuspended once the CSOs are activated (Dris et al. 2018). This would represent a pulse of very high concentrations of microplastic released into rivers during storm conditions.
Microplastics have also been observed in stormwater ponds (Liu et al. 2019a, 2019b; Olesen et al. 2019). These receive runoff from a range of urban environments and aim to retain particles. The role of these systems in conveying microplastics to recipient water bodies – often freshwater systems – has not yet been quantified. These ponds do show potential for accumulating microplastic particles in their sediments, which may act as a temporary sink (Liu et al. 2019b). However, a diverse range of polymer and particle types has been reported including many which might not be expected to settle out to sediments.
4.3.2.3 Road Runoff
As discussed in Sect. 4.2.2.1, there are a limited number of studies that have investigated RAMP, and this includes the pathways from land to freshwater systems. The highest concentrations of TWP in environmental samples are found close to the road environment: from road dust and soil at the roadside. Lower concentrations have been reported from environments further from the road (Fig. 4.2). Values for freshwater environment vary considerably: between 36 and 179,000 mg kg−1 for river sediments and 1.6 and 36 mg L−1 for surface water (Baensch-Baltruschat et al. 2020; Unice et al. 2013; Wik and Dave 2009). However, there may be large variations in river water as well, especially due to the input during rainfall, as seen in the study by Kamata et al. (2000), where they reported TWP concentrations of 2200–5200 mg kg−1 during a storm flow. However, this demonstrates that transfers to freshwater systems do occur. Comparing the concentrations found in river water and water from sedimentation ponds to river sediments and sediments from sedimentation ponds, the current data provide clear indications that TWP will accumulate in the sediments. Additionally, transport of TWP from the road to freshwater is expected to be limited in areas where there is soil , sediment, and vegetation between the road and the recipient water body, as TWP is more likely to be retained.
Direct releases from the road environment to freshwaters are likely to occur. For example, particles may be released from bridges passing over freshwater via splashing or direct water outlets. Additionally, some larger roads have in-built drainage systems that collect and transport road runoff material and release it directly into a freshwater recipient. Many of these systems employ gully-pots which are expected to trap larger particles, but they rarely include sedimentation ponds or treatment systems to remove particulate and contaminants from runoff waters. The retention of TWP and other RAMP constituents in gully-pots is expected to be limited (Vogelsang et al. 2019); hence, direct discharges to freshwater environments are likely to represent an important pathway for RAMP release. Tunnel wash water represents a third direct discharge pathway. All road tunnels are washed regularly in order to maintain traffic safety and to avoid damage to technical instruments. The frequency of these tunnel wash events differs, usually determined by the number of cars passing through per day (AADT). In between these wash events, the tunnel is typically dry, and, therefore, there is a limited release of runoff from the tunnel. During a tunnel wash, large volumes of water are used, and this is collected by the tunnel drainage system. In some cases, the water passes through sedimentation and filtration treatment systems, but in other instances, it may be directly discharged to receiving water bodies. For example, Norway has over 1200 road tunnels, the third highest value globally (Vegkart, 2020). Only a small fraction of these tunnels receives any kind of treatment of the tunnel drainage water, and most release the runoff directly into a water recipient (Rødland and Helgadottir 2018). Several studies have documented high levels of particulate matter in tunnel wash water (Hallberg et al. 2014; Meland et al. 2010a, b; Meland and Rødland 2018); however, there are very few that investigate concentrations of TWP or other microplastics in this matrix. It is expected that tunnel wash waters may be highly enriched in RAMP. This is an area of research that should be addressed in future studies.
4.3.2.4 Wastewater Treatment Plant (WWTP) Effluents
Wastewater treatment plants (WWTPs) typically discharge treated effluents directly into recipient water bodies. This has the potential to represent an important release pathway for microplastics to freshwater environments. As discussed in Sect. 4.2.1, WWTPs receive microplastics from a diverse range of sources. Many of these are captured by wastewater treatment processes and transferred to the sewage sludge phase (64.4–99.9%). Despite this, total discharges from WWTPs are significant, and so this remaining percentage is expected to represent a significant release pathway for microplastics across temporal scales (Carr et al. 2016; Lv et al. 2019b; Sun et al. 2019).
Several studies have now provided an estimation of this release. The average concentration of microplastic in treated effluents generally falls below 1 particle L−1; however, large WWTPs can process several million litres of wastewater each day, resulting in daily emissions that are significant (Gatidou et al. 2019; Sun et al. 2019). For example, Mason et al. 2016a, b estimated that on average approximately four million microplastic particles were released each day from a single plant, in a study of 17 WWTPs in the USA. This study had a lower size limit of detection of 125 μm. Studies that go below this report far higher concentrations in the lowest size fraction (<20 μm), so the number of particles that are released can be expected to be far higher (e.g. Simon et al. 2018). The release of microplastics by WWTPs is further supported by studies that have documented significant increases in fluvial microplastic concentrations downstream from WWTP effluent releases (e.g. Estahbanati and Fahrenfield 2016; Kay et al. 2018; Vermaire et al. 2017). The release of microplastics from WWTPs can be expected to vary through time and space. Smaller, rural WWTPs process far smaller volumes of wastewater per day but may also have low trapping efficiencies for microplastic particles (e.g. Wei et al. 2020). It is also of interest to capture the influence of precipitation events on microplastic release by WWTPs, where plants may release pulses of untreated wastewater into recipient waterbodies. Moreover, approximately 80% of the world’s wastewater is emitted without sufficient treatment (UNESCO 2017), for which the potential microplastic release is unknown.
Fibres are commonly reported to be the dominant particle type present in WWTP effluents (Mason et al. 2016a, b; Ruan et al. 2019; Yang et al. 2019). A single garment can shed more than 1900 fibres per domestic laundry wash, resulting in >100 fibres L−1 of laundry effluent (Browne et al. 2011). Many different treatment steps are employed by WWTPs globally, with capture rates ranging from 0 to 99.9% (Zhang and Chen 2020). The efficacy of these clean-up steps is expected to differ across the spectrum of particle sizes and morphologies (Carr et al. 2016; Lusher et al. 2019; Sun et al. 2019). This is particularly relevant for fibrous particles, which have been noted as the most challenging microplastic type for removal in wastewater due to their high length to width ratio and potential to curve and bend (Ngo et al. 2019). In some cases, the presence of microplastics can actually exhibit a negative impact on the efficiency of wastewater treatment processes (Zhang and Chen 2020), further complicating their removal.
It is important to note that comparing studies of microplastic releases from WWTP is challenging, as the methodologies used in each study, from sample collection, sample processing, and the size range of particles may differ substantially, and no harmonised methodology has yet emerged. There are also different approaches to sampling microplastic particles in streams receiving WWTP effluents, many of which may not capture very small microplastic particles. Excluding the smaller fraction of microplastics from studies may result in underestimating the microplastics released from WWTPs as they may be less likely to be captured by treatment processes (Lusher et al. 2019). Further research is required to quantify the scale of microplastic release by WWTP effluents.
4.4 Microplastics in Freshwater Systems
4.4.1 Microplastics in Rivers
Fluvial systems comprise running bodies of water that connect terrestrial, lacustrine, glacial, and marine environments. They represent important long-range transport pathways and act as conduits for suspended sediments and contamination through the landscape. Rivers and streams are expected to be highly complex and dynamic regarding the accumulation and transfer of microplastic particles. As has been established in the previous section, fluvial environments are connected, with many sources and release pathways for microplastic particles, including both point and diffuse releases across different spatial and temporal scales. Numerous studies have now documented microplastic contamination in rivers or streams (Scherer et al. 2020).
Variation is observed in the microplastic contamination reported within river systems. This spatial and temporal heterogeneity is not common across all studied catchments. These differences point towards some of the complexity associated with river systems. For example, some studies report a common longitudinal pattern of increasing microplastic concentrations with distance downstream (e.g. Jiang et al. 2019; Shruti et al. 2019), which likely represents a culmination of microplastic sources and pathways. In contrast, other studies show a less clear-cut pattern of microplastic abundance, especially in highly urbanised systems (e.g. Hurley et al. 2018). River sediments, on average, present higher concentrations than overlying waters (Li et al. 2020) and may act as a temporary store for microplastic particles (Castañeda et al. 2014). However, relating microplastic abundance in moving waters to that in more static sediments is complicated.
The different methodological approaches to sampling fluvial environments make data comparability challenging. Many variations exist between samples; for example, methods of sampling (sediment grabs, sediment cores, nets, pumps, etc.), sample matrices (surface waters, water column, sediments, etc.), particle size fractions, laboratory analytical methods (sample purification, density separation, pore sizes of filtration approaches, etc.), and reporting units (particles m−2, m−3, L−1, kg−1, etc.) (Blettler et al. 2018). Table 4.2 presents a selection of studies reporting fluvial microplastic contamination that utilise a range of different approaches. Based on this degree of discrepancy between methodologies, it is difficult to partition the observed differences in reported concentrations between methodological and environmental factors. This is further hampered by the wide range of potential controls on microplastic release and distribution in river systems including varying hydrological and geomorphological conditions, density and proximity of sources and release pathways, catchment characteristics and land use, anthropogenic modifications such as dams, and seasonal variability in microplastic releases and river characteristics (Blettler et al. 2018; Mai et al. 2019; McCormick et al. 2016). Furthermore, it has been suggested that existing assessments of riverine microplastic contamination may miss significant variability due to their selected spatial and temporal scales (Stanton et al. 2020).
In dynamic systems such as rivers, there is a need to examine how microplastic distributions change across spatial and temporal scales and in response to different controls. Figure 4.1 presents some of the processes likely to be relevant for microplastic transport in river systems. For example, microplastics can settle in riverbeds but may be resuspended during high energy events, such as floods, and transported further along the river (Hurley et al. 2018). The extent and controls of this remobilisation are essentially unknown (Alimi et al. 2018), and only a small number of studies have begun to investigate important hydrological controls on particle retention and transport (e.g. Ockelford et al. 2020). It is assumed that the smaller the microplastic particle, the lower its retention in river systems based upon the lower flow velocities required for entrainment (Besseling et al. 2017). Connectivity between river channels and the overbank zone during flood events may lead to deposition or mobilisation of microplastic particles. Seasonal variability is also likely to play an important role in some systems. Watkins et al. (2019) identified that hydrological differences between spring high flow and summer low flow were the dominant factor determining microplastic concentrations in two streams in New York, USA. These studies suggest that the hydrodynamics of the river strongly impact microplastic distributions and emissions to the marine environment (Besseling et al. 2017; McCormick et al. 2016). Furthermore, although more attention is typically directed to larger river systems, smaller streams should also be investigated as in many cases they are the primary interface between land, usage of plastics, and drainage networks (Dikareva and Simon 2019). Microplastic contamination of headwater streams has been reported (Hurley et al. 2018), demonstrating the pervasive nature of fluvial microplastic contamination.
4.4.2 Microplastics in Lakes
Microplastics were first recorded in a lake environment in 2012 (Faure et al. 2012). Since then, 36 additional published studies have investigated the occurrence of microplastics in the waters or sediments of lakes, globally. The majority of these studies can be broadly grouped into three key locations: Great Lakes system, European lakes, and Chinese lakes (Fig. 4.3). Lakes may receive microplastic particles from a wide range of potential sources or release pathways, including WWTP effluents (Uurasjärvi et al. 2020), industrial discharges (Eriksen et al. 2013), fisheries (Wang et al. 2018; Yuan et al. 2019), and inflowing rivers (Ballent et al. 2016; Corcoran et al. 2015). They represent complex environmental systems that have the potential to transport, disperse, or accumulate particles according to an array of different processes (Fig. 4.1).
Methodological disparities complicate efforts to compare findings from different studies. This includes the field sampling procedure, which has been shown to result in significant differences between reported microplastic concentrations for different sampling apparatus types (Uurasjärvi et al. 2020). Further variation in the particle sizes classes analysed introduces additional uncertainty. The lakes investigated also represent a spectrum of lake and catchment sizes and types. Hence, it is difficult to determine whether differences between studies are mainly derived from the methodological approach or to a higher extent relate to environmental factors. Despite this, variability between spatial or temporal concentrations within single studies that apply one methodology indicates that environmental factors are important in governing levels of microplastic contamination (Nan et al. 2020; Scherer et al. 2020). It is notable that despite possible methodological difference, the presence of microplastics has been reported in all lakes studied thus far, even in remote locations (Free et al. 2014; Zhang et al. 2016a, b).
Assessments of surface waters report concentrations ranging from 0.21 (Fischer et al. 2016) to 34,000 particles m−3 (Yuan et al. 2019). Low-density plastic types such as polyethylene and polypropylene are commonly reported as the dominant microplastic types (Sighicelli et al. 2018; Wang et al. 2018; Xiong et al. 2018). This concurs with the expected buoyant properties of these polymer types. Fibres are also commonly reported (Anderson et al. 2017; Wang et al. 2018), despite typically being composed of polymer types that are denser than water. Several studies show high concentrations close to population centres and point towards the surface currents generated by prevailing winds as an important process governing microplastic distributions at the water surface (Fischer et al. 2016; Free et al. 2014; Migwi et al. 2020).
Lake sediment concentrations vary between studies. In smaller lakes, concentrations of microplastics in lake sediments have been shown to reflect processes influencing surface water distributions, such as prevailing wind or proximity to inputs, suggesting that denser polymer types undergo transport through the lake prior to sedimentation (Vaughan et al. 2017). In larger lake systems, a lack of correlation between surface water and sediment concentrations is often reported (Yuan et al. 2019), but this may be due to the spatial resolution of sampling campaigns. River tributaries have been identified as a depositional environment for microplastics as energy conditions change during the transition into the lake environment (Ballent et al. 2016). Lenaker et al. (2019) demonstrated that partitioning between surface waters, sub-surface waters, and sediments occurred at a density threshold of 1.1 g cm−3 in a North American freshwater lake system. Despite this, low-density polymer types are sometimes observed in lake sediments (e.g. Sruthy and Ramasamy 2017). This is contrary to the expected buoyancy of these particles but may be explained by processes such as biofouling that increases particle bulk density (Chen et al. 2019). Lake sediments also have the potential to accumulate and preserve microplastic particles through processes of sedimentation and burial. This has been reported for Lake Ontario (Corcoran et al. 2015); Hampstead Pond, London, UK (Turner et al. 2019); and Donghu Lake, Wuhan, China (Dong et al. 2020).
4.4.3 Occurrence of Microplastic in Freshwater Organisms
To assess the status of freshwater systems, there has been a long tradition to use macroinvertebrates as indicator species. They represent a diverse group of organisms that show tolerance and sensitivities towards different stressors, present different feeding strategies, inhabit different environments, and have a range of lifespans (including long life cycles allowing for accumulation of contaminants). Research has now begun to investigate macroinvertebrates as a measure for microplastic contamination.
The majority of studies on freshwater macroinvertebrates has been conducted in the laboratory to measure the ecotoxicity of different polymers and particles types. Only a comparatively small number of studies have documented the occurrence of microplastics in macroinvertebrates in environmental samples (Akindele et al. 2019; Hurley et al. 2017; Nan et al. 2020; Nel et al. 2018; Su et al. 2018; Windsor et al. 2019a). These investigate a range of organisms including mayflies, caddisflies, gastropods, clams, and shrimp. All of the 20 different investigated species of six classes: Insecta, Clitellata, Bivalvia, Amphibia, Malacostraca, and Gastropoda were found to contain microplastic particles. The investigated sites covered lakes, river deltas, urban rivers, and small streams. Concentrations vary from 0.07 to 5 particles individual−1, 0.01 to 0.042 particles mg−1 d.w., and 0.0003 to 1.12 particles mg−1 w.w (Akindele et al. 2019; Hurley et al. 2017; Nan et al. 2020; Nel et al. 2018; Su et al. 2018; Windsor et al. 2019a). At present, there is insufficient data to draw conclusions regarding differences in microplastic uptake based on different feeding traits, trophic interactions, or microplastic particle size. However, it has been suggested that generalist species are more likely to ingest microplastic than predators (Scherer et al. 2018) and that non-selective feeders are more likely to ingest microplastic particles than selective feeders (Scherer et al. 2017). Fibres are the most commonly reported particle type (e.g. Akindele et al. 2019; Hurley et al. 2017; Nan et al. 2020; Su et al. 2018), but it is not known whether this reflects the feeding behaviour of macroinvertebrate species or the dominant particle type present in the local environment.
These studies have thus far applied several different methods for separating microplastic from organisms. These include alkaline hydrolysis (KOH or NaOH), digestion with hydrogen peroxide, acid digestion (HNO3), and combined approaches (KOH + H2O2). Some of the studies rely on visual assessment of microplastic particles only – two thirds verify a subsample of particles using chemical analytical techniques. Since most macroinvertebrates are small, and the majority of their food items are thereby also small, a visual analysis may not be sufficient to capture the full-size spectrum of plastic particles that may be ingested. There is also an absence of quality assurance and quality control measures such as including both blank and spiked samples in many of the studies, emphasising the need for methodological improvements.
A larger number of studies have reported the occurrence of microplastic in freshwater fish (Andrade et al. 2019; Biginagwa et al. 2016; Horton et al. 2018; Jabeen et al. 2017; Phillips and Bonner 2015; Sanchez et al. 2014; Silva-Cavalcanti et al. 2017). Over 50 species of fish have been analysed for microplastic ingestion under field conditions thus far. This has mostly been documented for riverine specimens, but studies have also been conducted in lakes (Biginagwa et al. 2016) and a stormwater pond (Olesen et al. 2019). Concentrations range from 0 to 65 microplastic particles individual−1. It is important to note, however, that most studies only investigate the gut content so total concentrations are not known. Although, it is expected that only small microplastic particles have the potential to pass gut membranes. A single study found no plastic particles in the gut contents of fish: northern pikes, roach, and bream from Lake Geneva (Faure et al. 2012). The majority of studies perform a visual examination of the gastrointestinal tract or digest the gut contents using KOH or H2O2; however, small fish may be freeze-dried and digested (e.g. Olesen et al. 2019), and one study has also documented the occurrence of polyethylene and polystyrene in liver samples digested using sodium hypochlorite (e.g. Collard et al. 2018). Some trends have been reported. For example, McGoran et al. (2017) found that benthic-feeding fish ingested more microplastics (75%) than pelagic-feeding fish (20%) in the Thames River. Moreover, Horton et al. (2018) observed that microplastics in gut content was positively correlated with fish size, which is in turn typically associated with sex. The reported microplastic burden on freshwater fish species may also represent an underestimate due to the lower size limit of the studies; Roch et al. (2019) reported that the majority of microplastic ingested may be below 40 μm. This is supported by the high concentrations reported when using high-resolution μFT-IR imaging methodologies (e.g. Olesen et al. 2019).
Thus far, there are no studies that document the occurrence of RAMP in organisms under field conditions. Several studies have demonstrated the uptake of hazardous compounds associated with tyres during laboratory toxicity testing, which are summarised in Table 4.3. However, only a single study was able to confirm uptake of tyre particles by any of the organisms (Redondo-Hasselerharm et al. 2018); on average between 2.5 and 4 tyre tread particles were ingested by freshwater benthic macroinvertebrates. No published, peer-reviewed studies, in the field or laboratory, have yet looked for ingestion of RWPRM or RWPPMB.
4.5 Microplastics in the Atmosphere
The potential for microplastic occurrence in the atmosphere above both land and sea is as yet largely unexplored. Processes of initial entrainment, localised updraft, convective mixing and advection, horizontal conveyance, and settling are expected to be relevant for the suspension, dispersion, transport, and deposition of particles across spatial scales (Fig. 4.1). These processes are likely affected by the size, morphology, and density of plastic particles. For example, particle size influences the movement of particles by wind at the land-air interface. Larger particles may move in a rolling motion, known as ‘creep’, whilst smaller particles may be transported through saltation motion, hopping along the land surface, or by suspension, based upon thresholds for particle motion and entrainment (Raupach and Lu 2004). Obstacles in the landscape may also represent temporary stores for microplastic particles, as has been demonstrated for terrestrial plants (Liu et al. 2020a).
Atmospheric deposition represents a pathway to terrestrial and freshwater environments. Several studies have reported deposition rates of between 0 and 11,130 particles m−2 day−1 (Allen et al. 2019; Cai et al. 2017; Dris et al. 2016, 2017; Klein and Fischer 2019; Liu et al. 2019a, b, c, d; Wright et al. 2020; Zhou et al. 2017); however, methodological differences, including discrepancies in the particle size classes analysed, hinder comparisons between datasets. For most studies, deposition appears to be higher in urban areas, which is likely associated with the quantity and proximity of sources. However, Klein and Fischer (2019) report higher concentrations in rural areas, which they attribute to the influence of forest canopy textures in combing out suspended particles. Rayon, polyamides, and polyesters are the dominant polymer types associated with fibrous microplastic , whilst polyethylene, polypropylene, and polystyrene are regularly reported for other particles types. For particle types such as fragments and films, studies typically report a higher deposition of microplastics concentrated in the smallest size categories and associated with lower-density polymer types. This demonstrates the influence of particle characteristics on atmospheric transport. For fibres, however, larger particles are commonly observed, and particles are composed of higher-density polymer types. Here, shape likely plays a dominant role in initial entrainment, transport, and deposition, where the irregular form of fibres encourages continued suspension. This has been demonstrated by Abbasi et al. (2019) who analysed urban dust from Asaluyeh county in Iran and found that, whilst deposited dusts were composed of a diverse range of particles types, suspended dusts contained only fine or fibrous particles. Furthermore, several studies of atmospheric deposition of microplastics report fibres as the dominant particle shape (e.g. Wright et al. 2020; Zhou et al. 2017; Dris et al. 2017). It has now been estimated that between 7.64 and 33.76 tonnes of fibrous atmospheric microplastics were generated globally during the year 2018 (Liu et al. 2020b).
It is difficult to quantify the role of atmospheric deposition as a pathway for microplastics to freshwater systems, due to the influence of multiple potential sources of microplastic contamination which complicates the assessment of individual inputs (Free et al. 2014). However, a recent study of atmospheric contamination of glacial ice shed some light on the potential contribution of atmospheric deposition. Ambrosini et al. (2019) found 74 ± 28 microplastics kg−1 of supraglacial sediments found on the glacier surface, which indicate a baseline level of contamination for that region. Further research is required to quantify the rates of deposition across different spatial and temporal scales. Moreover, the cryosphere represents a vastly understudied environmental compartment, which may yield insights into baseline atmospheric deposition rates and the dynamics of long-range transport (Windsor et al. 2019b).
Factors such as precipitation and wind speed have been positively correlated with microplastic deposition in a remote catchment in the Pyrenees (Allen et al. 2019). Both rain and snowfall events led to increased deposition of particles, where event occurrence and intensity were found to be more important than the duration of precipitation. This is supported by a recent study identifying high concentrations of microplastics in snow samples from Europe and the Arctic (Bergmann et al. 2019).
Tracking air mass trajectory through atmospheric modelling has successfully demonstrated medium-range transport of microplastic particles over an extended sampling duration (Allen et al. 2019); however, transport over longer distances within a regional context was also likely to have occurred. This is further studied by an assessment of microplastics in the sea air, which used the same backward trajectory modelling approach to identify the terrestrial-to-marine transfer of microplastics in the west Pacific Ocean (Liu et al. 2019a, b, c, d). The same study reported that trajectory modelling indicates that suspended microplastic particles from that region could be transported to the Arctic through the movement of air masses. Microplastics have been observed in several remote regions that are typically considered ‘pristine’ due to the very low levels of anthropogenic influence in the vicinity (Allen et al. 2019; Free et al. 2014; Zhang et al. 2016b, 2019). Medium- and long-range transport of particles is considered to be a key mechanism delivering microplastic contamination to these locations. Questions remain regarding the potential for long-range atmospheric transport of microplastic particles, atmospheric residence times, and transformation (e.g. degradation, fragmentation) of microplastic within the atmosphere.
4.6 Microplastics: Where Do They End Up?
4.6.1 Export to the Marine Environment
Due to catchment dynamics, erosion, and transport processes, many of the microplastic particles released on land are expected to eventually end up in the marine environment (Hale et al. 2020). Several seminal studies have estimated significant fluxes of plastic from land to the ocean (Jambeck et al. 2015; Lebreton et al. 2017; Schmidt et al. 2017); however, these deal with mass estimates, which predominately illustrate flows of macroplastic. Some studies have instead modelled microplastic release, demonstrating increases in microplastic export over the next several decades (Siegfried et al. 2017; van Wijnen et al. 2019). These studies highlight some geographical hotspots for release, such as South East Asia, and highlight wastewater treatment and TWP as important origins for microplastic that reaches the oceans. Improvements in sewage treatment were identified as a potential solution to significantly reduce future marine export of microplastics from land-based sources.
As previously discussed, microplastic transport in freshwater systems may vary in regard to seasonal or episodic changes in hydrological conditions. This is likely to influence the flux of microplastic to the oceans. Flood events are important for the transport of suspended sediments; over 90% of the annual suspended sediment flux of a river may be associated with storm events (Walling et al. 1992). Hurley et al. (2018) reported an export of 0.85 tonnes of microplastic particles from bed sediments in a medium-sized catchment in the UK. This was associated with a high-magnitude flood event that scoured accumulated microplastics from riverbeds and transferred them downstream and potentially out to the ocean . This is supported by evidence for significant increases in coastal microplastic contamination in the vicinity of river outlets following flood events (e.g. Gündoğdu et al. 2018; Lee et al. 2013; Veerasingam et al. 2016).
This transfer from land to sea may not always be unidirectional. Rivers influenced by tidal changes see a reversal in flow direction for some, or all, of the cross section during high-tide conditions, which may transport plastics upstream (van Emmerik et al. 2019). Moreover, coastal flooding may return marine microplastics to the land through deposition during the inundation of land. The impact of these factors should be considered when establishing robust flux estimations and assessing the fate of microplastic particles.
4.6.2 Microplastic Sinks in Terrestrial and Freshwater Environments
In some cases, land-based sources of microplastic and the terrestrial and freshwater systems involved in their dispersal and transformation are perceived as vectors for marine microplastic contamination. In regard to hydrological and geological cycles, across long timescales the majority of waters and sediments can be expected to reach the marine environment. Hence, microplastics are also likely to end up at this ultimate destination. Despite this, it is important not to overlook the need to better understand terrestrial and freshwater contamination dynamics, the risks posed to these ecosystems, and measures to limit or remediate contamination in these settings. Microplastics in the environment may pass through several terrestrial and marine cycles related to continuous and complex movement between both biotic and abiotic environmental compartments (Bank and Hansson 2019). Without a thorough understanding of the transfer of particles from the source to the ocean , efforts to reduce or remediate microplastic contamination will be hindered.
In addition, within terrestrial and freshwater systems, there are several candidate environments that may act as environmental sinks for microplastic particles, interrupting their ultimate transport to the coast. These may represent temporary or permanent sinks across different temporal and spatial scales. For example, lake sediments, where microplastics may accumulate and become buried by sediment deposits, have been identified as sites of plastic preservation and storage (Corcoran et al. 2015; Dong et al. 2020; Turner et al. 2019). At the bottom of a lake and beneath sediment layers, plastic particles are isolated from many of the degradative forces that initiate weathering, such as photodegradation (Corcoran et al. 2015). Microplastic particles have been identified in sediment layers as deep as 75 cm and dated to have been deposited during the early twentieth century, at the onset of plastic production (Turner et al. 2019). Once particles are buried to that depth, a significant disturbance event is required to remobilise sediments – such as dredging activities or a very high-magnitude storm. In the absence of such disturbance, lake sediments can be considered permanent or very long-term sinks for microplastic particles.
Other environments that may represent environmental sinks – but have not yet been studied regarding this specific question – include a range of sedimentary landscapes. These comprise settings that have been identified as environmental sinks for other contaminant types. For example, alluvial environments act as stores for many sediment-bound contaminants (e.g. Lecce and Pavlowsky 1997; Walling et al. 2003; Winter et al. 2001). Floodplain soils have already been shown to contain microplastic particles (Scheurer and Bigalke 2018). Depending on the geomorphological conditions of the environmental setting, floodplains may represent long-term stores of microplastic particles. Additionally, they may constitute future diffuse sources of microplastic particles as sediments with connectivity to freshwater systems may be reworked into active channels.
Environmental sinks can be defined by their temporal frame. From this perspective, not all sinks may be sedimentary. For example, residence times of waters in large lake systems, such as the Great Lakes, can reach close to 100 years (Mason et al. 2016a, b). If particles are also retained in these water masses, surface and sub-surface waters in lacustrine environments may represent a short- to medium-term sink for microplastic particles. This is particularly relevant for lakes that are not consistently connected with fluvial systems, such as floodplain lakes. Additionally, entrapment in low-energy zones in fluvial systems – such as in dense riparian vegetation – may constitute short-term storage of buoyant microplastics. The residence times associated with these stores for microplastics particles, as well as the thresholds required to transition these environments to ‘sources’, require further investigation.
4.7 Future Research Agendas
Microplastic contamination is globally pervasive across terrestrial and freshwater environments. This review has drawn together research on several important sources and release pathways for microplastics including roads, agriculture, and wastewater treatment. Many environmental settings are expected to receive significant microplastic loadings, which likely represents a greater annual release than that estimate for the marine environment. Terrestrial and freshwater systems can be characterised by considerable complexity, whereby a range of dynamic processes are expected to influence the distribution, transport, and fate of microplastic particles.
Through this review, a set of specific directives for future research have been identified:
-
i. Harmonisation of Methods and Reporting, Including Improved Quality Assurance and Control (QA/QC) Practices, to Ensure Sufficient Data Quality and Permit Comparability Between Datasets
-
The review of studies of microplastic occurrence in terrestrial and freshwater environments, including samples of water, sediment, and biota, is characterised by the wide range of methodological approaches undertaken. This includes discrepancies between the sampling techniques, sample treatment, analytical technologies employed, and particle size classes analysed. In addition, many studies do not employ a similar set of QA/QC measures, so it is not possible to assess the quality of reported data. Findings are also often reported in different ways, for example, using different units or publishing only summary statistics that also differ (e.g. minimum/maximum, mean, median). The culmination of this variability is the lack of comparability between different studies. Harmonisation of analytical methods and reporting formats, and the publication of data in appropriate repositories, will help to reduce uncertainties in a holistic, global overview of the status of contamination, as well as providing meaningful baselines from which to track the impact of reduction or remediation measures.
-
ii. A Thorough Assessment of Microplastic Sources, Fate, and Impacts in Agricultural Environments
-
Agricultural environments represent the convergence of several sources and release pathways of microplastic particles. Particles may also be associated with higher chemical burdens from plastic additives (such as for decreasing photodegradation of mulching films) or sorbed contaminants (such as from WWTPs), although the extent and significance of this are relatively unknown. The status of contamination of agricultural settings is relatively unknown, and the fate of particles in agricultural soils remains under-researched, which makes it difficult to assess the relative contributions of different sources under relevant environmental conditions or the accumulation of particles over time. A small body of research on the impacts of microplastic contamination in agricultural environments is just now emerging. More research is required to gain a holistic perspective on the risks posed by microplastics across spatial and temporal scales. This is particularly important given the potential for any identified negative effects to impact upon soil health and food security.
-
iii. Quantification of Road-Associated Microplastic Particles (RAMP) as a Source of Microplastic to the Environment
-
For road-associated microplastic particles (RAMP), there are major knowledge gaps concerning environmental loadings, transport from the road to different matrices, and retention in gully-pots and water treatment systems. More research is urgently needed on RAMP in order to accurately assess how much is released into the environment, including the relative contributions from different road-related sources (TWP, RAMPPMB, RAMPRM). This is important given the spotlight that has now been placed on RAMP in several assessments of globally significant sources of microplastic to the environment. Quantification should be achieved through a new and optimised approach to analysing RAMP in environmental samples.
-
iv. Measures and Technologies to Reduce Microplastic Emissions to Wastewater or to Separate Particles Within WWTP Systems
-
This review highlighted the role of wastewater systems, such as WWTPs and CSOs, as a release pathway for microplastic particles to both terrestrial and freshwater environments. A diverse range of sources input microplastic particles to wastewater, and efforts should be made to reduce these at the source to reduce the burden on WWTPs and limit releases from untreated discharges such as CSOs. Much of the world’s wastewater is not connected to a WWTP and is instead released untreated. Improvements in the global capacity of wastewater treatment would limit the environmental release of microplastics in many countries. Land application of sewage sludge has been identified as a primary release pathway for many microplastic types (ECHA 2019). Technologies to capture and remove plastic particles in WWTPs may help to reduce the burden on global soil environments.
-
v. A Better Understanding of the Controls Underpinning the Retention and Transport of Microplastic Particles in Freshwater Systems, Including More Accurate Flux Estimates to the Marine Environment
-
Microplastic particles in freshwater systems are likely to follow a complex pathway from their release to their ultimate fate. This may include several processes that interrupt downstream transport. These dynamics require further investigation to establish thresholds and controls on microplastic transport in freshwater environments. The majority of particles are expected to eventually end up in the marine environment, via fluvial systems. Estimates for this flux need to draw upon process-based research to incorporate appropriate complexity and identify the relevant controls on microplastic release to the marine environment. Moreover, further research on the dynamics of microplastic transport and spatial patterns of contamination will identify zones of microplastic accumulation in freshwater systems and can highlight the areas at greatest risk to potential negative impacts of contamination. This will help to focus efforts to protect freshwater ecosystems.
-
vi. Further Investigation of the Occurrence of Microplastics in Terrestrial and Freshwater Organisms, with a Specific Focus on Particle Types Such as RAMP
-
Many ecotoxicological studies are determining the effects associated with different microplastic particles and loadings, but evidence for uptake under field conditions is still scarce. Exposure represents half of the equation to evaluate risk, and so a more detailed investigation of the uptake of particles in real environment conditions is essential to contextualise ecotoxicological studies and inform risk assessments.
-
vii. Assessment of the Spatial and Temporal Scales of Environment Sinks for Microplastic Particles
-
This review highlighted several candidate environments that may act as temporary, long-term, or even permanent sinks for microplastic particles in freshwater and terrestrial settings. Some initial studies have investigated particle accumulation and potential residence times for some of these, but further research is required to establish the spatial and temporal scales upon which these environments act as stores, including the potential for them to become future sources of microplastic contamination through reworking and remobilisation. This is necessary to gain a better long-term perspective of environmental contamination and build more appropriate and better-targeted approaches to remediation instead of short-term fixes (Table 4.4).
References
Abbasi S, Keshavarzi B, Moore F, Delshab H, Soltani N, Sorooshian A (2017) Investigation of microrubbers, microplastics and heavy metals in street dust: a study in Bushehr city, Iran. Environ Earth Sci 76(23):798. https://doi.org/10.1007/s12665-017-7137-0
Abbasi S, Keshavarzi B, Moore F, Turner A, Kelly FJ, Dominguez AO, Jaafarzadeh N (2019) Distribution and potential health impacts of microplastics and microrubbers in air and street dusts from Asaluyeh County, Iran. Environ Pollut 244:153–164. https://doi.org/10.1016/j.envpol.2018.10.039
Accinelli C, Abbas HK, Shier WT, Vicari A, Little NS, Aloise MR, Giacomini S (2019) Degradation of microplastic seed film-coating fragments in soil. Chemosphere 226:645–650. https://doi.org/10.1016/j.chemosphere.2019.03.161
Akindele EO, Ehlers SM, Koop JHE (2019) First empirical study of freshwater microplastics in West Africa using gastropods from Nigeria as bioindicators. Limnologica 78:125708. https://doi.org/10.1016/j.limno.2019.125708
Alimi OS, Farner Budarz J, Hernandez LM, Tufenkji N (2018) Microplastics and nanoplastics in aquatic environments: aggregation, deposition, and enhanced contaminant transport. Environ Sci Technol 52(4):1704–1724. https://doi.org/10.1021/acs.est.7b05559
Allen S, Allen D, Phoenix VR, Roux GL, Jiménez PD, Simonneau A et al (2019) Atmospheric transport and deposition of microplastics in a remote mountain catchment. Nat Geosci 12(5):339–344. https://doi.org/10.1038/s41561-019-0335-5
Ambrosini R, Azzoni RS, Pittino F, Diolaiuti G, Franzetti A, Parolini M (2019) First evidence of microplastic contamination in the supraglacial debris of an alpine glacier. Environ Pollut 253:297–301. https://doi.org/10.1016/j.envpol.2019.07.005
Anderson PJ, Warrack S, Langen V, Challis JK, Hanson ML, Rennie MD (2017) Microplastic contamination in Lake Winnipeg, Canada. Environ Pollut 225:223–231. https://doi.org/10.1016/j.envpol.2017.02.072
Andrade MC, Winemiller KO, Barbosa PS, Fortunati A, Chelazzi D, Cincinelli A, Giarrizzo T (2019) First account of plastic pollution impacting freshwater fishes in the Amazon: ingestion of plastic debris by piranhas and other serrasalmids with diverse feeding habits. Environ Pollut 244:766–773. https://doi.org/10.1016/j.envpol.2018.10.088
Armitage N (2007) The reduction of urban litter in the stormwater drains of South Africa. Urban Water J 4(3):151–172. https://doi.org/10.1080/15730620701464117
Armitage N, Rooseboom A (2000) The removal of urban litter from stormwater conduits and streams: paper 1-The quantities involved and catchment litter management options. Water SA 26(2):181–188. Retrieved from https://journals.co.za/content/waters/26/2/AJA03784738_2350
Armitage N, Marais M, Pithey S (2001) Reducing urban litter in South Africa through catchment based litter management plans. Models and Applications to Urban Water Systems, Monograph 9:37. Retrieved from https://www.chijournal.org/R207-03
Avio CG, Gorbi S, Regoli F (2015) Experimental development of a new protocol for extraction and characterization of microplastics in fish tissues: first observations in commercial species from Adriatic Sea. Mar Environ Res 111(Supplement C):18–26. https://doi.org/10.1016/j.marenvres.2015.06.014
Baensch-Baltruschat B, Kocher B, Stock F, Reifferscheid G (2020) Tyre and road wear particles (TRWP) – a review of generation, properties, emissions, human health risk, ecotoxicity, and fate in the environment. Sci Total Environ 733:137823. https://doi.org/10.1016/j.scitotenv.2020.137823
Ballent A, Corcoran PL, Madden O, Helm PA, Longstaffe FJ (2016) Sources and sinks of microplastics in Canadian Lake Ontario nearshore, tributary and beach sediments. Mar Pollut Bull 110(1):383–395. https://doi.org/10.1016/j.marpolbul.2016.06.037
Bank MS, Hansson SV (2019) The plastic cycle: a novel and holistic paradigm for the anthropocene. Environ Sci Technol 53(13):7177–7179. https://doi.org/10.1021/acs.est.9b02942
Baumann M, Ismeier M (1998) Emissionen bei bestimmungsgemäßem Gebrauch von Reifen. In: KGK Kautschuk Gummi Kunststoffe, 51st year, No.3/98, 182-186 (in German)
Benckiser G (2019) Plastics, micro- and nanomaterials, and virus-soil microbe-plant interactions in the environment. In: Prasad R (ed) Plant nanobionics. Springer, pp 83–101. https://doi.org/10.1007/978-3-030-12496-0
Bergmann M, Mützel S, Primpke S, Tekman MB, Trachsel J, Gerdts G (2019) White and wonderful? Microplastics prevail in snow from the Alps to the Arctic. Sci Adv 5(8):eaax1157. https://doi.org/10.1126/sciadv.aax1157
Besseling E, Quik JTK, Sun M, Koelmans AA (2017) Fate of nano- and microplastic in freshwater systems: a modeling study. Environ Pollut 220:540–548. https://doi.org/10.1016/j.envpol.2016.10.001
Biginagwa FJ, Mayoma BS, Shashoua Y, Syberg K, Khan FR (2016) First evidence of microplastics in the African Great Lakes: recovery from Lake Victoria Nile perch and Nile tilapia. J Great Lakes Res 42(1):146–149. https://doi.org/10.1016/j.jglr.2015.10.012
Blettler MCM, Ulla MA, Rabuffetti AP, Garello N (2017) Plastic pollution in freshwater ecosystems: macro-, meso-, and microplastic debris in a floodplain lake. Environ Monit Assess 189(11):581. https://doi.org/10.1007/s10661-017-6305-8
Blettler MC, Abrial E, Khan FR, Sivri N, Espinola LA (2018) Freshwater plastic pollution: Recognizing research biases and identifying knowledge gaps. Water Res 143:416–424. https://doi.org/10.1016/j.watres.2018.06.015
Bordós G, Urbányi B, Micsinai A, Kriszt B, Palotai Z, Szabó I, Hantosi Z, Szoboszlay S (2019) Identification of microplastics in fish ponds and natural freshwater environments of the Carpathian basin, Europe. Chemosphere 216:110–116. https://doi.org/10.1016/j.chemosphere.2018.10.110
Boucher J, Friot D (2017) Primary microplastics in the oceans: A global evaluation of sources. IUCN International Union for Conservation of Nature. https://doi.org/10.2305/IUCN.CH.2017.01.en
Browne MA, Crump P, Niven SJ, Teuten E, Tonkin A, Galloway T, Thompson R (2011) Accumulation of microplastic on shorelines worldwide: sources and sinks. Environ Sci Technol 45(21):9175–9179. https://doi.org/10.1021/es201811s
Cai L, Wang J, Peng J, Tan Z, Zhan Z, Tan X, Chen Q (2017) Characteristic of microplastics in the atmospheric fallout from Dongguan city, China: preliminary research and first evidence. Environ Sci Pollut Res:1–8. https://doi.org/10.1007/s11356-017-0116-x
Camponelli KM, Casey RE, Snodgrass JW, Lev SM, Landa ER (2009) Impacts of weathered tire debris on the development of Rana sylvatica larvae. Chemosphere 74(5):717–722. https://doi.org/10.1016/j.chemosphere.2008.09.056
Carlin J, Craig C, Little S, Donnelly M, Fox D, Zhai L, Walters L (2020) Microplastic accumulation in the gastrointestinal tracts in birds of prey in central Florida, USA. Environ Pollut 264:114633. https://doi.org/10.1016/j.envpol.2020.114633
Carpenter EJ, Smith KL (1972) Plastics on the Sargasso Sea Surface. Science 175(4027):1240–1241. https://doi.org/10.1126/science.175.4027.1240
Carpenter EJ, Anderson SJ, Harvey GR, Miklas HP, Peck BB (1972) Polystyrene spherules in coastal waters. Science 178(4062):749–750. https://doi.org/10.1126/science.178.4062.749
Carr SA, Liu J, Tesoro AG (2016) Transport and fate of microplastic particles in wastewater treatment plants. Water Res 91:174–182. https://doi.org/10.1016/j.watres.2016.01.002
Castañeda RA, Avlijas S, Simard MA, Ricciardi A (2014) Microplastic pollution in St. Lawrence River sediments. Can J Fish Aquat Sci 71(12):1767–1771. https://doi.org/10.1139/cjfas-2014-0281
Chen X, Xiong X, Jiang X, Shi H, Wu C (2019) Sinking of floating plastic debris caused by biofilm development in a freshwater lake. Chemosphere 222:856–864. https://doi.org/10.1016/j.chemosphere.2019.02.015
Chen Y, Leng Y, Liu X, Wang J (2020) Microplastic pollution in vegetable farmlands of suburb Wuhan, central China. Environ Pollut 257:113449. https://doi.org/10.1016/j.envpol.2019.113449
Collard F, Gasperi J, Gilbert B, Eppe G, Azimi S, Rocher V, Tassin B (2018) Anthropogenic particles in the stomach contents and liver of the freshwater fish Squalius cephalus. Sci Total Environ 643:1257–1264. https://doi.org/10.1016/j.scitotenv.2018.06.313
Corcoran PL, Norris T, Ceccanese T, Walzak MJ, Helm PA, Marvin CH (2015) Hidden plastics of Lake Ontario, Canada and their potential preservation in the sediment record. Environ Pollut 204:17–25. https://doi.org/10.1016/j.envpol.2015.04.009
Corradini F, Meza P, Eguiluz R, Casado F, Huerta-Lwanga E, Geissen V (2019) Evidence of microplastic accumulation in agricultural soils from sewage sludge disposal. Sci Total Environ 671:411–420. https://doi.org/10.1016/j.scitotenv.2019.03.368
Dean BY, Corcoran PL, Helm PA (2018) Factors influencing microplastic abundances in nearshore, tributary and beach sediments along the Ontario shoreline of Lake Erie. J Great Lakes Res 44(5):1002–1009. https://doi.org/10.1016/j.jglr.2018.07.014
Dehghani S, Moore F, Akhbarizadeh R (2017) Microplastic pollution in deposited urban dust, Tehran metropolis, Iran. Environ Sci Pollut Res 24(25):20360–20371. https://doi.org/10.1007/s11356-017-9674-1
Dikareva N, Simon KS (2019) Microplastic pollution in streams spanning an urbanisation gradient. Environ Pollut 250:292–299. https://doi.org/10.1016/j.envpol.2019.03.105
Dong H, Liu T, Li Y, Liu H, Wang D (2013) Effects of plastic film residue on cotton yield and soil physical and chemical properties in Xinjiang. Trans Chinese Soc Agric Eng 29(8):91–99. https://doi.org/10.3969/j.issn.1002-6819.2013.08.011
Dong M, Luo Z, Jiang Q, Xing X, Zhang Q, Sun Y (2020) The rapid increases in microplastics in urban lake sediments. Sci Rep 10(1):1–10. https://doi.org/10.1038/s41598-020-57933-8
Dris R, Imhof H, Sanchez W, Gasperi J, Galgani F, Tassin B, Laforsch C (2015) Beyond the ocean: contamination of freshwater ecosystems with (micro-)plastic particles. Environ Chem 12(5):539–550. https://doi.org/10.1071/EN14172
Dris R, Gasperi J, Saad M, Mirande C, Tassin B (2016) Synthetic fibers in atmospheric fallout: a source of microplastics in the environment? Mar Pollut Bull 104(1):290–293. https://doi.org/10.1016/j.marpolbul.2016.01.006
Dris R, Gasperi J, Mirande C, Mandin C, Guerrouache M, Langlois V, Tassin B (2017) A first overview of textile fibers, including microplastics, in indoor and outdoor environments. Environ Pollut (Barking, Essex: 1987) 221:453–458. https://doi.org/10.1016/j.envpol.2016.12.013
Dris R, Gasperi J, Tassin B (2018) Sources and fate of microplastics in urban areas: a focus on Paris megacity. In: Freshwater microplastics. Springer, Cham, pp 69–83. https://doi.org/10.1007/978-3-319-61615-5_4
ECHA (2019) ANNEX XV RESTRICTION REPORT, PROPOSAL FOR A RESTRICTION: intentionally added microplastics, p 146. Retrieved from https://echa.europa.eu/documents/10162/05bd96e3-b969-0a7c-c6d0-441182893720
Eisentraut P, Dümichen E, Ruhl AS, Jekel M, Albrecht M, Gehde M, Braun U (2018) Two birds with one stone—fast and simultaneous analysis of microplastics: microparticles derived from thermoplastics and tire wear. Environ Sci Technol Lett 5(10):608–613. https://doi.org/10.1021/acs.estlett.8b00446
Eriksen M, Mason S, Wilson S, Box C, Zellers A, Edwards W et al (2013) Microplastic pollution in the surface waters of the Laurentian Great Lakes. Mar Pollut Bull 77(1):177–182. https://doi.org/10.1016/j.marpolbul.2013.10.007
Espí E, Salmerón A, Fontecha A, García Y, Real AI (2006) Plastic Films for Agricultural Applications. J Plastic Film Sheeting 22(2):85–102. https://doi.org/10.1177/8756087906064220
Estahbanati S, Fahrenfield NL (2016) Influence of wastewater treatment plant discharges on microplastic concentrations in surface water. Chemosphere 162:277–284. https://doi.org/10.1016/j.chemosphere.2016.07.083
Evans-Pughe C (2017) All at sea: cleaning up the great Pacific garbage patch. Eng Technol 12(1):52–55. https://doi.org/10.1049/et.2017.0105
Fan Y, Zheng K, Zhu Z, Chen G, Peng X (2019) Distribution, sedimentary record, and persistence of microplastics in the Pearl River catchment, China. Environ Pollut 251:862–870. https://doi.org/10.1016/j.envpol.2019.05.056
Faure F, Corbaz M, Baecher H, Alencastro D, Felippe L (2012) Pollution due to plastics and microplastics in Lake Geneva and in the Mediterranean Sea. Arch Sci 65:157–164. Retrieved from https://pdfs.semanticscholar.org/5744/79e4e81161ff2d93615a2af7a3e83432035e.pdf
Fischer EK, Paglialonga L, Czech E, Tamminga M (2016) Microplastic pollution in lakes and lake shoreline sediments–a case study on Lake Bolsena and Lake Chiusi (central Italy). Environ Pollut 213:648–657. https://doi.org/10.1016/j.envpol.2016.03.012
França D, Messa LL, Souza CF, Faez R (2019) Nano and microencapsulated nutrients for enhanced efficiency fertilizer. In: Gutiérrez TJ (ed) Polymers for agri-food applications. Springer, Cham, pp 29–44. https://doi.org/10.1007/978-3-030-19416-1_3
Free CM, Jensen OP, Mason SA, Eriksen M, Williamson NJ, Boldgiv B (2014) High-levels of microplastic pollution in a large, remote, mountain lake. Mar Pollut Bull 85(1):156–163. https://doi.org/10.1016/j.marpolbul.2014.06.001
Frias JPGL, Pagter E, Nash R, O’Connor I, Carretero O, Filgueiras A et al (2018) Standardised protocol for monitoring microplastics in sediments. https://doi.org/10.13140/RG.2.2.36256.89601/1
Fuller S, Gautam A (2016) A procedure for measuring microplastics using pressurized fluid extraction. Environ Sci Technol 50(11):5774–5780. https://doi.org/10.1021/acs.est.6b00816
Gatidou G, Arvanti OS, Stasinakis AS (2019) Review on the occurrence and fate of microplastics in Sewage Treatment Plants. J Hazard Mater 367:504–512. https://doi.org/10.1016/j.jhazmat.2018.12.081
GESAMP (2015) Sources, fate and effects of microplastics in the marine environment (part 1), No. 2015 #90. IMO, p 98. Retrieved from: http://www.gesamp.org/publications/reports-and-studies-no-90
GESAMP (2016) Sources, fate and effects of microplastics in the marine environment (part 2), No. 2016 #93. IMO, p 221. Retrieved from: http://www.gesamp.org/publications/microplastics-in-the-marine-environment-part-2
GESAMP (2019) Guidelines for the monitoring and assessment of plastic litter in the ocean, No. 2019 #99. IMO, p 123. Retrieved from: http://www.gesamp.org/publications/guidelines-for-the-monitoring-and-assessment-of-plastic-litter-in-the-ocean
Geyer R, Jambeck JR, Law KL (2017) Production, use, and fate of all plastics ever made. Sci Adv 3(7):e1700782. https://doi.org/10.1126/sciadv.1700782
Grung M, Kringstad A, Bæk K, Allan IJ, Thomas KV, Meland S, Ranneklev SB (2017) Identification of non-regulated polycyclic aromatic compounds and other markers of urban pollution in road tunnel particulate matter. J Hazard Mater 323:36–44. https://doi.org/10.1016/j.jhazmat.2016.05.036
Gualtieri M, Andrioletti M, Vismara C, Milani M, Camatini M (2005a) Toxicity of tire debris leachates. Environ Int 31(5):723–730. https://doi.org/10.1016/j.envint.2005.02.001
Gualtieri M, Andrioletti M, Mantecca P, Vismara C, Camatini M (2005b) Impact of tire debris on in vitro and in vivo systems. Part Fibre Toxicol 2(1):1. https://doi.org/10.1186/1743-8977-2-1
Gündoğdu S, Cevik C, Ayat B, Aydoğan B, Karaca S (2018) How microplastics quantities increase with flood events? An example from Mersin Bay NE Levantine coast of Turkey. Environ Pollut 239:342–350. https://doi.org/10.1016/j.envpol.2018.04.042
Hale RC, Seeley ME, La Guardia MJ, Mai L, Zeng EY (2020) A global perspective on microplastics. J Geophys Res Oceans 125(1):5, e2018JC014719. https://doi.org/10.1029/2018JC014719
Hallberg M, Renman G, Byman L, Svenstam G, Norling M (2014) Treatment of tunnel wash water and implications for its disposal. Water Sci Technol J Int Assoc Water Pollut Res 69(10):2029–2035. https://doi.org/10.2166/wst.2014.113
He P, Chen L, Shao L, Zhang H, Lü F (2019) Municipal solid waste (MSW) landfill: a source of microplastics? -Evidence of microplastics in landfill leachate. Water Res 159:38–45. https://doi.org/10.1016/j.watres.2019.04.060
Hendrickson E, Minor EC, Schreiner K (2018) Microplastic abundance and composition in western Lake Superior as determined via microscopy, Pyr-GC/MS, and FTIR. Environ Sci Technol 52(4):1787–1796. https://doi.org/10.1021/acs.est.7b05829
Holland ER, Mallory ML, Shutler D (2016) Plastics and other anthropogenic debris in freshwater birds from Canada. Sci Total Environ 571:251–258. https://doi.org/10.1016/j.scitotenv.2016.07.158
Horton AA, Svendsen C, Williams RJ, Spurgeon DJ, Lahive E (2017a) Large microplastic particles in sediments of tributaries of the River Thames, UK – Abundance, sources and methods for effective quantification. Mar Pollut Bull 114(1):218–226. https://doi.org/10.1016/j.marpolbul.2016.09.004
Horton AA, Walton A, Spurgeon DJ, Lahive E, Svendsen C (2017b) Microplastics in freshwater and terrestrial environments: evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci Total Environ 586:127–141. https://doi.org/10.1016/j.scitotenv.2017.01.190
Horton AA, Jürgens MD, Lahive E, van Bodegom PM, Vijver MG (2018) The influence of exposure and physiology on microplastic ingestion by the freshwater fish Rutilus rutilus (roach) in the River Thames, UK. Environ Pollut 236:188–194. https://doi.org/10.1016/j.envpol.2018.01.044
Huang Y, Liu Q, Jia W, Yan C, Wang J (2020) Agricultural plastic mulching as a source of microplastics in the terrestrial environment. Environ Pollut 114096. https://doi.org/10.1016/j.envpol.2020.114096
Huerta Lwanga E, Gertsen H, Gooren H, Peters P, Salánki T, van der Ploeg M et al (2017) Incorporation of microplastics from litter into burrows of Lumbricus terrestris. Environ Pollut 220:523–531. https://doi.org/10.1016/j.envpol.2016.09.096
Hurley RR, Nizzetto L (2018) Fate and occurrence of micro(nano)plastics in soils: knowledge gaps and possible risks. Curr Opin Environ Sci Health 1:6–11. https://doi.org/10.1016/j.coesh.2017.10.006
Hurley RR, Woodward JC, Rothwell JJ (2017) Ingestion of microplastics by freshwater Tubifex worms. Environ Sci Technol 51(21):12844–12851. https://doi.org/10.1021/acs.est.7b03567
Hurley RR, Woodward JC, Rothwell JJ (2018) Microplastic contamination of river beds significantly reduced by catchment-wide flooding. Nat Geosci. https://doi.org/10.1038/s41561-018-0080-1
Imhof HK, Ivleva NP, Schmid J, Niessner R, Laforsch C (2013) Contamination of beach sediments of a subalpine lake with microplastic particles. Curr Biol 23(19):R867–R868. https://doi.org/10.1016/j.cub.2013.09.001
Imhof HK, Wiesheu AC, Anger PM, Niessner R, Ivleva NP, Laforsch C (2018) Variation in plastic abundance at different lake beach zones-A case study. Sci Total Environ 613:530–537
Isobe A, Buenaventura NT, Chastain S, Chavanich S, Cózar A, DeLorenzo M et al (2019) An interlaboratory comparison exercise for the determination of microplastics in standard sample bottles. Mar Pollut Bull 146:831–837. https://doi.org/10.1016/j.marpolbul.2019.07.033
Jabeen K, Su L, Li J, Yang D, Tong C, Mu J, Shi H (2017) Microplastics and mesoplastics in fish from coastal and fresh waters of China. Environ Pollut 221:141–149. https://doi.org/10.1016/j.envpol.2016.11.055
Jambeck JR, Geyer R, Wilcox C, Siegler TR, Perryman M, Andrady A et al (2015) Plastic waste inputs from land into the ocean. Science 347(6223):768–771. https://doi.org/10.1126/science.1260352
Jiang C, Yin L, Li Z, Wen X, Luo X, Hu S et al (2019) Microplastic pollution in the rivers of the Tibet Plateau. Environ Pollut 249:91–98. https://doi.org/10.1016/j.envpol.2019.03.022
Johnsen JP, Bye NH (2019) Assessment of tire wear emission in a road tunnel, using benzothiazoles as tracer in tunnel wash water 79. Retrieved from https://nmbu.brage.unit.no/nmbu-xmlui/handle/11250/2612289
Kamata H, Masuda K, Yamada J, Takada H (2000) Water-Particle distribution of hydrophobic micro pollutants in storm water runoff. Polycycl Aromat Compd 20(1–4):39–54. https://doi.org/10.1080/10406630008034774
Kay P, Hiscoe R, Moberley I, Bajic L, McKenna N (2018) Wastewater treatment plants as a source of microplastics in river catchments. Environ Sci Pollut Res 25(20):20264–20267. https://doi.org/10.1007/s11356-018-2070-7
Keller AS, Jimenez-Martinez J, Mitrano DM (2020) Transport of nano- and Microplastic through unsaturated porous media from sewage sludge application. Environ Sci Technol 54(2):911–920. https://doi.org/10.1021/acs.est.9b06483
Khan FR, Halle LL, Palmqvist A (2019) Acute and long-term toxicity of micronized car tire wear particles to Hyalella azteca. Aquat Toxicol 213:105216. https://doi.org/10.1016/j.aquatox.2019.05.018
Klein M, Fischer EK (2019) Microplastic abundance in atmospheric deposition within the Metropolitan area of Hamburg, Germany. Sci Total Environ 685:96–103. https://doi.org/10.1016/j.scitotenv.2019.05.405
Klöckner P, Reemtsma T, Eisentraut P, Braun U, Ruhl AS, Wagner S (2019) Tire and road wear particles in road environment – Quantification and assessment of particle dynamics by Zn determination after density separation. Chemosphere 222:714–721. https://doi.org/10.1016/j.chemosphere.2019.01.176
Koelmans AA, Mohamed Nor NH, Hermsen E, Kooi M, Mintenig SM, De France J (2019) Microplastics in freshwaters and drinking water: critical review and assessment of data quality. Water Res 155:410–422. https://doi.org/10.1016/j.watres.2019.02.054
Kole PJ, Löhr AJ, Van Belleghem FGAJ, Ragas AMJ (2017) Wear and tear of tyres: a stealthy source of microplastics in the environment. Int J Environ Res Public Health 14(10):1265. https://doi.org/10.3390/ijerph14101265
Kum V, Sharp A, Harnpornchai N (2005) Improving the solid waste management in Phnom Penh city: a strategic approach. Waste Manag 25(1):101–109. https://doi.org/10.1016/j.wasman.2004.09.004
Lambert S, Wagner M (2018) Microplastics are contaminants of emerging concern in freshwater environments: an overview. In: Freshwater microplastics. Springer, Cham, pp 1–23. https://doi.org/10.1007/978-3-319-61615-5_1
Lassen, C., Hansen, S. F., Magnusson, K., Hartmann, N. B., Jensen, P. R., Nielsen, T. G., & Brinch, A. (2015). Microplastics: occurrence, effects and sources of releases to the environment in Denmark. Danish Environmental Protection Agency. Retrieved from https://orbit.dtu.dk/en/publications/microplastics-occurrence-effects-and-sources-of-releases-to-the-e
Lebreton LCM, van der Zwet J, Damsteeg J-W, Slat B, Andrady A, Reisser J (2017) River plastic emissions to the world’s oceans. Nat Commun 8:15611. https://doi.org/10.1038/ncomms15611
Lecce SA, Pavlowsky RT (1997) Storage of mining-related zinc in floodplain sediments, Blue River, Wisconsin. Phys Geogr 18(5):424–439. https://doi.org/10.1080/02723646.1997.10642628
Lee J, Hong S, Song YK, Hong SH, Jang YC, Jang M et al (2013) Relationships among the abundances of plastic debris in different size classes on beaches in South Korea. Mar Pollut Bull 77(1–2):349–354. https://doi.org/10.1016/j.marpolbul.2013.08.013
Lenaker PL, Baldwin AK, Corsi SR, Mason SA, Reneau PC, Scott JW (2019) Vertical distribution of microplastics in the water column and surficial sediment from the Milwaukee River Basin to Lake Michigan. Environ Sci Technol. https://doi.org/10.1021/acs.est.9b03850
Li X, Chen L, Mei Q, Dong B, Dai X, Ding G, Zeng EY (2018) Microplastics in sewage sludge from the wastewater treatment plants in China. Water Res 142:75–85. https://doi.org/10.1016/j.watres.2018.05.034
Li C, Busquets R, Campos LC (2020) Assessment of microplastics in freshwater systems: a review. Sci Total Environ 707:135578. https://doi.org/10.1016/j.scitotenv.2019.135578
Liu EK, He WQ, Yan CR (2014) ‘White revolution’ to ‘white pollution’—agricultural plastic film mulch in China. Environ Res Lett 9(9):091001. https://doi.org/10.1088/1748-9326/9/9/091001
Liu M, Lu S, Song Y, Lei L, Hu J, Lv W et al (2018) Microplastic and mesoplastic pollution in farmland soils in suburbs of Shanghai, China. Environ Pollut 242:855–862. https://doi.org/10.1016/j.envpol.2018.07.051
Liu F, Vianello A, Vollertsen J (2019a) Retention of microplastics in sediments of urban and highway stormwater retention ponds. Environ Pollut 113335. https://doi.org/10.1016/j.envpol.2019.113335
Liu K, Wu T, Wang X, Song Z, Zong C, Wei N, Li D (2019b) Consistent transport of terrestrial microplastics to the ocean through atmosphere. Environ Sci Technol 53(18):10612–10619. https://doi.org/10.1021/acs.est.9b03427
Liu S, Jian M, Zhou L, Li W (2019c) Distribution and characteristics of microplastics in the sediments of Poyang Lake, China. Water Sci Technol 79(10):1868–1877. https://doi.org/10.2166/wst.2019.185
Liu X, Yuan W, Di M, Li Z, Wang J (2019d) Transfer and fate of microplastics during the conventional activated sludge process in one wastewater treatment plant of China. Chem Eng J 362:176–182. https://doi.org/10.1016/j.cej.2019.01.033
Liu K, Wang X, Song Z, Wei N, Li D (2020a) Terrestrial plants as a potential temporary sink of atmospheric microplastics during transport. Sci Total Environ 742:140523. https://doi.org/10.1016/j.scitotenv.2020.140523
Liu K, Wang X, Song Z, Wei N, Ye H, Cong, X. … Li, D. (2020b) Global inventory of atmospheric fibrous microplastics input into the ocean: an implication from the indoor origin. J Hazard Mater 400:123223. https://doi.org/10.1016/j.jhazmat.2020.123223
Lorenz K, Kandeler E (2005) Biochemical characterization of urban soil profiles from Stuttgart, Germany. Soil Biol Biochem 37(7):1373–1385. https://doi.org/10.1016/j.soilbio.2004.12.009
Lusher AL, Hurley RR, Vogelsang C, Nizzetto L, Olsen M (2018) Mapping microplastics in sludge, No. M907, p 55. Retrieved from http://www.miljodirektoratet.no/Documents/publikasjoner/M907/M907.pdf
Lusher AL, Hurley RR, Vogelsang C (2019) Microplastics in sewage sludge: captured but released? In: Karapanagioti HK, Kalavrouziotis IK (eds) Microplastics in water and wastewater. IWA Publishing, London, pp 85–100
Lv W, Zhou W, Lu S, Huang W, Yuan Q, Tian M et al (2019a) Microplastic pollution in rice-fish co-culture system: a report of three farmland stations in Shanghai, China. Sci Total Environ 652:1209–1218. https://doi.org/10.1016/j.scitotenv.2018.10.321
Lv X, Dong Q, Zuo Z, Liu Y, Huang X, Wu W-M (2019b) Microplastics in a municipal wastewater treatment plant: fate, dynamic distribution, removal efficiencies, and control strategies. Retrieved from https://pubag.nal.usda.gov/catalog/6367168
Maaß S, Daphi D, Lehmann A, Rillig MC (2017) Transport of microplastics by two collembolan species. Environ Pollut 225:456–459. https://doi.org/10.1016/j.envpol.2017.03.009
Magnusson K, Norén F (2014) Screening of microplastic particles in and down-stream a wastewater treatment plant, No. C 55, p 22. Retrieved from http://www.diva-portal.org/smash/record.jsf?pid=diva2:773505
Magnusson K, Eliasson K, Fråne A, Haikonen K, Hultén J, Olshammar M et al (2016) Swedish sources and pathways for microplastics to the marine environment. A Review of Existing Data IVL, C, 183. Retrieved from https://www.naturvardsverket.se/upload/miljoarbete-i-samhallet/miljoarbete-i-sverige/regeringsuppdrag/2016/mikroplaster/swedish-sources-and-pathways-for-microplastics-to-marine%20environment-ivl-c183.pdf
Mahon AM, O’Connell B, Healy MG, O’Connor I, Officer R, Nash R, Morrison L (2017) Microplastics in sewage sludge: effects of treatment. Environ Sci Technol 51(2):810–818. https://doi.org/10.1021/acs.est.6b04048
Mai L, Bao L-J, Wong CS, Zeng EY (2018) Microplastics in the terrestrial environment. In: Zeng EY (ed) Microplastic contamination in aquatic environments; an emerging matter of environmental urgency. Elsevier, pp 365–378. https://doi.org/10.1016/B978-0-12-813747-5.00012-6
Mai L, You S-N, He H, Bao L-J, Liu L, Zeng EY (2019) Riverine microplastic pollution in the Pearl River Delta, China: are modeled estimates accurate? Environ Sci Technol. https://doi.org/10.1021/acs.est.9b04838
Mani T, Hauk A, Walter U, Burkhardt-Holm P (2015) Microplastics profile along the Rhine River. Sci Rep 5:17988. https://doi.org/10.1038/srep17988
Mantecca P, Gualtieri M, Andrioletti M, Bacchetta R, Vismara C, Vailati G, Camatini M (2007) Tire debris organic extract affects Xenopus development. Environ Int 33(5):642–648. https://doi.org/10.1016/j.envint.2007.01.007
Marais M, Armitage N, Pithey S (2001) A study of the litter loadings in urban drainage systems – methodology and objectives. Water Sci Technol 44(6):99–108. https://doi.org/10.2166/wst.2001.0350
Marais M, Armitage N, Wise C (2004) The measurement and reduction of urban litter entering stormwater drainage systems: paper 1 – quantifying the problem using the City of Cape Town as a case study [Text]. Retrieved February 24, 2020, from https://www.ingentaconnect.com/content/sabinet/waters/2004/00000030/00000004/art00006
Mason SA, Garneau D, Sutton R, Chu Y, Ehmann K, Barnes J et al (2016a) Microplastic pollution is widely detected in US municipal wastewater treatment plant effluent. Environ Pollut 218:1045–1054. https://doi.org/10.1016/j.envpol.2016.08.056
Mason SA, Kammin L, Eriksen M, Aleid G, Wilson S, Box C, Williamson N, Riley A (2016b) Pelagic plastic pollution within the surface waters of Lake Michigan, USA. J Great Lakes Res 42(4):753–759. https://doi.org/10.1016/j.jglr.2016.05.009
McCormick AR, Hoellein TJ, London MG, Hittie J, Scott JW, Kelly JJ (2016) Microplastic in surface waters of urban rivers: concentration, sources, and associated bacterial assemblages. Ecosphere 7(11):e01556. https://doi.org/10.1002/ecs2.1556
McGoran AR, Clark PF, Morritt D (2017) Presence of microplastic in the digestive tracts of European flounder, Platichthys flesus, and European smelt, Osmerus eperlanus, from the River Thames. Environ Pollut 220:744–751. https://doi.org/10.1016/j.envpol.2016.09.078
Mehlhart G, Blepp M (2012) Study on land-sourced litter (LSL) in the marine environment: review of sources and literature. Öko-Institut. https://www.oeko.de/oekodoc/1487/2012-058-en.pdf
Meland S, Rødland E (2018) Forurensning i tunnelvaskevann –en studie av 34 veitunneler i Norge. VANN 1:54–65. Retrieved from: https://vannforeningen.no/wp-content/uploads/2018/07/Meland.pdf
Meland S, Borgstrøm R, Heier LS, Rosseland BO, Lindholm O, Salbu B (2010a) Chemical and ecological effects of contaminated tunnel wash water runoff to a small Norwegian stream. Sci Total Environ 408(19):4107–4117. https://doi.org/10.1016/j.scitotenv.2010.05.034
Meland S, Heier LS, Salbu B, Tollefsen KE, Farmen E, Rosseland BO (2010b) Exposure of brown trout (Salmo trutta L.) to tunnel wash water runoff—chemical characterisation and biological impact. Sci Total Environ 408(13):2646–2656. https://doi.org/10.1016/j.scitotenv.2010.03.025
Migwi FK, Ogunah JA, Kiratu JM (2020) Occurrence and spatial distribution of microplastics in the surface waters of Lake Naivasha, Kenya. Environ Toxicol Chem n/a(n/a). https://doi.org/10.1002/etc.4677
Muñoz-Cadena CE, Lina-Manjarrez P, Estrada-Izquierdo I, Ramón-Gallegos E (2012) An approach to litter generation and littering practices in a Mexico City neighborhood. Sustainability 4(8):1733–1754. https://doi.org/10.3390/su4081733
Nan B, Su L, Kellar C, Craig NJ, Keough MJ, Pettigrove V (2020) Identification of microplastics in surface water and Australian freshwater shrimp Paratya australiensis in Victoria, Australia. Environ Pollut 259:113865. https://doi.org/10.1016/j.envpol.2019.113865
Nel HA, Dalu T, Wasserman RJ (2018) Sinks and sources: assessing microplastic abundance in river sediment and deposit feeders in an Austral temperate urban river system. Sci Total Environ 612:950–956. https://doi.org/10.1016/j.scitotenv.2017.08.298
Ngo PL, Pramanik BK, Shah K, Roychand R (2019) Pathway, classification and removal efficiency of microplastics in wastewater treatment plants. Environ Pollut 255(2):113326. https://doi.org/10.1016/j.envpol.2019.113326
Nizzetto L, Futter M, Langaas S (2016) Are agricultural Soils dumps for microplastics of urban origin? Environ Sci Technol 50(20):10777–10779. https://doi.org/10.1021/acs.est.6b04140
O’Connor D, Pan S, Shen Z, Song Y, Jin Y, Wu W-M, Hou D (2019) Microplastics undergo accelerated vertical migration in sand soil due to small size and wet-dry cycles. Environ Pollut. https://doi.org/10.1016/j.envpol.2019.03.092
Ockelford A, Cundy A, Ebdon JE (2020) Storm response of fluvial sedimentary microplastics. Sci Rep 10(1):1–10. https://doi.org/10.1038/s41598-020-58765-2
Olesen KB, Stephansen DA, van Alst N, Vollertsen J (2019) Microplastics in a stormwater pond. Water 11(7):1466. https://doi.org/10.3390/w11071466
Panebianco A, Nalbone L, Giarratana F, Ziino G (2019) First discoveries of microplastics in terrestrial snails. Food Control 106:106722. https://doi.org/10.1016/j.foodcont.2019.106722
Panko JM, Kreider ML, McAtee BL, Marwood C (2013) Chronic toxicity of tire and road wear particles to water- and sediment-dwelling organisms. Ecotoxicology 22(1):13–21. https://doi.org/10.1007/s10646-012-0998-9
Phillips MB, Bonner TH (2015) Occurrence and amount of microplastic ingested by fishes in watersheds of the Gulf of Mexico. Mar Pollut Bull 100(1):264–269. https://doi.org/10.1016/j.marpolbul.2015.08.041
Piehl S, Leibner A, Löder MGJ, Dris R, Bogner C, Laforsch C (2018) Identification and quantification of macro- and microplastics on an agricultural farmland. Sci Rep 8(1):17950. https://doi.org/10.1038/s41598-018-36172-y
Qin Y, Wang Z, Li W, Chang X, Yang J, Yang F (2019) Microplastics in the sediment of Lake Ulansuhai of Yellow River Basin, China. Water Environ Res 92(6). https://doi.org/10.1002/wer.1275
Ranneklev SB, Hurley R, Bråte ILN, Vogelsang C (2019) Plast i landbruket: kilder, massebalanse og spredning til lokale vannforekomster (Plastland). Norsk insitutt for vannforskning. Retrieved from https://niva.brage.unit.no/niva-xmlui/handle/11250/2632595
Raupach MR, Lu H (2004) Representation of land-surface processes in aeolian transport models. Environ Model Softw 19(2):93–112. https://doi.org/10.1016/S1364-8152(03)00113-0
Redondo-Hasselerharm PE, de Ruijter VN, Mintenig SM, Verschoor A, Koelmans AA (2018) Ingestion and chronic effects of car tire tread particles on freshwater benthic macroinvertebrates. Environ Sci Technol 52(23):13986–13994. https://doi.org/10.1021/acs.est.8b05035
Rezaei M, Riksen MJ, Sirjani E, Sameni A, Geissen V (2019) Wind erosion as a driver for transport of light density microplastics. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2019.02.382
Roch S, Walter T, Ittner LD, Friedrich C, Brinker A (2019) A systematic study of the microplastic burden in freshwater fishes of south-western Germany – Are we searching at the right scale? Sci Total Environ 689:1001–1011. https://doi.org/10.1016/j.scitotenv.2019.06.404
Rochman CM (2018) Microplastics research—from sink to source. Science 360(6384):28–29. https://doi.org/10.1126/science.aar7734
Rødland E (2019) Ecotoxic potential of road-associated microplastic particles (RAMP). VANN 3:166–183. Retrieved from: https://vannforeningen.no/wp-content/uploads/2019/12/R%C3%B8dland.pdf
Rødland E, Helgadottir D (2018) Prioriteringsverktøy for vurdering av forurensingsbelastning fra urenset tunnelvaskevann. VANN 4:367–376. Retrieved from: https://vannforeningen.no/wp-content/uploads/2019/04/R%C3%B8dland.pdf
Rødland E, Okoffo ED, Rauert C, Heier LS, Lind OC, Reid M, Thomas KV, Meland S (2020) Road de-icing salt: assessment of a potential new source and pathway of microplastics particles from roads. Sci Total Environ 738:139352. https://doi.org/10.1016/j.scitotenv.2020.139352
Rodrigues MO, Abrantes N, Gonçalves FJM, Nogueira H, Marques JC, Gonçalves AMM (2018) Spatial and temporal distribution of microplastics in water and sediments of a freshwater system (Antuã River, Portugal). Sci Total Environ 633:1549–1559. https://doi.org/10.1016/j.scitotenv.2018.03.233
Ruan Y, Zhang K, Wu C, Wu R, Lam PKS (2019) A preliminary screening of HBCD enantiomers transported by microplastics in wastewater treatment plants. Sci Total Environ 674:171–178. https://doi.org/10.1016/j.scitotenv.2019.04.007
Sanchez W, Bender C, Porcher J-M (2014) Wild gudgeons (Gobio gobio) from French rivers are contaminated by microplastics: Preliminary study and first evidence. Environ Res 128:98–100. https://doi.org/10.1016/j.envres.2013.11.004
Scarascia-Mugnozza G, Sica C, Russo G (2012) Plastic materials in European agriculture: actual use and perspectives. J Agric Eng 42(3):15–28. https://doi.org/10.4081/jae.2011.3.15
Scherer C, Brennholt N, Reifferscheid G, Wagner M (2017) Feeding type and development drive the ingestion of microplastics by freshwater invertebrates. Sci Rep 7(1):17006. https://doi.org/10.1038/s41598-017-17191-7
Scherer C, Weber A, Lambert S, Wagner M (2018) Interactions of microplastics with freshwater biota. In: Freshwater microplastics. Springer, Cham, pp 153–180. https://doi.org/10.1007/978-3-319-61615-5_8
Scherer C, Weber A, Stock F, Vurusic S, Egerci H, Kochleus, C. … Reifferscheid, G. (2020) Comparative assessment of microplastics in water and sediment of a large European river. Sci Total Environ 738:139866. https://doi.org/10.1016/j.scitotenv.2020.139866
Scheurer M, Bigalke M (2018) Microplastics in Swiss floodplain soils. Environ Sci Technol 52(6):3591–3598. https://doi.org/10.1021/acs.est.7b06003
Schmidt C, Krauth T, Wagner S (2017) Export of plastic debris by rivers into the sea. Environ Sci Technol 51(21):12246–12253. https://doi.org/10.1021/acs.est.7b02368
Scopetani C, Chelazzi D, Cincinelli A, Esterhuizen-Londt M (2019) Assessment of microplastic pollution: occurrence and characterisation in Vesijärvi lake and Pikku Vesijärvi pond, Finland. Environ Monit Assess 191(11):652. https://doi.org/10.1007/s10661-019-7843-z
Shiber JG (1979) Plastic pellets on the coast of Lebanon. Mar Pollut Bull 10(1):28–30. https://doi.org/10.1016/0025-326X(79)90321-7
Shruti VC, Jonathan MP, Rodriguez-Espinosa PF, Rodríguez-González F (2019) Microplastics in freshwater sediments of Atoyac River basin, Puebla City, Mexico. Sci Total Environ 654:154–163. https://doi.org/10.1016/j.scitotenv.2018.11.054
Siegfried M, Koelmans AA, Besseling E, Kroeze C (2017) Export of microplastics from land to sea. A modelling approach. Water Res 127(Supplement C):249–257. https://doi.org/10.1016/j.watres.2017.10.011
Sighicelli M, Pietrelli L, Lecce F, Iannilli V, Falconieri M, Coscia L et al (2018) Microplastic pollution in the surface waters of Italian Subalpine Lakes. Environ Pollut 236:645–651. https://doi.org/10.1016/j.envpol.2018.02.008
Silva-Cavalcanti JS, Silva JDB, de França EJ, de Araújo MCB, Gusmão F (2017) Microplastics ingestion by a common tropical freshwater fishing resource. Environ Pollut 221:218–226. https://doi.org/10.1016/j.envpol.2016.11.068
Simon M, van Alst N, Vollertsen J (2018) Quantification of microplastic mass and removal rates at wastewater treatment plants applying Focal Plane Array (FPA)-based Fourier Transform Infrared (FT-IR) imaging. Water Res 142:1–9. https://doi.org/10.1016/j.watres.2018.05.019
Sinha T, Bhagwatwar P, Krishnamoorthy C, Chidambaram R (2019) Polymer based micro- and nanoencapsulation of agrochemicals. In: Gutiérrez TJ (ed) Polymers for agri-food applications. Springer, Cham, pp 5–28. https://doi.org/10.1007/978-3-030-19416-1_2
Sommer F, Dietze V, Baum A, Sauer J, Gilge S, Maschowski C, Gieré R (2018) Tire abrasion as a major source of microplastics in the environment. Aerosol Air Qual Res 18(8):2014–2028. https://doi.org/10.4209/aaqr.2018.03.0099
Sruthy S, Ramasamy EV (2017) Microplastic pollution in Vembanad Lake, Kerala, India: the first report of microplastics in lake and estuarine sediments in India. Environ Pollut 222:315–322. https://doi.org/10.1016/j.envpol.2016.12.038
Stanton T, Johnson M, Nathanail P, MacNaughton M, Gomes RL (2020) Freshwater microplastic concentrations vary through both space and time. Environ Pollut 263(B):114481. https://doi.org/10.1016/j.envpol.2020.114481
Steinmetz Z, Wollmann C, Schaefer M, Buchmann C, David J, Tröger J et al (2016) Plastic mulching in agriculture. Trading short-term agronomic benefits for long-term soil degradation? Sci Total Environ 550:690–705. https://doi.org/10.1016/j.scitotenv.2016.01.153
Su L, Xue Y, Li L, Yang D, Kolandhasamy P, Li D, Shi H (2016) Microplastics in Taihu Lake, China. Environ Pollut 216(Supplement C):711–719. https://doi.org/10.1016/j.envpol.2016.06.036
Su L, Cai H, Kolandhasamy P, Wu C, Rochman CM, Shi H (2018) Using the Asian clam as an indicator of microplastic pollution in freshwater ecosystems. Environ Pollut 234:347–355. https://doi.org/10.1016/j.envpol.2017.11.075
Su Y, Zhang Z, Wu D, Zhan L, Shi H, Xie B (2019) Occurrence of microplastics in landfill systems and their fate with landfill age. Water Res 164:114968. https://doi.org/10.1016/j.watres.2019.114968
Sun J, Dai X, Wang Q, van Loosdrecht MCM, Ni B-J (2019) Microplastics in wastewater treatment plants: detection, occurrence and removal. Water Res 152:21–37. https://doi.org/10.1016/j.watres.2018.12.050
Sundt P, Schulze P-E, Syversen F (2014) Sources of microplastics pollution to the marine environment, No. M-321 | 2015. MEPEX fpr the Norwegian Environment Agency, p 86. Retrieved from http://www.miljodirektoratet.no/Documents/publikasjoner/M321/M321.pdf
Sundt P, Syversen F, Skogesal O, Schulze P-E (2016) Primary microplasticpollution: measures and reduction potentials in Norway, No. M-545. Norwegian Environment Agency, p 117. Retrieved from http://www.miljodirektoratet.no/Documents/publikasjoner/M545/M545.pdf
Townsend AK, Barker CM (2014) Plastic and the nest entanglement of urban and agricultural crows. PLoS One 9(1):e88006. https://doi.org/10.1371/journal.pone.0088006
Turner S, Horton AA, Rose NL, Hall C (2019) A temporal sediment record of microplastics in an urban lake, London. UK J Paleolimnol:1–14. https://doi.org/10.1007/s10933-019-00071-7
UNEP (2009) Marine litter: a global challenge. Regional Seas, United Nations Environment Programme, Nairobi. Retrieved from: http://wedocs.unep.org/handle/20.500.11822/7787
UNESCO (2017) Waste water: the untapped resources. The United Nations World Water Development Report. Retrieved from: https://www.unido.org/sites/default/files/2017-03/UN_World_Water_Development_Report_-_Full_0.pdf
Unice KM, Kreider ML, Panko JM (2013) Comparison of tire and road wear particle concentrations in sediment for watersheds in France, Japan, and the United States by quantitative pyrolysis GC/MS Analysis. Environ Sci Technol 47(15):8138–8147. https://doi.org/10.1021/es400871j
Uurasjärvi E, Hartikainen S, Setälä O, Lehtiniemi M, Koistinen A (2020) Microplastic concentrations, size distribution, and polymer types in the surface waters of a northern European lake. Water Environ Res 92(1):149–156. https://doi.org/10.1002/wer.1229
van den Berg P, Huerta-Lwanga E, Corradini F, Geissen V (2020) Sewage sludge application as a vehicle for microplastics in eastern Spanish agricultural soils. Environ Pollut 261:114198. https://doi.org/10.1016/j.envpol.2020.114198
van Emmerik T, Loozen M, van Oeveren K, Buschman F, Prinsen G (2019) Riverine plastic emission from Jakarta into the ocean. Environ Res Lett 14(8):084033. https://doi.org/10.1088/1748-9326/ab30e8
van Wijnen J, Ragas AMJ, Kroeze C (2019) Modelling global river export of microplastics to the marine environment: Sources and future trends. Sci Total Environ 673:392–401. https://doi.org/10.1016/j.scitotenv.2019.04.078
Vaughan R, Turner SD, Rose NL (2017) Microplastics in the sediments of a UK urban lake. Environ Pollut 229:10–18. https://doi.org/10.1016/j.envpol.2017.05.057
Veerasingam S, Mugilarasan M, Venkatachalapathy R, Vethamony P (2016) Influence of 2015 flood on the distribution and occurrence of microplastic pellets along the Chennai coast, India. Mar Pollut Bull 109(1):196–204. https://doi.org/10.1016/j.marpolbul.2016.05.082
Vegkart (2020) Road statistics from the road map applications Vegkart https://vegkart.atlas.vegvesen.no/. Accessed 8th May 2020
Vermaire JC, Pomeroy C, Herczegh SM, Haggart O, Murphy M (2017) Microplastic abundance and distribution in the open water and sediment of the Ottawa River, Canada, and its tributaries. FACETS 2:201–314. https://doi.org/10.1139/facets-2016-0070
Villena OC, Terry I, Iwata K, Landa ER, LaDeau SL, Leisnham PT (2017) Effects of tire leachate on the invasive mosquito Aedes albopictus and the native congener Aedes triseriatus. PeerJ 5:e3756. https://doi.org/10.7717/peerj.3756
Vogelsang C, Lusher AL, Dadkhah ME, Sundvor I, Umar M, Ranneklev SB et al (2019) Microplastics in road dust – characteristics, pathways and measures. Norsk institutt for vannforskning. Retrieved from https://brage.bibsys.no/xmlui/handle/11250/2493537
Vollertsen J, Hansen AA (2017) Microplastic in Danish wastewater: Sources, occurrences and fate, Environmental Project No. 1906. Danish Environmental Protection Agency, pp 1–55. Retrieved from https://www2.mst.dk/Udgiv/publications/2017/03/978-87-93529-44-1.pdf
Wagner S, Hüffer T, Klöckner P, Wehrhahn M, Hofmann T, Reemtsma T (2018) Tire wear particles in the aquatic environment – a review on generation, analysis, occurrence, fate and effects. Water Res 139:83–100. https://doi.org/10.1016/j.watres.2018.03.051
Walling DE, Webb BW, Woodward JC (1992) Some sampling considerations in the design of effective strategies for monitoring sediment-associated transport. Erosion and Sediment Transport Monitoring Programmes in River Basins (Proceedings of the Oslo Symposium, August 1992), IAHS Publication no. 210, pp 279–288. Retrieved from: https://pdfs.semanticscholar.org/c3db/f67dbe41df9f7194ebe55d3748015fd816ad.pdf?_ga=2.165832254.18612053.1583661455-2018662021.1583059758
Walling DE, Owens P, Carter J, Leeks GJ, Lewis S, Meharg A, Wright J (2003) Storage of sediment-associated nutrients and contaminants in river channel and floodplain systems. Appl Geochem 18(2):195–220. https://doi.org/10.1016/S0883-2927(02)00121-X
Wang W, Ndungu AW, Li Z, Wang J (2017) Microplastics pollution in inland freshwaters of China: a case study in urban surface waters of Wuhan, China. Sci Total Environ 575:1369–1374. https://doi.org/10.1016/j.scitotenv.2016.09.213
Wang W, Yuan W, Chen Y, Wang J (2018) Microplastics in surface waters of Dongting Lake and Hong Lake, China. Sci Total Environ 633:539–545. https://doi.org/10.1016/j.scitotenv.2018.03.211
Wang T, Li B, Zou X, Wang Y, Li Y, Xu Y et al (2019a) Emission of primary microplastics in mainland China: Invisible but not negligible. Water Res 162:214–224. https://doi.org/10.1016/j.watres.2019.06.042
Wang Z, Qin Y, Li W, Yang W, Meng Q, Yang J (2019b) Microplastic contamination in freshwater: first observation in Lake Ulansuhai, Yellow River Basin, China. Environ Chem Lett 17(4):1821–1830. https://doi.org/10.1007/s10311-019-00888-8
Watkins L, Sullivan PJ, Walter MT (2019) A case study investigating temporal factors that influence microplastic concentration in streams under different treatment regimes. Environ Sci Pollut Res 26(21):21797–21807. https://doi.org/10.1007/s11356-019-04663-8
Wei S, Luo H, Zou J, Chen J, Pan X, Rousseau DPL, Li J (2020) Characteristics and removal of microplastics in rural domestic wastewater treatment facilities of China. Sci Total Environ 739:139935. https://doi.org/10.1016/j.scitotenv.2020.139935
Wen X, Du C, Xu P, Zeng G, Huang D, Yin L, Yin Q, Hu L, Wan J, Zhang J, Tan S, Deng R (2018) Microplastic pollution in surface sediments of urban water areas in Changsha, China: Abundance, composition, surface textures. Mar Pollut Bull 136:414–423. https://doi.org/10.1016/j.marpolbul.2018.09.043
Wik A, Dave G (2005) Environmental labeling of car tires—toxicity to Daphnia magna can be used as a screening method. Chemosphere 58(5):645–651. https://doi.org/10.1016/j.chemosphere.2004.08.103
Wik A, Dave G (2006) Acute toxicity of leachates of tire wear material to Daphnia magna—variability and toxic components. Chemosphere 64(10):1777–1784. https://doi.org/10.1016/j.chemosphere.2005.12.045
Wik A, Dave G (2009) Occurrence and effects of tire wear particles in the environment – a critical review and an initial risk assessment. Environ Pollut 157(1):1–11. https://doi.org/10.1016/j.envpol.2008.09.028
Wik A, Nilsson E, Källqvist T, Tobiesen A, Dave G (2009) Toxicity assessment of sequential leachates of tire powder using a battery of toxicity tests and toxicity identification evaluations. Chemosphere 77(7):922–927. https://doi.org/10.1016/j.chemosphere.2009.08.034
Windsor FM, Tilley RM, Tyler CR, Ormerod SJ (2019a) Microplastic ingestion by riverine macroinvertebrates. Sci Total Environ 646:68–74. https://doi.org/10.1016/j.scitotenv.2018.07.271
Windsor FM, Durance I, Horton AA, Thompson RC, Tyler CR, Ormerod SJ (2019b) A catchment-scale perspective of plastic pollution. Glob Chang Biol 25:1207–1221
Winter LT, Foster IDL, Charlesworth SM, Lees JA (2001) Floodplain lakes as sinks for sediment-associated contaminants—a new source of proxy hydrological data? Sci Total Environ 266(1–3):187–194. https://doi.org/10.1016/s0048-9697(00)00745-2
Wright SL, Ulke J, Font A, Chan KLA, Kelly FJ (2020) Atmospheric microplastic deposition in an urban environment and an evaluation of transport. Environ Int 136:105411. https://doi.org/10.1016/j.envint.2019.105411
Xiong X, Zhang K, Chen X, Shi H, Luo Z, Wu C (2018) Sources and distribution of microplastics in China’s largest inland lake – Qinghai Lake. Environ Pollut 235:899–906. https://doi.org/10.1016/j.envpol.2017.12.081
Xu Q, Gao Y, Xu L, Shi W, Wang F, LeBlanc GA et al (2020) Investigation of the microplastics profile in sludge from China’s largest Water reclamation plant using a feasible isolation device. J Hazard Mater 388:122067. https://doi.org/10.1016/j.jhazmat.2020.122067
Yang L, Li K, Cui S, Kang Y, An L, Lei K (2019) Removal of microplastics in municipal sewage from China’s largest water reclamation plant. Water Res 155:175–181. https://doi.org/10.1016/j.watres.2019.02.046
Yin L, Jiang C, Wen X, Du C, Zhong W, Feng Z, Long Y, Ma Y (2019) Microplastic pollution in surface water of urban lakes in Changsha, China. Int J Environ Res Public Health 16(9):1650. https://doi.org/10.3390/ijerph16091650
Yu M, van der Ploeg M, Lwanga EH, Yang X, Zhang S, Ma X et al (2019) Leaching of microplastics by preferential flow in earthworm (Lumbricus terrestris) burrows. Environ Chem 16(1):31–40. https://doi.org/10.1071/EN18161
Yuan W, Liu X, Wang W, Di M, Wang J (2019) Microplastic abundance, distribution and composition in water, sediments, and wild fish from Poyang Lake, China. Ecotoxicol Environ Saf 170:180–187. https://doi.org/10.1016/j.ecoenv.2018.11.126
Zhang Z, Chen Y (2020) Effects of microplastics on wastewater and sewage sludge treatment and their removal: a review. Chem Eng J 382:122955. https://doi.org/10.1016/j.cej.2019.122955
Zhang GS, Liu YF (2018) The distribution of microplastics in soil aggregate fractions in southwestern China. Sci Total Environ 642:12–20. https://doi.org/10.1016/j.scitotenv.2018.06.004
Zhang K, Su J, Xiong X, Wu X, Wu C, Liu J (2016a) Microplastic pollution of lakeshore sediments from remote lakes in Tibet plateau, China. Environ Pollut 219:450–455. https://doi.org/10.1016/j.envpol.2016.05.048
Zhang K, Su J, Xiong X, Wu X, Wu C, Liu J (2016b) Microplastic pollution of lakeshore sediments from remote lakes in Tibet plateau, China. Environ Pollut 219:450–455. https://doi.org/10.1016/j.envpol.2016.05.048
Zhang Y, Gao T, Kang S, Sillanpää M (2019) Importance of atmospheric transport for microplastics deposited in remote areas. Environ Pollut 254:112953. https://doi.org/10.1016/j.envpol.2019.07.121
Zhao S, Zhu L, Li D (2016) Microscopic anthropogenic litter in terrestrial birds from Shanghai, China: Not only plastics but also natural fibers. Sci Total Environ 550:1110–1115. https://doi.org/10.1016/j.scitotenv.2016.01.112
Zhou P, Huang C, Fang H, Cai W, Li D, Li X, Yu H (2011) The abundance, composition and sources of marine debris in coastal seawaters or beaches around the northern South China Sea (China). Mar Pollut Bull 62(9):1998–2007. https://doi.org/10.1016/j.marpolbul.2011.06.018
Zhou Q, Tian C, Luo Y (2017) Various forms and deposition fluxes of microplastics identified in the coastal urban atmosphere. Chin Sci Bull 62(33):3902–3909. https://doi.org/10.1360/N972017-00956
Zhou B, Wang J, Zhang H, Shi H, Fei Y, Huang S et al (2019) Microplastics in agricultural soils on the coastal plain of Hangzhou Bay, east China: multiple sources other than plastic mulching film. J Hazard Mater 121814. https://doi.org/10.1016/j.jhazmat.2019.121814
Acknowledgements
EK was supported by the Innovation Fund Denmark under the project Udvikling af nye metoder til at detektere mikroplastik i vandmiljøet. ER was supported by both the MicroROAD project, funded by both the NordFoU-project REHIRUP, consisting of the Norwegian Public Roads Administration, the Swedish Transport Administration and the Danish Road Directorate, and the Norwegian Institute for Water Research (NIVA). RH was supported by the Research Council of Norway (grant number 271825/E50), in the frame of the collaborative international consortium (IMPASSE) financed under the ERA-NET WaterWorks2015 co-funded call. This ERA-NET is an integral part of the 2016 Joint Activities developed by the Water Challenges for a Changing World Joint Programme Initiative (Water JPI).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2022 The Author(s)
About this chapter
Cite this chapter
Kallenbach, E.M.F., Rødland, E.S., Buenaventura, N.T., Hurley, R. (2022). Microplastics in Terrestrial and Freshwater Environments. In: Bank, M.S. (eds) Microplastic in the Environment: Pattern and Process. Environmental Contamination Remediation and Management. Springer, Cham. https://doi.org/10.1007/978-3-030-78627-4_4
Download citation
DOI: https://doi.org/10.1007/978-3-030-78627-4_4
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-78626-7
Online ISBN: 978-3-030-78627-4
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)