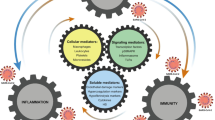
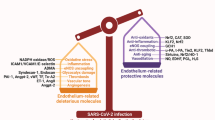
Abstract
A number of different viral species are known to have effects on the endothelium. These include dengue, Ebola, Marburg, Lassa fever, yellow fever and influenza viruses, cytomegalovirus and coronaviruses. There are currently seven human endemic coronaviruses, all of which cause respiratory diseases and bind to receptors found within the endothelium. The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) that causes the coronavirus disease 2019 (COVID-19) is highly infectious. Like its predecessor, SARS-CoV, it binds to angiotensin-converting enzyme-2 (ACE-2), which is expressed in many cell types, particularly in the lung, including endothelial cells. The initiation of a cytokine storm by the virus along with infection of endothelial cells leads to apoptosis and structural and functional changes that attenuate vascular integrity in many organs including the lungs, heart, liver and kidney. Endothelial damage also enhances the coagulation pathway leading to thrombus formation in major vessels and capillaries. Infection with SARS-CoV-2 has an adverse outcome for individuals with particular comorbid diseases, e.g. hypertension, obesity, type 2 diabetes and cardiovascular disease. It is possible that this is due to the presence of pre-existing endothelial dysfunction and systemic inflammation in subjects with these diseases. Therapies for COVID-19 that target the endothelium, the inflammatory response and the coagulation pathway are currently under trial.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Estola T (1970) Coronaviruses, a new group of animal RNA viruses. Avian Dis 14:330–336
Mäkelä MJ, Puhakka T, Ruuskanen O, Leinonen M, Saikku P, Kimpimäki M et al (1998) Viruses and bacteria in the etiology of the common cold. J Clin Microbiol 36:539–542
Drosten C, Günther S, Preiser W, Van Der Werf S, Brodt HR, Becker S et al (2003) Identification of a novel coronavirus in patients with severe acute respiratory syndrome. N Engl J Med 348:1967–1976
Zaki AM, Van Boheemen S, Bestebroer TM, Osterhaus AD, Fouchier RA (2012) Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med 367:1814–1820
Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y et al (2020) Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet 395:507–513
Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao Y et al (2020) Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis 71:762–768
Jiang F, Deng L, Zhang L, Cai Y, Cheung CW, Xia Z (2020) Review of the clinical characteristics of coronavirus disease 2019 (COVID-19). J Gen Intern Med 35:1545–1549
WHO (2020) WHO Coronavirus Disease (COVID-19) Dashboard [Online]. Available: https://covid19.who.int/. Accessed 23rd July 2020
Perlman S, Dandekar AA (2005) Immunopathogenesis of coronavirus infections: implications for SARS. Nat Rev Immunol 5:917–927
Zhou P, Yang X-L, Wang X-G, Hu B, Zhang L, Zhang W et al (2020) A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579:270–273
Jiang S, Hillyer C, Du L (2020) Neutralizing antibodies against SARS-CoV-2 and other human coronaviruses. Trends Immunol 41:355–359
Brevetti G, Schiano V, Chiariello M (2008) Endothelial dysfunction: a key to the pathophysiology and natural history of peripheral arterial disease? Atherosclerosis 197:1–11
Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS et al (2020) Endothelial cell infection and endotheliitis in COVID-19. Lancet 395:1417–1418
Klein D, Hurley LB, Quesenberry JC (1999) Sidney S (2002) Do protease inhibitors increase the risk for coronary heart disease in patients with HIV-1 infection? J Acquir Immune Defic Syndr 30:471–477
Wang T, Green LA, Gupta SK, Kim C, Wang L, Almodovar S et al (2014) Transfer of intracellular HIV Nef to endothelium causes endothelial dysfunction. PLoS One 9:e91063. https://doi.org/10.1371/journal.pone.0091063
Chang C-H, Huang Y, Issekutz AC, Griffith M, Lin K-H, Anderson R (2002) Interleukin-1α released from epithelial cells after adenovirus type 37 infection activates intercellular adhesion molecule 1 expression on human vascular endothelial cells. J Virol 76:427–431
Fourie C, Van Rooyen J, Pieters M, Conradie K, Hoekstra T, Schutte A (2011) Is HIV-1 infection associated with endothelial dysfunction in a population of African ancestry in South Africa? Cardiovasc J Afr 22:134–140
Arildsen H, Sorensen KE, Ingerslev JM, Ostergaard LJ, Laursen AL (2013) Endothelial dysfunction, increased inflammation, and activated coagulation in HIV-infected patients improve after initiation of highly active antiretroviral therapy. HIV Med 14:1–9
Van Vonderen MG, Hassink EA, Van Agtmael MA, Stehouwer CD, Danner SA, Reiss P et al (2009) Increase in carotid artery intima-media thickness and arterial stiffness but improvement in several markers of endothelial function after initiation of antiretroviral therapy. J Infect Dis 199:1186–1194
Hemmat N, Ebadi A, Badalzadeh R, Memar MY, Baghi HB (2018) Viral infection and atherosclerosis. J Clin Microbiol Infect Dis 37:2225–2233
Popović M, Smiljanić K, Dobutović B, Syrovets T, Simmet T, Isenović ERJJOT et al (2012) Human cytomegalovirus infection and atherothrombosis. J Thromb Thrombolysis 33:160–172
Peretz A, Azrad M, Blum A (2019) Influenza virus and atherosclerosis. QJM 112:749–755
Basler CF (2017) Molecular pathogenesis of viral hemorrhagic fever. Semin Immunopathol 39:551–561
Feldmann H, Bugany H, Mahner F, Klenk H-D, Drenckhahn D, Schnittler HJ (1996) Filovirus-induced endothelial leakage triggered by infected monocytes/macrophages. J Virol 70:2208–2214
Ter Meulen J, Sakho M, Koulemou K, Magassouba NF, Bah A, Preiser W et al (2004) Activation of the cytokine network and unfavorable outcome in patients with yellow fever. J Infect Dis 190:1821–1827
Rastogi M, Sharma N, Singh SK (2016) Flavivirus NS1: a multifaceted enigmatic viral protein. Virol J 13:131–131
Beatty PR, Puerta-Guardo H, Killingbeck SS, Glasner DR, Hopkins K, Harris E (2015) Dengue virus NS1 triggers endothelial permeability and vascular leak that is prevented by NS1 vaccination. Sci Transl Med 7:304ra141. https://doi.org/10.1126/scitranslmed.aaa3787
Noronha L, Zanluca C, Azevedo ML, Luz KG, Santos CN (2016) Zika virus damages the human placental barrier and presents marked fetal neurotropism. Mem Inst Oswaldo Cruz 111:287–293
Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F et al (2020) Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med 383:120–128
Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S et al (2020) SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 181:271–280
Hamming I, Timens W, Bulthuis M, Lely A, Navis G, Van Goor H (2004) Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol 203:631–637
Li W, Moore MJ, Vasilieva N, Sui J, Wong SK, Berne MA et al (2003) Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature 426:450–454
Raj VS, Mou H, Smits SL, Dekkers DH, Müller MA, Dijkman R et al (2013) Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC. Nature 495:251–254
Yeager CL, Ashmun RA, Williams RK, Cardellichio CB, Shapiro LH, Look AT et al (1992) Human aminopeptidase N is a receptor for human coronavirus 229E. Nature 357:420–422
Li F (2015) Receptor recognition mechanisms of coronaviruses: a decade of structural studies. J Virol 89:1954–1964
Zhao M, Wang M, Zhang J, Ye J, Xu Y, Wang Z et al (2020) Advances in the relationship between coronavirus infection and cardiovascular diseases. Biomed Pharmacother 127:110230. https://doi.org/10.1016/j.biopha.2020.110230
Tong M, Jiang Y, Xia D, Xiong Y, Zheng Q, Chen F et al (2020) Elevated serum endothelial cell adhesion molecules expression in COVID-19 patients. J Infect Dis:jiaa349. https://doi.org/10.1093/infdis/jiaa349
Escher R, Breakey N, Lämmle BJTR (2020) Severe COVID-19 infection associated with endothelial activation. Thromb Res 190:62. https://doi.org/10.1016/j.thromres.2020.04.014
Mezoh G, Crowther NJ (2019) Deciphering endothelial dysfunction in the HIV-infected population. Adv Exp Med Biol 1134:193–215
Sadler JE (1998) Biochemistry and genetics of von Willebrand factor. Annu Rev Biochem 67:395–424
Munro JM (1993) Endothelial-leukocyte adhesive interactions in inflammatory diseases. Eur Heart J 14:72–77
Koraka P, Murgue B, Deparis X, Van Gorp EC, Setiati TE, Osterhaus AD et al (2004) Elevation of soluble VCAM-1 plasma levels in children with acute dengue virus infection of varying severity. J Med Virol 72:445–450
Cardier JE, Rivas B, Romano E, Rothman AL, Perez-Perez C, Ochoa M et al (2006) Evidence of vascular damage in dengue disease: demonstration of high levels of soluble cell adhesion molecules and circulating endothelial cells. Endothelium 13:335–340
Umehara H, Bloom ET, Okazaki T, Nagano Y, Yoshie O, Imai T (2004) Fractalkine in vascular biology: from basic research to clinical disease. Arterioscler Thromb Vasc Biol 24:34–40
Nizzoli ME, Merati G, Tenore A, Picone C, Consensi E, Perotti L et al (2020) Circulating endothelial cells in COVID-19. Am J Hematol 95:E187–E188. https://doi.org/10.1002/ajh.25881
Khider L, Gendron N, Goudot G, Chocron R, Hauw-Berlemont C, Cheng C et al (2020) Curative anticoagulation prevents endothelial lesion in COVID-19 patients. J Thromb Haemost. https://doi.org/10.1111/jth.14968
Blann AD, Woywodt A, Bertolini F, Bull TM, Buyon JP, Clancy RM et al (2005) Circulating endothelial cells. Thromb Haemost 93:228–235
López M, Vispo E, San Román J, Herrero D, Peris A, Corral A et al (2012) High risk of endothelial dysfunction in HIV individuals may result from deregulation of circulating endothelial cells and endothelial progenitor cells. AIDS Res Hum Retrov 28:656–659
Liu J, Li S, Liu J, Liang B, Wang X, Wang H et al (2020) Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine 55:102763. https://doi.org/10.1016/j.ebiom.2020.102763
Zhang C, Wu Z, Li J-W, Zhao H, Wang GQ (2020) The cytokine release syndrome (CRS) of severe COVID-19 and Interleukin-6 receptor (IL-6R) antagonist Tocilizumab may be the key to reduce the mortality. Int J Antimicrob Agents 55:105954. https://doi.org/10.1016/j.ijantimicag.2020.105954
Teuwen L-A, Geldhof V, Pasut A, Carmeliet P (2020) COVID-19: the vasculature unleashed. Nat Rev Immunol 20:389–391
Helms J, Tacquard C, Severac F, Leonard-Lorant I, Ohana M, Delabranche X et al (2020) High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Med 46:1089–1098
Argenziano MG, Bruce SL, Slater CL, Tiao JR, Baldwin MR, Barr RG et al (2020) Characterization and clinical course of 1000 patients with coronavirus disease 2019 in New York: retrospective case series. BMJ 369:m1996. https://doi.org/10.1136/bmj.m1996
Atkins JL, Masoli JA, Delgado J, Pilling LC, Kuo C-L, Kuchel GA et al (2020) Preexisting comorbidities predicting COVID-19 and mortality in the UK biobank community cohort. J Gerontol A Biol Sci Med Sci:glaa183. https://doi.org/10.1093/gerona/glaa183
Onder G, Rezza G, Brusaferro S (2020) Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA. https://doi.org/10.1001/jama.2020.4683
Davies MA (2020) HIV and risk of COVID-19 death: a population cohort study from the Western Cape Province, South Africa. medRxiv:20145185. https://doi.org/10.1101/2020.07.02.20145185
Li X, Sun X, Carmeliet P (2019) Hallmarks of endothelial cell metabolism in health and disease. Cell Metab 30:414–433
Donath MY, Meier DT, Böni-Schnetzler M (2019) Inflammation in the pathophysiology and therapy of cardiometabolic disease. Endocr Rev 40:1080–1091
Zhu Y, Xian X, Wang Z, Bi Y, Chen Q, Han X et al (2018) Research progress on the relationship between atherosclerosis and inflammation. Biomol Ther 8:80. https://doi.org/10.3390/biom8030080
Vinciguerra M, Romiti S, Fattouch K, De Bellis A, Greco E (2020) Atherosclerosis as pathogenetic substrate for Sars-Cov-2 cytokine storm. J Clin Med 9:E2095. https://doi.org/10.3390/jcm9072095
Wu Q, Zhou L, Sun X, Yan Z, Hu C, Wu J et al (2017) Altered lipid metabolism in recovered SARS patients twelve years after infection. Sci Rep 7:9110. https://doi.org/10.1038/s41598-017-09536-z
Johnson RM, Vinetz JM (2020) Dexamethasone in the management of COVID-19. BMJ 370:m2648. https://doi.org/10.1136/bmj.m2648
Luo P, Liu Y, Qiu L, Liu X, Liu D, Li J (2020) Tocilizumab treatment in COVID-19: a single center experience. J Med Virol 92:814–818
Xu X, Han M, Li T, Sun W, Wang D, Fu B et al (2020) Effective treatment of severe COVID-19 patients with tocilizumab. Proc Natl Acad Sci U S A 117:10970–10975
Morena V, Milazzo L, Oreni L, Bestetti G, Fossali T, Bassoli C et al (2020) Off-label use of tocilizumab for the treatment of SARS-CoV-2 pneumonia in Milan, Italy. Eur J Intern Med 76:36–42
Feldmann M, Maini RN, Woody JN, Holgate ST, Winter G, Rowland M et al (2020) Trials of anti-tumour necrosis factor therapy for COVID-19 are urgently needed. Lancet 395:1407–1409
Lee KC, Sewa DW, Phua GC (2020) Potential role of statins in COVID-19. Int J Infect Dis 96:615–617
Fedson D, Opal S, Rordam OM (2020) Treating patients with severe COVID-19 infection. mBio 11:e00398–e00320. doi:https://doi.org/10.1128/mBio.00398-20
Danser AHJ, Epstein M, Batlle D (2020) Renin-angiotensin system blockers and the COVID-19 pandemic. At present there is no evidence to abandon renin-angiotensin system blockers. Hypertension 75:1382–1385
Pons S, Fodil S, Azoulay E, Zafrani L (2020) The vascular endothelium: the cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Crit Care 24:353. https://doi.org/10.1186/s13054-020-03062-7
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2021 The Editor(s) (if applicable) and The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Cite this chapter
Mezoh, G., Crowther, N.J. (2021). Endothelial Dysfunction as a Primary Consequence of SARS-CoV-2 Infection. In: Guest, P.C. (eds) Clinical, Biological and Molecular Aspects of COVID-19. Advances in Experimental Medicine and Biology(), vol 1321. Springer, Cham. https://doi.org/10.1007/978-3-030-59261-5_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-59261-5_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-59260-8
Online ISBN: 978-3-030-59261-5
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)