Abstract
Potassium (K) outputs comprise removals in harvested crops and losses via a number of pathways. No specific environmental issues arise from K losses to the wider environment, and so they have received little attention. Nevertheless, K is very soluble and so can be leached to depth or to surface waters. Also, because K is bound to clays and organic materials, and adsorbed K is mostly associated with fine soil particles, it can be eroded with particulate material in runoff water and by strong winds. It can also be lost when crop residues are burned in the open. Losses represent a potential economic cost to farmers and reduce soil nutritional status for plant growth. The pathways of loss and their relative importance can be related to: (a) the general characteristics of the agricultural ecosystem (tropical or temperate regions, cropping or grazing, tillage management, interactions with other nutrients such as nitrogen); (b) the specific characteristics of the agricultural ecosystem such as soil mineralogy, texture, initial soil K status, sources of K applied (organic, inorganic), and rates and timing of fertilizer applications. This chapter provides an overview of the main factors affecting K removals in crops and losses through runoff, leaching, erosion, and open burning.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
Potassium is removed in harvested crops, a necessary and important part of agriculture and food production, but it is also lost by erosion, leaching, and open burning. Unlike nitrogen (N), there are no gaseous losses of K. Management practices can reduce losses, sustain plant-available soil K supplies, and improve the recovery efficiency of K applications. Compared to N and phosphorus (P), K losses have received little attention because they have few, if any, environmental impacts. Nevertheless, they represent a potential economic cost to farmers. They also increase the risk of poor soil K fertility and unbalanced nutrition for plant growth.
Higher losses are expected in soils with a lower K holding capacity. We define potassium holding capacity as the maximum quantity of K that can be retained by a given volume of soil. A better understanding of the main pathways of K losses and the key factors controlling them can improve practical recommendations to ensure farmers optimize the K-holding capacity of their soils to increase or sustain productivity and economic returns.
3.1 Removal in Harvested Crops
Harvested plant K is the quantity of K in plant material removed from a given area. The rate of K removal per unit area increases when the total K accumulation in harvested plant organs increases. Such increases occur with an increased K concentration in plant tissue, higher yields, or a shift toward removing additional plant organs from the area, such as straw and grain rather than just grain.
An associated term is Harvest Index (HI), which is the fraction of the harvested yield divided by the total amount of biomass produced (Unkovich et al. 2010). Although the denominator is defined as the total biomass produced (shoots and roots), it is most common to only measure the above-ground biomass due to the difficulty of measuring root biomass. The unharvested portion that remains on the field following harvest is defined as crop residue. Knowing the proportion of biomass or nutrients (such as K) that will be removed during harvest and what proportion will remain as residue or be returned to the soil is essential for estimating K budgets.
The HI can vary depending on the time of measurement. For example, many legume crops begin to shed leaves prior to physiological maturity and harvest. To correctly calculate the K requirement, the HI for these crops should be calculated using maximum biomass dry matter, including the leaves that fall to the ground. During maize (Zea mays L.) harvest, the grain may be the only product removed from the field or in some circumstances the stalks, leaves, and husks may be all removed for economic purposes (such as for silage or bioenergy). Additionally, the residue stalks and stems are cut at varying heights above the ground.
For determining the HI of horticultural crops, “maturity” is determined by market-driven parameters such as gelatinous mass filling of tomato fruit (Solanum lycopersicum L.), coloring of peppers (Capsicum annuum L.), head formation of lettuce (Lactuca sativa L.), plant size for spinach (Spinacia oleracea L.), tuber size for potatoes (Solanum tuberosum L.), or sugar accumulation of grapes (Vitis vinifera L.)
Indeterminate crops may be harvested multiple times. The unharvested portion of annual crops is generally returned to the soil. For perennial crops, the woody portion will continue to accumulate biomass and nutrients, with only the leaves dropping to the soil. In some cases, crop residues may not be returned to the same field from which they were harvested (e.g., pomace and bagasse).
Understanding both the rate of uptake and the total amount of K accumulated in the crop during the growing season, and in the harvested portion removed during harvest, is required for assessing the seasonal crop demand. For example, Bender et al. (2013) examined seasonal K uptake of six modern maize hybrids. They reported that K accumulation occurred in a sigmoidal pattern over the growing season, with most K uptake already completed by the time vegetative growth transitioned to reproductive growth. After this, a large amount of K was translocated from the vegetative tissue to the reproductive organs. At harvest, 30% of the total K was in the grain.
Rogers et al. (2019) measured total K accumulation of five barley (Hordeum vulgare L.) cultivars and found it peaked at the soft dough stage (253 kg K ha−1) and then declined to 172 K kg ha−1 at physiological maturity. This loss of 81 kg K ha−1 from the biomass occurred at the same time as a small increase in K in the barley heads (from 37 to 42 kg K kg ha−1). Sugarbeet (Beta vulgaris L.) had a sigmoidal pattern of K accumulation, with approximately half of the total accumulated K (>500 kg K ha−1) in the vegetative tops and half in the roots by the time of harvest (De et al. 2019).
Perennial crops can accumulate and export a large quantity of K at harvest. For example, almonds (Prunus dulcis (Mill.) D. A. Webb) remove 400 kg K in a typical 5 t ha−1 yield in the harvested hulls, shells, and kernels (Muhammad et al. 2009), and bananas (Musa spp.) remove even more ~750 kg K in a typical 50 t ha−1 yield during the extended fruit harvest period (Lahav and Turner 1989).
Crops grown for hay production, such as alfalfa (Medicago sativa L.), also remove large amounts of nutrients from the soil, especially K. For example, a healthy alfalfa crop may remove 25 kg K t−1 in a typical 25–35 t ha−1 yield. Cultivating high-yielding hay crops therefore requires special attention to avoid depleting the soil nutrient supply.
The K concentration in the residue of agricultural crops varies widely, as does the rate of subsequent K release from the residue (e.g., Anguria et al. 2017). Estimating both the quantity of crop residue and its K concentration are necessary steps for measuring the K in residues and the potential for K loss or recycling from residues remaining in the field.
Although the general differences in K removal among grain crops are well documented (e.g., soybeans seeds Glycine max (L.) Merr. contain approximately 4 times more K per t than maize), there is important variation in nutrient content among species based on the growing environment, yield level, and crop genetics. For example, Nathan et al. (2009) analyzed maize grain samples (n = 141 in 2006 and n = 214 in 2007) and measured a mean K concentration of 3.4 mg K kg−1 and 2.7 mg K kg−1 in the 2 years, respectively (±0.5 mg K kg−1 standard deviation). Even within the fairly small geographic region sampled, they found >25% difference in corn grain K concentrations in the 2 years.
This temporal difference in nutrient removal during crop harvest illustrates the challenge of using “average values” for estimating K offtake. A number of published databases exist that provide average nutrient removal coefficients for most harvested crops. However, many of these tables and databases do not use up-to-date measurements, do not properly cite where the information came from, or are not reliable for accounting for the significant spatial, cultural, and temporal variation in nutrient concentrations that arise during routine crop production practices. General nutrient removal databases can be useful for making nutrient offtake estimates but should not be used for more precise planning. Samples of harvested crops should be periodically analyzed in the laboratory in order to confirm the quantity of nutrients being removed from the field.
3.1.1 Whole-Plant Removal
The practice of removing straw from grain fields is common in many parts of the world and has important implications for both soil health and nutrient cycling. Residue removal may be locally useful for purposes such as animal feed or bedding, fuel, or for use in cellulose-based ethanol production. However, some level of organic matter input is required for maintaining the long-term ecological function and the agricultural productivity of soils.
Crop residues contain valuable plant nutrients, so removing them from the field will speed nutrient depletion and have economic impacts, especially for K. For example, small grain straw contains less P and N than the grain, but a higher proportion of K; i.e., the average straw:grain mass nutrient ratio in wheat is 0.47 for N, 0.26 for P, but 4.12 for K; the straw:grain nutrient ratio in barley is 0.49 for N, 0.35 for P, but 5.04 for K (Tarkalson et al. 2009). Therefore, when both grain and straw are removed from fields, soil K depletion is accelerated compared with harvesting only grain. The financial expense associated with purchasing K fertilizer to replace this harvested K should be accounted for in long-term nutrient budgets and decisions on residue removal.
Methods of handling the straw and crop residues also need to be considered when calculating potential K losses. Since K is readily leached from crop residues with rainfall and irrigation, the length of time the residue remains in the field before removal and how the residue is distributed before removal (e.g., windrows, piles, or broadcast) will significantly impact the amount of K ultimately removed in the biomass.
3.2 Erosion
Erosion loss is K lost from the movement of soil particles out of a given volume of soil. Losses can occur in both water and wind erosion. Soil particles eroded from the field carry adsorbed K with them. Water erosion occurs mostly across the soil surface or at shallow depths by runoff, but particles can also be transported to depth and lost through field drains, if the land is drained.
3.2.1 Water Erosion
Runoff loss arises from surface and subsurface movement of water. Surface runoff loss is K in water moving laterally over the soil surface in the direction of the slope. Subsurface runoff loss is K in water that infiltrates the soil surface to shallow depths and then moves laterally in the direction of the slope.
Korucu et al. (2018) used a collection pan at the soil surface to measure only surface runoff. They conducted their study on a site with 2% slope composed of loam and clay loam soils. A day after maize silage harvest, they planted cereal rye to test the effects of a cover crop on surface runoff. Approximately 1 month after planting rye, they broadcast 13 kg NH4-N ha−1, 27 kg P ha−1, and 83 kg K ha−1 as monoammonium phosphate and potassium chloride. An hour later, they simulated a 10-year extreme rainfall event, using spray nozzles to deliver 65 mm of water in 60 min. Such conditions favored runoff losses of fertilizer P and K. On treatments with no rye cover crop, it took an average of 4.9 min for runoff to begin after the start of the rainfall simulation (Table 3.1). Runoff averaged 27.3 mm. Total suspended solids averaged 444 kg ha−1 and K loss averaged 12.42 kg K ha−1. Average concentration of K in the runoff was 43.0 mg L−1. The month-old rye cover crop doubled the time for runoff to begin and reduced the total runoff amount by 65%, the total suspended solids by 68%, the K concentration by 75%, and the total K loss by 91%. Thus, even when a simulated 10-year rainfall event occurred just 1 h after a surface application of K, the rye cover crop reduced the K runoff loss to an average of 1.08 kg K ha−1. Where no cover crops were present, K losses across replications were 4–32% of the amount applied an hour before the start of the rainfall simulation. These results and others indicate that K losses in surface runoff depend on rainfall intensity, the timing of precipitation events, and the management of K applications (Alfaro et al. 2004b, 2008).
In temperate systems, favorable conditions for the development of surface runoff are mainly found during winter months. Although no mineral fertilizers are usually applied at that time, livestock farmers may apply substantial amounts of organic manures and slurries because there is more labor capacity available and the application does not interfere with grazing and cropping. Spreading in winter also helps to reduce storage requirements for manure or slurry. Some countries ban the spreading of manures and slurries in winter to reduce losses of nitrate and P to waters. However, the practice, as well as fertilizer applications in early spring, is still common and increases the risk of K losses in runoff (Alfaro et al. 2004a). The effect is likely to be greater in grazing areas because of the excessive trampling by animals that modifies soil structure, puddling the soil surface and reducing soil porosity (Heathwaite et al. 1996). In this case, the control of stocking rates at critical points during the grazing season is a key factor to reduce K losses from grazed paddocks (Alfaro et al. 2004a, b).
Rain reduces the porosity of the soil over time, increasing the likelihood of K runoff losses. Raindrop impact destroys soil aggregates and increases the thickness of the compacted surface layer (Rousseva et al. 2002), especially at high rainfall intensities, such as those in tropical regions (e.g., Acharya et al. 2007). This risk increases in soils with poor drainage. Under no-till, surface-applied K fertilizer increases the K concentration in runoff; however, because surface crop residue reduces the force of raindrop impact, sediment loss in the runoff is reduced, resulting in an overall decrease in total K loss (Bertol et al. 2005).
Zöbisch et al. (1995) measured total K loss from water erosion and the impact of cropping on losses in a soil with 8% slope at the Kabete Steep Lands Research Station in Nairobi, Kenya. The four treatments were maize (Zea mays L.), common bean (Phaseolus vulgaris L.), maize intercropped with common bean, and bare fallow. Sediment was collected after each rainfall event during the rainy season. There were 22 rainfall events during that period, eight of which, totaling 189.6 mm, produced runoff and erosion. Table 3.2 shows that bare fallow lost the most total K. Losses from the cropped treatments did not differ significantly, although losses from maize tended to be higher. Erosion contributed most to K loss—over 90% for all treatments; K dissolved in runoff comprised less than 10% of the total across all treatments.
Bertoluzzi et al. (2013) observed that the composition of suspended sediment differed significantly from that of the soil from which it was lost. During a 39-mm rainfall event with a peak intensity of 3.25 mm min−1, they sampled suspended solids from a stream monitoring point at the outlet of a 36-farm, 480 ha watershed in the Rio Grande do Sul State in southern Brazil. They divided stream flow occurring 120 min after the start of rainfall into three periods: phase A (15-min duration) was the initial period when streamflow was still near background levels and suspended solid concentrations were low; phase B (70-min duration) was characterized by high suspended solid concentrations and a rapid increase in flow in response to the rainfall event, followed by a slow decrease; and phase C (35-min duration) was when flow rate returned to background levels but contained low concentrations of suspended solids composed of fine particles. The total transported sediment was 29.2 Mg. Clay-sized particles dominated the sediment composition (Table 3.3) even though clay contents of the soils in the watershed were all less than 21%. Smectite comprised more than 90% of the clay, with most of the remainder being kaolinite. Illite, present in quantities up to 25% in some of the soils in the watershed, was not detected in the sediment. Potassium, defined as “labile K,” was lost in both phases. Bertoluzzi et al. (2013) defined labile K as the quantity of K in the soil solution plus the quantity of K most readily desorbed into solution from particle surfaces (i.e., the most soluble K in soil). To quantify labile K, they extracted K from soil with a cation/anion exchange resin for 16 h and then measured the K adsorbed to the resin. They repeated the extraction on the same sample of soil for a total of four successive extractions but considered labile K to be the K desorbed from the soil in only the first extraction. The sum of all the K desorbed during the second through fourth extractions was considered to be additional, potentially bioavailable K desorbed more slowly than the labile K (Table 3.3). They found that labile K was greatest in phase A and second highest in phase C, even though phase C had the highest clay content and most persistent suspension of fine particulates. Phases A and C also contained the highest quantities of total bioavailable K (labile plus additional, more slowly desorbed bioavailable K). Both of these phases were associated with slower streamflow.
3.2.2 Wind Erosion
Potassium is also lost through wind erosion. The greater the velocity of wind, the more soil is eroded (Wang et al. 2018). The smallest dry particle sizes are most susceptible to wind erosion (Yan et al. 2018). Dry particle size can be measured by air-drying soil samples and then passing them through a series of sieves of progressively smaller mesh sizes. (Dry particle size is not the same as soil texture. Soil texture is determined by using a dispersing solution, typically sodium hexametaphosphate, to break up aggregates into sand-, silt-, and clay-sized fractions.) Depending on the soil and its management, dry particles of a given size can be made up of a variety of percentages of clay, silt, and sand. Yan et al. (2018) observed that wind-eroded soils lost more fine dry particles (<0.2 mm in diameter), than larger dry particles; however, the sand, silt, and clay composition did not change. They also found that, compared to the composition of the bulk soil, a disproportionate amount of K was lost with the fine dry particles.
These few studies elucidate key points about erosion. First, erosion losses of K, though they have not been studied to the extent that leaching losses have, can be a dominant form of loss. Second, the composition of eroded soil can be very different from the bulk soil. Smaller-sized particles are more subject to erosion, and those smaller particles contain a significant portion of the bulk soil’s K supply.
3.3 Leaching
Leaching is the displacement of K below the rhizosphere volume by water percolating down the soil profile. The rhizosphere volume is the volume of soil adjacent to and influenced by plant roots. Leaching losses can be expected in the presence of drainage when K inputs exceed the sum of K holding capacity and plant uptake (Johnston 2003). Leaching losses can be as low as 0.2 kg K ha−1 year−1 in the prairies of northern America (Brye and Norman 2004) and as high as 185 kg K ha−1 year−1 under urine patches in a silt loam soil in New Zealand (Di and Cameron 2004). These losses are influenced by the rate of K applied, the timing of fertilizer or manure application, soil type and land use, and the amount and pathways of drainage.
It has been proposed that K leaching losses follow a two-phase pattern (Fig. 3.1). Phase A (fast) arises from macropore flow and the presence of K in solution. Macropore flow, or preferential flow is the rapid movement of water and solutes through large pores. These large pores may be channels left by roots or worms, cracks in the soil, or other larger voids formed from biological, geological, or anthropogenic causes. The presence of K in solution at the beginning of the drainage season may result from any one or more of the following: release of K from soil particles upon rewetting; applications of K as fertilizer or manure; leaching from crop residues; or soil biological activity (Alfaro et al. 2004b; Askegaard et al. 2003). Phase B (slow) is dependent on the amount and intensity of rainfall and the associated development of matrix flow later in the drainage season (Alfaro et al. 2004b). Matrix flow is the slow movement of water and solutes from soil volumes of higher total soil water potential to soil volumes of lower total soil water potential.
Simplified diagram showing the initial rapid leaching phase arising from macropore flow and the presence of K in solution (Phase A), and the subsequent slow leaching phase (Phase B) caused by matrix flow. (Alfaro et al. 2004a)
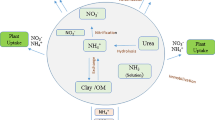
Potassium-holding capacity is determined by the presence of soil particles with adsorption sites that hold K on the planar, edge, wedge, and interlayer sites of phyllosilicate minerals. These sites bind K with sufficient energy to keep it from readily re-entering the soil solution. The largest number of these sites is usually present in the clay fraction where the smallest particle sizes maximize surface area. Fewer sites are available in the silt fraction and fewer still are in the sand fraction. Thus, the expectation is that K leaching is least in clay soils, greatest in sandy soils, and somewhere between in loamy soils. Figure 3.2 shows this conceptual relationship for sand and clay for a layer of soil 1-cm thick. As progressively more K is added, an exponentially lower proportion of it is adsorbed as adsorption sites are saturated with K, leaving a greater proportion of the added K in the soil solution. This soil solution K is free to move downward with the wetting front to the next soil layer.
Modelled relationships between the fraction of K adsorbed to soil particle surfaces and the rate of K applied for a soil layer 1-cm thick. (Johnston and Goulding 1992)
For a given K-holding capacity, a history of higher application rates of K reduces the quantity of additional K that can be retained. Rosolem et al. (2010) observed greater movement of K down the soil profile with successively greater rates of previously applied K. High application rates of manure can have the same effect. Not accounting for K when using organic sources (Askegaard and Eriksen 2002; Bernal et al. 1993) or wastewater (Arienzo et al. 2009) can result in overapplications of K, saturating adsorption sites and exceeding the K holding capacity.
Proper manure management should require farmers to account for K in manures, which is much more variable than in commercial fertilizers. Ideally, each load of manure should be tested for its K content to allow the farmer to back calculate how much K was applied. While some testing is done, in practice, it is more common to use standardized estimates of K concentration and plant-availability. Software decision support tools like MANNER-NPK estimate plant-available nutrients in manures and other organic materials (Nicholson et al. 2013) to help farmers account for the K applied. Another decision support tool is OVERSEER (Wheeler et al. 2003), a farm-scale nutrient budget model used by farmers and consultants throughout New Zealand. The model strives to optimize nutrient input (both inorganic and organic) to maximize production while minimizing nutrient losses to water (Wheeler et al. 2008).
Soils with higher clay contents may not necessarily have greater K-holding capacities. Macropore flow has such a dominant effect on K leaching that it can override differences in soil texture. As an example, Alfaro et al. (2004b) conducted leaching studies on monolith lysimeters of four soils: one sand, two loams, and one clay. Each lysimeter was 80 cm in diameter and 135 cm deep. The study used four lysimeters of each soil as replicates, buried in a field so that the top of the lysimeter was level with the soil surface. The excavated monolith columns preserved the original structure of each soil. Dairy slurry containing 5.7% K (dry basis) was applied at a rate of 24 L per lysimeter, split evenly across four applications during the year. Table 3.4 presents the results for leachate collected between October 2000 and April 2001. As expected, total K leaching losses from the two loam soils were both less than those from the sandy soil, even though flow from all three soils was the same. Unexpectedly, the clay soil lost as much K as the sand. Leachate from the clay soil had the lowest flow but the greatest concentration of K. The dominance of macropore flow in two of the four lysimeters containing the clay soil prevented K in the percolating water from diffusing into clay aggregates and being adsorbed. In those two lysimeters, the average K concentrations across the sampling period were 25.5 and 36.3 mg L−1. In the other two clay soil lysimeters, matrix flow dominated, resulting in average K concentrations of 1.6 and 0.9 mg L−1. When all four replicates of the clay soil were averaged it had the highest losses of K, but with very large variation. These results show how important macropore flow can be for determining the quantity of K leaching losses and the likely spatial variation of leaching in clay soils.
Macropore flow has been shown to occur immediately after a cattle urination event. The quantity of K in the event is large, and is deposited to a small volume of soil. Also, the K supply is usually in excess of the short-term requirements of the plants growing in the urine patch. Consequently, K penetrates to depth in the profile (Williams and Haynes 1992). As with the lysimeters above, percolation occurs too quickly for any significant sorption reactions between the soil and solutes in the urine (Williams and Haynes 1992). In dairy systems, urine from dairy cattle is responsible for 74 to 92% of total K losses (Williams et al. 1990), accounting for 3–29 kg K ha−1 year−1 in grazing areas of Chile and New Zealand (Alfaro et al. 2006; Williams et al. 1990), and up to 185 kg K ha−1 year−1 under urine patches in New Zealand soils (Di and Cameron 2004).
While practitioners in the field often associate greater K retention with higher cation exchange capacity (CEC), CEC has not proven to be a good predictor of K loss from soils (Quémener 1986). An important confounding factor is organic matter, which has a high CEC but does not bind K as strongly as mineral adsorption sites (Quémener 1986; Thomas and Hipp 1968). In high organic matter soils, heavy rains can seriously deplete the amount of soluble K in a matter of days (Thomas and Hipp 1968). In addition, the presence of clay-sized phyllosilicate minerals with high CEC does not limit K leaching losses if macropore flow is present. Thus, CEC alone is not a good predictor of K leaching losses; however, it could be a useful factor in prediction models that incorporate additional factors.
When N increases crop K uptake and yield, lower K leaching losses may follow. For instance, the use of nitrification inhibitors such as dicyandiamide in grassland soils has been found to reduce K leaching losses by up to 65%, probably as an indirect effect of its increasing yield (Di and Cameron 2004). However, N applications may also result in larger quantities of leached K, even when system productivity is increased. In a study conducted in southwest England (Alfaro et al. 2003), larger K leaching losses occurred even though N applications increased system productivity so much that K outputs exceeded K inputs, resulting in a net negative K budget.
In intensively managed agricultural systems with nutrient surpluses, greater K leaching losses are usually linked to greater N leaching losses (Brye and Norman 2004). Nitrate leached through the soil profile forms ion pairs with other solution cations to balance charge. When K+ is part of the ion pair, it will move with NO3− down the soil profile. This effect has also been observed after liming. In acidic soils, liming promotes nitrification and increases nitrate concentration in the uppermost soil layers, resulting in higher K leaching losses (Crusciol et al. 2011). Additionally, calcium (Ca2+) and magnesium (Mg2+) in lime can exchange with K+ on soil particle surfaces, moving it into the soil solution. Greater K leaching can also occur when competing cations are present in irrigation water (Kolahchi and Jalali 2007; Sekhon 1982).
3.4 Modeling Potassium Losses
Several models, ranging from conceptual to computational, have been developed to estimate K behavior. This section highlights some of these past efforts to model K losses from soils.
3.4.1 Conceptual Model of Leaching
Alfaro et al. (2004a) proposed a conceptual model that combined surface runoff, macropore flow, and matrix flow. Their model was based on research conducted on field drainage plots (Armstrong and Garwood 1991). Each of those plots collected surface runoff and subsurface runoff downslope from the upper 30 cm of soil. Subsurface runoff from this layer was associated with macropore flow. On drained plots, mole channels placed below 30 cm were added along with an associated second drainage collection point downslope. Flow collected at that second point was associated with matrix flow and classified as leaching. The conceptual model is presented in Fig. 3.3. The solid black line represents the combination of surface runoff, macropore flow through the upper 30 cm of soil, and subsurface runoff from that upper layer. The dashed line represents matrix flow. At the start of the time period considered, the soil is dry, and there is little runoff or matrix flow. When rainfall starts, runoff flow increases, but matrix flow remains low. Once enough time has passed for water to infiltrate the soil, matrix flow subsequently increases. According to these principles, higher intensity rainfall events favor larger losses by runoff, because larger quantities of water fall before matrix flow become significant.
Idealized hydrograph of water flow after rainfall on a dry soil. (Alfaro et al. 2004a)
3.4.2 EPIC
De Barros et al. (2004) modified the EPIC model to estimate K losses through leaching, surface runoff, and subsurface runoff. They adjusted the model to match the climatic (semiarid), pedological, and cropping system conditions in Brazil. Soil is modeled as a stack of soil layers. For only the first layer, K lost in surface runoff is subtracted and not added to any remaining soil layers. For all layers, K lost in subsurface runoff and leaching are subtracted from a given layer and added to the next layer until the bottom of the soil profile is reached.
3.4.3 KLEACH
Johnston and Goulding (1992) used relationships similar to Fig. 3.2 as the basis for the model KLEACH. This model considers soil to be a series of consecutively stacked 1-cm thick layers. Added K not adsorbed by one layer moves to the adjacent layer below it. The model estimates K adsorbed by each successive layer as K moves down the soil profile. Figure 3.4 shows the results of two simulations for the cultivated layer (20 cm) of soil. The first (Fig. 3.4a) is for 200 kg K ha−1 applied as KCl to a clay loam soil. The second (Fig. 3.4b) models 800 kg K ha−1 applied as manure slurry to a sandy soil. In both cases, KLEACH models 100% adsorption of the applied K when summed over all soil depths. Compared to the clay loam, the sand is predicted to adsorb less K in each 1-cm layer, resulting in deeper penetration of K down the soil profile. The model predicts the movement of K through the soil profile and so the risk of leaching, rather than actual leaching loss.
Predicted distribution of K applied (a) at a rate of 200 kg K ha−1 as KCl on a clay loam soil, and (b) 800 kg K ha−1 as manure slurry on a sandy soil in the cultivated layer (20 cm) of each soil. (Johnston and Goulding 1992)
3.4.4 NUTMON
Smaling and Fresco (1993) developed the multilevel decision support model for monitoring the soil nutrient balance (NUTMON). Data collected from the Kisii District in Kenya were used to develop the model (Smaling et al. 1993), and the authors stated that the regional level was most appropriate for operating it.
In NUTMON, loss of K by erosion is based on estimates of soil erosion, using the universal soil loss equation (Smaling et al. 1993). Soil loss is multiplied by estimated total K concentration in the 0–20 cm layer of soil, which varies by clay content and exchangeable K. To account for the enrichment of K in eroded sediment, compared to the soil in situ, the estimated total K loss is multiplied by an enrichment factor. The result is multiplied by 0.75 to quantify net K loss. In a related effort, although not formally part of NUTMON, Lesschen et al. (2007) used the LAPSUS model (Schoorl et al. 2000, 2002) to estimate sediment transport rates, quantifying soil erosion as well as sedimentation.
NUTMON estimates the amount of K leached as a percentage of the K applied as fertilizer and manure (Smaling and Fresco 1993; Smaling et al. 1993). These percentages range from 0.5 to 1.0% and are adjusted for annual rainfall and clay content, with higher annual rainfall adjusting percentages toward 1.0% and higher clay content adjusting percentages the opposite direction, toward 0.5%. Lesschen et al. (2007) revised this methodology based on new algorithms developed for nitrogen (N) in NUTMON. They developed the following multiple regression model (Eq. 3.1) to estimate K leaching for a wide range of soils and climates:
where:
- P:
-
annual precipitation (mm)
- FK:
-
mineral and organic fertilizer addition (kg K ha−1)
- CEC:
-
cation exchange capacity (cmol(+) kg−1).
This equation was used to develop the K-leaching estimates used to calculate the K balances of sub-Saharan African countries presented in Chap. 11.
3.4.5 SVMLEACH-NK POTATO
Fortin et al. (2015) developed SVMLEACH-NK POTATO to simulate the daily dynamics of N and K leaching losses during the potato-growing season in Quebec, Canada. The model uses the least squares support vector machines (LS-SVMs) method, a machine learning technique that looks for patterns in data and performs regression in a high-dimension feature space (Fortin et al. 2014). The best-fit empirical model built from the training data set used the following variables: seasonal precipitation, seasonal temperature, N rate, day of year, and percent clay. Consequently, model users enter planting date, N application rate, percent clay, daily mean temperature, and daily total precipitation. The model relates nitrate leaching to K leaching, consistent with K+ being one cation forming an ion pair with NO3− in the soil solution during leaching.
3.4.6 SWAT-K
Wang et al. (2016) modified the Soil and Water Assessment Tool (SWAT) to simulate stream K load and K budgets of the Shibetwu River Watershed, an area of dairy farming in Japan where 95% of the agricultural land is under pasture. The SWAT-K model is an example of a more integrative K management approach. The model considers K losses in surface runoff, subsurface runoff (lateral flow), leaching, and erosion and simulates total K load at the outlet of the watershed. Wang et al. (2016) calibrated the model to measured levels of streamflow, suspended sediment load, and dissolved K.
3.5 Open Burning
Open burning is the unenclosed combustion of materials in an ambient environment (Lemieux et al. 2004). We limit our discussion to the burning of plant material in place at or near the soil surface, including wildfires, prescribed burning, burning for land clearing, and stubble burning (the burning of crop residues on agricultural land). Emissions from burning include particulate matter, nutrients, water, carbon dioxide, carbon monoxide, methane, volatile and semi-volatile organics, acid aerosols, metals, polychlorinated dibenzo-p-dioxins, polychlorinated dibenzofurans, and polychlorinated biphenyls (Lemieux et al. 2004). Potassium is among the nutrients lost in burning.
Gaudichet et al. (1995) identified three K-containing particles emitted from biomass burning: carbonaceous, drop-shaped microsoot particles ranging from 0.2 to 0.5μm contain K associated with sulfur (S); potassium salt condensed as cubic crystals of KCl less than 1μm in size and occurring either separately or embedded in other constituents associated with open flames; larger vegetation relics (greater than 1μm) containing K in partially combusted material. Long-distance transport is possible for all of these particles.
Potassium is the most abundant of the nutrients in particles emitted from burning vegetation. This is in contrast to emissions from fossil fuels that contain little K (Ruscio et al. 2016). Gaudichet et al. (1995) suggested that the presence of K could be used to differentiate biomass emissions from fossil fuel emissions. Support for such differentiation was provided by Amici et al. (2011) who used hyperspectral imaging to confirm that K emission was a characteristic of open flames from biomass burning.
We know relatively little about the quantity of K lost by open burning. The fact that K can be lost in this way is contrary to the belief that burning simply leaves K in the ash on the field. While ash does contain K, it clearly does not contain all the K that was in the plant material prior to burning. Further research will need to determine just how much K is lost during open burning.
3.6 Considerations for Potassium Recommendations
The development and implementation of Best Management Practices (BMP) for fertilizer use, with a focus on source, rate, time, and placement (i.e., the “4R” approach: right source, right rate, right time and right place), is necessary in the short-term to increase productivity and economic returns and in the long-term to provide more efficient ways of using non-renewable resources upon which food, feed, fiber, and fuel production depend (Fixen and Johnston 2012).
Source of K can be a factor when adjusting recommendations to limit losses. Most K sources (fertilizers, manures, composts, crop residues, and wastewaters) contain K in the simple cationic form of K+ (Arienzo et al. 2009; Stockdale et al. 2002), and inorganic fertilizers and organic manures are equally effective for meeting the K requirements of crops (Johnston and Goulding 1990). Organic sources, however, may sustain higher concentrations of solution K (Addiscott and Johnston 1975). To what extent these higher levels contribute to greater leaching or more efficient plant utilization has yet to be clarified in research. Enhanced-efficiency fertilizers (for example, Di and Cameron 2004; Gillman and Noble 2005; Yang et al. 2016) are a technological approach to increasing K use efficiency. The main advantage of these is that the K release may more closely match plant requirements through the season, reducing the risk of losses. However, in a greenhouse experiment, Bley et al. (2017) found that using slow-release (polymer coated) KCl reduced leaching compared to traditional KCl, but the slow rate of release was not sufficient to meet crop K demand during the initial phase of growth. The rate of adoption of technologies such as these is limited because of: the cost of the materials in comparison to traditional sources; the existence of regulatory policies (Gillman and Noble 2005); health and safety issues associated with their application (Timilsena et al. 2015); and sometimes a lack of information available on its impacts on productivity at the farm level.
The rate of K fertilizer recommended to farmers should account for K losses. While some decision tools exist, the focus has been on calculating more accurate nutrient budgets to eliminate overapplications. Adjusting K inputs in relation to estimated outputs to avoid a surplus at risk to leaching is a key factor for reducing K leaching losses. In tropical, coarse-textured soils managed under no-till, K leaching is high when fertilizer application exceeds plant demand (Rosolem and Steiner 2017). Process models that simulate K loss by water and wind erosion have yet to be developed, and even those that exist and estimate runoff and leaching losses for a given set of conditions have yet to be incorporated into algorithms that adjust K fertilizer rates recommended to farmers.
Timing of fertilizer applications is another approach to managing K losses. High K application rates may generate high K leaching losses, especially when drainage exceeds 500 mm (Bolton et al. 1970; Thomas and Hipp 1968). In fact, rainfall distribution and intensity are often more important than total precipitation (Quémener 1986) because of their impacts on surface runoff relative to the amount of matrix flow (Heathwaite et al. 1996). Potassium leaching losses may significantly increase when K fertilizer is applied to drained soils because of preferential flow, as discussed previously. In these situations, a key aspect to reduce K losses is the time interval between K application and the rainfall event: the longer this interval, the lower the losses (Alfaro et al. 2004a). Splitting a large application into two or more smaller applications is recommended when the risk of loss is high. On organic soils, K should be applied close to the time of active uptake by crops to avoid leaching, since these soils do not bind K tightly, even though they have a high CEC.
Fertilizer placement is also likely to be an effective way to reduce K loss, but almost no research has examined how various K placement methods affect it. Work on P has shown that subsurface banding of P fertilizers reduces runoff P losses compared to broadcast applications (Kimmel et al. 2001). The work cited earlier by Korucu (2018) showed that when K fertilizer was applied by surface broadcasting, a cover crop reduced K runoff losses. Sato et al. (2009), examining K placement in raised beds with seepage irrigation, suggested that evaluating K losses will require information about the height and seasonal dynamics of the water table. At the time of writing, it appears that no general guidance exists for placement strategies to minimize K losses.
3.7 Conclusions
Relative to other nutrients, K losses and transfers have not been well researched. Leaching has received the most attention, but erosion appears to be equally important in terms of the quantity of K lost. Dissolved K in runoff may contribute less to total loss than leaching and erosion. How much K is lost from open burning is still not well known. Potassium losses are associated particularly with losses of smaller soil particulates, which in turn are associated with loss of clay minerals. Not only is this loss of fertility detrimental in the short term, but it also appears to lead to reductions in K-holding capacity in the long term. Improved K management strategies must go beyond considering only fertilizer source, rate, time, and placement and be developed to incorporate strategies to maintain soil cover so that nutrients can be recycled more effectively. Building better decision support tools that incorporate process models will better inform farmers and help them make decisions that achieve the desired outcomes of efficient K use, including minimal losses.
References
Acharya GP, McDonald MA, Tripathi BP, Gardner RM, Mawdesley KJ (2007) Nutrient losses from rain-fed bench terraced cultivation systems in high rainfall areas of the mid-hills of Nepal. Land Degrad Dev 18(5):486–499. https://doi.org/10.1002/ldr.792
Addiscott TM, Johnston AE (1975) Potassium in soils under different cropping systems: 3. Nonexchangeable potassium in soils from long-term experiments at Rothamsted and Woburn. J Agric Sci 84(3):513–524. https://doi.org/10.1017/S0021859600052734
Alfaro MA, Jarvis SC, Gregory PJ (2003) Potassium budgets in grassland systems as affected by nitrogen and drainage. Soil Use Manag 19(2):89–95. https://doi.org/10.1111/j.1475-2743.2003.tb00286.x
Alfaro MA, Gregory PJ, Jarvis SC (2004a) Dynamics of potassium leaching on a hillslope grassland soil. J Environ Qual 33(1):192–200. https://doi.org/10.2134/jeq2004.1920
Alfaro MA, Jarvis SC, Gregory PJ (2004b) Factors affecting potassium leaching in different soils. Soil Use Manag 20(2):182–189. https://doi.org/10.1111/j.1475-2743.2004.tb00355.x
Alfaro M, Salazar F, Teuber N (2006) Potassium surface runoff and leaching losses in a beef cattle grazing system on volcanic soil. Better Crops 90(4):20–22
Alfaro M, Salazar F, Iraira S, Teuber N, Villarroel D, Ramírez L (2008) Nitrogen, phosphorus and potassium losses in a grazing system with different stocking rates in a volcanic soil. Chil J Agric Res 68(2):146–155
Amici S, Wooster MJ, Piscini A (2011) Multi-resolution spectral analysis of wildfire potassium emission signatures using laboratory, airborne and spaceborn remote sensing. Remote Sens Environ 115:1811–1823. https://doi.org/10.1016/j.rse.2011.02.022
Anguria P, Chemining’wa GN, Onwonga RN, Ugen MA (2017) Decomposition and nutrient release of selected cereal and legume crop residues. J Agric Sci 9(6):108–119. https://doi.org/10.5539/jas.v9n6p108
Arienzo M, Christen EW, Quayle W, Kumar A (2009) A review of the fate of potassium in the soil–plant system after land application of wastewaters. J Hazard Mater 164(2–3):415–422. https://doi.org/10.1016/j.jhazmat.2008.08.095
Armstrong AC, Garwood EA (1991) Hydrological consequences of artificial drainage of grassland. Hydrol Process 5:157–174. https://doi.org/10.1002/hyp.3360050204
Askegaard M, Eriksen J (2002) Exchangeable potassium in soil as indicator of potassium status in an organic crop rotation on loamy sand. Soil Use Manag 18(2):84–90. https://doi.org/10.1111/j.1475-2743.2002.tb00224.x
Askegaard M, Eriksen J, Olesen JE (2003) Exchangeable potassium and potassium balances in organic crop rotations on a coarse sand. Soil Use Manag 19(2):96–103. https://doi.org/10.1111/j.1475-2743.2003.tb00287.x
Bender RR, Haegele JW, Ruffo ML, Below FE (2013) Nutrient uptake, partitioning, and remobilization in modern, transgenic insect-protected maize hybrids. Agron J 105:161–170. https://doi.org/10.2134/agronj2012.0352
Bernal MP, Roig A, García D (1993) Nutrient balances in calcareous soils after application of different rates of pig slurry. Soil Use Manag 9(1):9–11. https://doi.org/10.1111/j.1475-2743.1993.tb00920.x
Bertol I, Guadagnin JC, González AP, do Amaral AJ, Brignoni LF (2005) Soil tillage, water erosion, and calcium, magnesium and organic carbon losses. Sci Agric 62(6):578–584
Bertoluzzi EC, dos Santos DR, Santanna MA, Caner L (2013) Mineralogy and nutrient desorption of suspended sediments during a storm event. J Soils Sediments 13:1093–1105. https://doi.org/10.1007/s11368-013-0692-4
Bley H, Gianello C, Santos LS, Selau LPR (2017) Nutrient release, plant nutrition, and potassium leaching from polymer-coated fertilizer. Rev Bras Ciênc Solo 41:e0160142. https://doi.org/10.1590/18069657rbcs20160142
Bolton EF, Aylesworth JW, Hore FR (1970) Nutrient losses through tile drains under three cropping systems and two fertility levels on a Brookston clay soil. Can J Soil Sci 50(3):275–279. https://doi.org/10.4141/cjss70-038
Brye KR, Norman JM (2004) Land-use effects on anion-associated cation leaching in response to above-normal precipitation. Acta Hydrochim Hydrobiol 32(3):235–248. https://doi.org/10.1002/aheh.200300534
Crusciol CAC, Garcia RA, Castro GSA, Rosolem CA (2011) Nitrate role in basic cation leaching under no-till. Rev Bras Ciênc Solo 35(6):1975–1984
de Barros I, Williams JR, Gaiser T (2004) Modeling soil nutrient limitations to crop production in semiarid NE of Brazil with a modified EPIC version. I. Changes in the source code of the model. Ecol Model 178:441–456. https://doi.org/10.1016/j.ecolmodel.2004.04.015
De M, Moore AD, Mikkelsen RL (2019) In-season accumulation and partitioning of macronutrients and micronutrients in irrigated sugar beet production. J Sugar Beet Res 56(3–4):54–78. https://doi.org/10.5274/Jsbr.56.3.56
Di HJ, Cameron KC (2004) Effects of the nitrification inhibitor dicyandiamide on potassium, magnesium and calcium leaching in grazed grassland. Soil Use Manag 20(1):2–7. https://doi.org/10.1111/j.1475-2743.2004.tb00330.x
Fixen PE, Johnston AM (2012) World fertilizer nutrient reserves: a view to the future. J Sci Food Agr 92(5):1001–1005. https://doi.org/10.1002/jsfa.4532
Fortin JG, Morais A, Anctil F, Parent LE (2014) Comparison of machine learning regression methods to simulate NO3 flux in soil solution under potato crops. Appl Math 5:832–841. https://doi.org/10.4236/am.2014.55079
Fortin JG, Morais A, Anctil F, Parent LE (2015) SVMLEACH-NK POTATO: a simple software tool to simulate nitrate and potassium co-leaching under potato crop. Comput Electron Agric 110:259–266. https://doi.org/10.1016/j.compag.2014.11.025
Gaudichet A, Echalar F, Chatenet B, Quisefit JP, Malingre G, Cachier H, Buat-Menard P, Artaxo P, Maenhaut W (1995) Trace elements in tropical African savanna biomass burning aerosols. J Atmos Chem 22:19–39. https://doi.org/10.1007/BF00708179
Gillman G, Noble A (2005) Environmentally manageable fertilizers: a new approach. Environ Qual Manag 15(2):59–70. https://doi.org/10.1002/tqem.20081
Heathwaite AL, Johnes PJ, Peters NE (1996) Trends in nutrients. Hydrol Process 10(2):263–293. https://doi.org/10.1002/(SICI)1099-1085(199602)10:2<263::AID-HYP441>3.0.CO;2-K
Johnston AE (2003) Understanding potassium and its use in agriculture. European Fertilizer Manufacturers Association, Brussels. http://www.pda.org.uk/wp/wp-content/uploads/2015/08/EFMA_Potassium_booklet_2003.pdf. Accessed 14 May 2020
Johnston AE, Goulding KWT (1990) The use of plant and soil analyses to predict the potassium supplying capacity of soil. In: Proceedings of the 22nd colloquium of the international potash institute: development of K-fertilizer recommendations, International Potash Institute, Basel, pp 177–204. https://ipipotash.org/en/. Accessed 14 May 2020
Johnston AE, Goulding KWT (1992) Potassium concentrations in surface and groundwaters and the loss of potassium in relation to land use. In: Proceedings of the 23rd colloquium of the International Potash Institute: potassium in ecosystems: biogeochemical fluxes of cations in agro- and forest-systems. International Potash Institute, Basel, pp 177–204. https://ipipotash.org/en/. Accessed 14 May 2020
Kimmel RJ, Pierzynski GM, Janssen KA, Barnes PL (2001) Effects of tillage and phosphorus placement on phosphorus runoff losses in a grain sorghum-soybean rotation. J Environ Qual 30:1324–1330. https://doi.org/10.2134/jeq2001.3041324x
Kolahchi Z, Jalali M (2007) Effect of water quality on the leaching of potassium from sandy soil. J Arid Environ 68(4):624–639. https://doi.org/10.1016/j.jaridenv.2006.06.010
Korucu T, Shipitalo MJ, Kaspar TC (2018) Rye cover crop increases earthworm populations and reduces losses of broadcast, fall-applied, fertilizers in surface runoff. Soil Till Res 180:99–106. https://doi.org/10.1016/j.still.2018.03.004
Lahav E, Turner DW (1989) Banana nutrition. International Potash Institute Bulletin 7. Berne
Lemieux PM, Lutes CC, Santoianni DA (2004) Emissions of organic air toxics from open burning: a comprehensive review. Prog Energ Combust 30:1–32. https://doi.org/10.1016/j.pecs.2003.08.001
Lesschen JP, Stoorvogel JJ, Smaling EMA, Heuvelink GBM, Veldkamp A (2007) A spatially explicit methodology to quantify soil nutrient balances and their uncertainties at the national level. Nutr Cycl Agroecosyst 78(2):111–131. https://doi.org/10.1007/s10705-006-9078-y
Muhammad S, Luedeling E, Brown PH (2009) A nutrient budget approach to nutrient management in almond. Proc Inter Plant Nutr Coll XVI. http://ucanr.org/sites/nm/files/76672.pdf. Accessed 14 May 2020
Nathan MV, Sun Y, Dunn D (2009) Nutrient removal values for major agronomic crops in Missouri report for 2006–2007. https://www.researchgate.net/publication/268082826. Accessed 14 May 2020
Nicholson FA, Bhogal A, Chadwick D, Gill E, Gooday RD, Lord E, Misselbrook T, Rollett AJ, Sagoo E, Smith KA, Thorman RE, Williams JR, Chambers BJ (2013) An enhanced software tool to support better use of manure nutrients: MANNER-NPK. Soil Use Manag 29(4):473–484. https://doi.org/10.1111/sum.12078
Quémener J (1986) Nutrient balances and need for potassium. In: Proceedings of the 13th international potash institute congress. International Potash Institute, Basel, pp 41–69. https://ipipotash.org/en/. Accessed 14 May 2020
Rogers CW, Dari B, Hu G, Mikkelsen R (2019) Dry matter production, nutrient accumulation, and nutrient partitioning of barley. J Plant Nutr Soil Sci 182:367–373. https://doi.org/10.1002/jpln.201800336
Rosolem CA, Steiner F (2017) Effects of soil texture and rates of K input on potassium balance in tropical soil. Eur J Soil Sci 68:658–666. https://doi.org/10.1111/ejss.12460
Rosolem CA, Sgariboldi T, Garcia RA, Calonego JC (2010) Potassium leaching as affected by soil texture and residual fertilization in tropical soils. Commun Soil Sci Plan 41(16):1934–1943. https://doi.org/10.1080/00103624.2010.495804
Rousseva S, Torri D, Pagliai M (2002) Effect of rain on the macroporosity at the soil surface. Eur J Soil Sci 53(1):83–94. https://doi.org/10.1046/j.1365-2389.2002.00426.x
Ruscio A, Kazanc F, Levendis YA (2016) Comparison of fine ash emissions generated from biomass and coal combustion and valuation of predictive furnace deposition indices: a review. J Energ Eng 142:E4015007. https://doi.org/10.1061/(ASCE)EY.1943-7897.0000310
Sato S, Morgan KT, Ozores-Hampton M, Simonne EH (2009) Spatial and temporal distributions in sandy soils with seepage irrigation: II. Phosphorus and potassium. Soil Sci Soc Am J 73:1053–1060. https://doi.org/10.2136/sssaj2008.0114
Schoorl JM, Sonneveld MPW, Veldkamp A (2000) Three-dimensional landscape process modelling: the effect of DEM resolution. Earth Surf Proc Land 25(9):1025–1034. https://doi.org/10.1002/1096-9837(200008)25%3A9<1025%3A%3AAID-ESP116>3.0.CO%3B2-Z
Schoorl JM, Veldkamp A, Bouma J (2002) Modeling water and soil redistribution in a dynamic landscape context. Soil Sci Soc Am J 66(5):1610–1619. https://doi.org/10.2136/sssaj2002.1610
Sekhon GS (1982) Potassium recycling in agriculture. In: Food and Fertilizer Technology Centre (ed) Recycling of potassium and phosphorus in agriculture, Tech Bull 69. Food and Fertilizer Technology Centre, Taiwan, pp 1–15
Smaling EMA, Fresco LO (1993) A decision-support model for monitoring nutrient balances under agricultural land use (NUTMON). Geoderma 60(1–4):235–256. https://doi.org/10.1016/0016-7061(93)90029-K
Smaling EMA, Stoorvogel JJ, Sindmeijer PN (1993) Calculating soil nutrient balances in Africa at different scales. II District scale. Fert Res 35:237–250. https://doi.org/10.1007/BF00750642
Stockdale EA, Shepherd MA, Fortune S, Cuttle SP (2002) Soil fertility in organic farming systems – fundamentally different? Soil Use Manag 18.(S1:301–308. https://doi.org/10.1111/j.1475-2743.2002.tb00272.x
Tarkalson DD, Brown B, Kok H, Bjorneberg DL (2009) Impact of removing straw from wheat and barley fields: a literature review. Better Crops 93(3):17–19
Thomas GW, Hipp BW (1968) Soil factors affecting potassium availability. In: Kilmer VJ (ed) The role of potassium in agriculture. American Society of Agronomy, Crop Science Society of America, Soil Science Society of America, Madison, pp 269–291. https://doi.org/10.2134/1968.roleofpotassium.c13
Timilsena YP, Adhikari R, Casey P, Muster T, Gill H, Adhikari B (2015) Enhanced efficiency fertilizers: a review of formulation and nutrient release patterns. J Sci Food Agr 95(6):1131–1142. https://doi.org/10.1002/jsfa.6812
Unkovich M, Baldock J, Forbes M (2010) Variability in harvest index of grain crops and potential significance for carbon accounting: examples from Australian agriculture. Adv Agron 105:173–219. https://doi.org/10.1016/S0065-2113(10)05005-4
Wang C, Jiang R, Boithias L, Sauvage S, Sánchez-Pérez J-M, Mao X, Han Y, Hayakawa A, Kuramochi K, Hatano R (2016) Assessing potassium environmental losses from a dairy farming watershed with the modified SWAT model. Agric Water Manage 175:91–104. https://doi.org/10.1016/j.agwat.2016.02.007
Wang X, Lang L, Hua T, Li H, Zhang C, Ma W (2018) Effects of aeolian processes on soil nutrient loss in the Gonghe Basin, Qinghai-Tibet Plateau: an experimental study. J Soils Sediments 18:229–238. https://doi.org/10.1007/s11368-017-1734-0
Wheeler DM, Ledgard SF, DeKlein CAM, Monaghan RM, Carey PL, McDowell RW, Johns KL (2003) OVERSEER® nutrient budgets – moving towards on-farm resource accounting. Proc NZ Grassland Assoc 65:191–194. https://www.grassland.org.nz/proceedings.php. Accessed 14 May 2020
Wheeler DM, Ledgard SF, DeKlein CAM (2008) Using the OVERSEER nutrient budget model to estimate on-farm greenhouse gas emissions. Aust J Exp Agric 48(1–2):99–103. https://doi.org/10.1071/EA07250
Williams PH, Haynes RJ (1992) Balance sheet of phosphorus, sulphur and potassium in a long-term grazed pasture supplied with superphosphate. Fert Res 31(1):51–60. https://doi.org/10.1007/BF01064227
Williams PH, Gregg PEH, Hedley MJ (1990) Mass balance modelling of potassium losses from grazed dairy pasture. New Zeal J Agric Res 33(4):661–668. https://doi.org/10.1080/00288233.1990.10428470
Yan Y, Wang X, Guo Z, Chen J, Xin X, Xu D, Yan R, Chen B, Xu L (2018) Influence of wind erosion on dry aggregate size distribution and nutrients in three steppe soils in northern China. Catena 170:159–168. https://doi.org/10.1016/j.catena.2018.06.013
Yang X, Geng J, Li C, Zhang M, Chen B, Tian X, Zheng W, Liu Z, Wang C (2016) Combined application of polymer coated potassium chloride and urea improved fertilizer use efficiencies, yield and leaf photosynthesis of cotton on saline soil. Field Crops Res 197:63–73. https://doi.org/10.1016/j.fcr.2016.08.009
Zöbisch MA, Richter C, Heiligtag B, Schlott R (1995) Nutrient losses from cropland in the Central Highlands of Kenya due to surface runoff and soil erosion. Soil Till Res 33:109–116. https://doi.org/10.1016/0167-1987(94)00441-G
Acknowledgments
Marta A. Alfaro, Deputy Director of National Research and Development, Instituto de Investigaciones Agropecuarias, Ministerio de Agricultura, Osorno, Chile, led the discussions on this topic at the workshop in Rome and contributed to an early version of the chapter.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Open Access This chapter is licensed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license and indicate if changes were made.
The images or other third party material in this chapter are included in the chapter's Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the chapter's Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
Copyright information
© 2021 The Author(s)
About this paper
Cite this paper
Goulding, K. et al. (2021). Outputs: Potassium Losses from Agricultural Systems. In: Murrell, T.S., Mikkelsen, R.L., Sulewski, G., Norton, R., Thompson, M.L. (eds) Improving Potassium Recommendations for Agricultural Crops. Springer, Cham. https://doi.org/10.1007/978-3-030-59197-7_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-59197-7_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-59196-0
Online ISBN: 978-3-030-59197-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)