Abstract
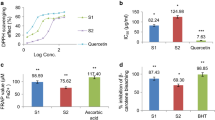
The anti-inflammatory steroidal and non-steroidal drugs are commonly used to treat inflammation. Be that as it may, with a number of side effects, these are disabled. We have therefore selected two plants, i.e., Mucuna pruriens and Allium sativum, which have various therapeutic agents used in traditional medicines and Ayurveda. The objective of this investigation was to test in vitro anti-inflammatory activities of Mucuna pruriens leaves and Allium sativum bulb extracts of both either alone or in combination by evaluating inhibition of cyclooxygenase and with determination of protein denaturation. The successive solvent extraction of shade dried leaves and peeled dry bulbs was performed for both plants with solvents of increased polarity. Then the two plants’ in vitro antioxidant assay was evaluated by estimating DPPH, H2O2, NO, ABTS and ascorbic acid as standard at various concentrations. The methanol and combined methanol extracts from the two plants showed significant antioxidant activity on DPPH, H2O2, NO and ABTS among the four extracts. In addition, the cyclooxygenase inhibition (76.23%) and protein denaturation (65%) were also tested for anti-inflammatory activity, which revealed that these two plants had strong antioxidant and anti-inflammatory principles.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
S. Bukke, P.S. Raghu, G. Sailaja, T.R. Kedam, The study on morphological, phytochemical and pharmacological aspects of Rhinacanthus nasutus. (L) Kurz (a review). J. Appl. Pharm. Sci. 1(8), 26–32 (2011)
C. Sangita, C. Priyanka, D. Protapaditya, B. Sanjib, Evaluation of in vitro anti-inflammatory activity of coffee against the denaturation of protein. Asian Pac. J. Trop. Biomed. 2, 178–180 (2012)
D. Arya, V. Patni, Pharmacognostic profile and phytochemical investigation of Pluchea Lanceolata Oliver & Hiern in vivo and in vitro. Int. J. Pharm. Sci. Rev. Res. 22(2), 157–161 (2013)
A. Gurib-Fakim, Medicinal plants: traditions of yesterday and drugs tomorrow. Mol. Aspects Med. 27, 1–93 (2006)
V.R. Winrow, P.G. Winyard, C.J. Morris, D.R. Blake, Free radicals in inflammation: second messengers and mediators of tissue destruction. Br. Med. Bull. 49, 506–522 (1993)
S.R. Maxwell, Prospects for the use of antioxidant therapies. Drugs 49, 345–361 (1995)
A. Hausladen, J.S. Stamer, Nirosative stress method in enzymology. 300, 389–395 (1999)
G. Leelaprakash, S. Mohan Dass, In vitro anti-inflammatory activity of methanol extract of Enicostemma axillare. Int. J. Drug Dev. Res. 3, 189–196 (2010)
P.V. Ingle, D.M. Patel, C-reactive protein in various disease conditions—an overview. Asian J. Pharm. Clin. Res. 4(1), 9–13 (2011)
S. Jamuna, S. Paulsamy, K. Karthika, Screening of in vitro antioxidant activity of methanolic leaf and root extracts of Hypochaeris radicata L. (Asteraceae). J. Appl. Pharm. Sci. 2(7), 149–154 (2012)
B.J. Divya, B. Suman, M. Venkataswamy, K. ThyagaRaju, The traditional uses and pharmacological activities of Mucuna pruriens (L) DC: a comprehensive review. Indo Am. J. Pharm. Res. 7(01), 7516–7525 (2017)
W. Lewis, M. Elvin-Lewis, Medical Botany: Plants Affecting Human Health, 2nd edn. (Wiley, New York, 2003)
B.J. Divya, B. Suman, L. Lakshman Kumar, M. Venkataswamy, B. Eswari, K. Thyagaraju, The role of Allium sativum (Garlic) in various diseases and its health benefits: a comprehensive review. Int. J. Adv. Res. 5(8), 592–602 (2017)
M. Burits, F. Bucar, Antioxidant activity of Nigella sativa essential oil. Phytother. Res. 14(5), 323–328 (2000)
M. Vijayabhaskaran, N. Venkateshwaramurthy, G. Babu, P. Perumal, In vitro antioxidant evaluation of Pseudarthria viscid. Int. J. Curr. Pharm. Res. 2, 21–23 (2010)
D.C. Garratt, The Quantitative Analysis of Drugs, vol. 3 (Japan, Chapman and Hall Ltd, 1964), pp. 456–458
N. Pellegrini, A. Proteggente, M.Y. Pannala, C. Rice-Evans, Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 26, 1231–1237 (1999)
S. Sakat, A.R. Juvekar, M.N. Gambhire, In vitro antioxidant and anti-inflammatory activity of methanol extract of Oxalis corniculata Linn. Int. J. Pharma Pharmacol. Sci. 2(1), 146–155 (2010)
B. Arne, Separation of white blood cells. J. Nat. 204, 793–794 (1964)
R.A. Copeland, J.M. Williams, J. Giannaras, S. Nurnberg, M. Covington, D. Pinto, S. Pick, J.M. Trazaskos, Mechanism of selective inhibition of the inducible isoform of prostaglandin G/H synthase. Proc. Natl. Acad. Sci. U.S.A. 91(23), 11202–11206 (1994)
Z.A. Zakaria, M.S. Rofiee, L.K. The, M.Z. Salleh, M.R. Sulaiman, M.N. Somchit, Bauhinia purpurea leaves extracts exhibited in vitro antiproliferative and antioxidant activities. Afr. J. Biotech. 10(1), 65–74 (2011)
M.A.A. Rahman, S.S. Moon, Antioxidant polyphenol glycosides from the plant Draba nemorosa. Bull. Korean Chem. Soc. 28(5), 827–831 (2007)
O.I. Aruoma, Free radicals, antioxidants and international nutrition. Asia Pac. J. Clin. Nut. 8, 53 (1999)
R. Puja, V.P. Karthik, A comparative study of in-vitro nitric oxide scavenging activity of Balofloxacinvs prulifloxacin. Asian J. Pharm. Clin. Res. 10(1), 380–382 (2017)
M.R. Saha, J. Rumana, M.M.I. Vhuiyan, I.J. Biva, In vitro nitric oxide scavenging activity of ethanol leaf extracts of four Bangladeshi medicinal plants. Stamford J. Pharm. Sci. 1(1&2), 57–62 (2008)
C. Rice-Evans, J. Sampson, P.M. Bramley, D.E. Holloway, Why do we expect carotenoids to be antioxidants in vivo. Free Rad. Res. 26, 381–398 (1997)
L.V. Jorgensen, H.L. Madsen, M.K. Thomsen, L.O. Dragsted, L.H. Skibsted, Regulation of phenolic antioxidants from phenoxyl radicals: an ESR and electrochemical study of antioxidant hierarchy. Free Rad. Res. 30, 207–220 (1999)
O.K. Kim, A. Murakami, Y. Nakamura, H. Oihigashi, Screening of edible Japanese plants for nitric oxide generation inhibitory activities in RAW 264.7 cells. Cancer Lett. 125, 199–207 (1998)
H.K. Kim, B.S. Choen, Y.H. Kim, S.Y. Kim, H.P. Kim, Effects of naturally occurring flavonoids on nitric oxide production in the macrophage cell line RAW 264.7 and their structure activity relationship. Biochem. Pharmacol. 58, 759–765 (1999)
G.C. Jagetia, S.K. Rao, M.S. Baliga, S.K. Babu, The evaluation of nitric oxide scavenging activity of certain herbal formulations in vitro: a preliminary study. Phytother. Res. 18(7), 561–565 (2004)
V. Steenkamp, M.J. Stewart, L. Chimuka, E. Cukrowska, Uranium concentrations in South African herbal remedies. Health Physiol. 89, 79–83 (2005)
L. Yu, S. Haley, J. Perret, M. Harris, J. Wilson, M. Qian, Free radical scavenging properties of wheat extracts. J. Agric. Food Chem. 50, 1619–1624 (2002)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this paper
Cite this paper
Divya, B.J., Suman, B., Venkataswamy, M., Mouli, K.C., Raju, K.T. (2020). An Analysis of In Vitro Antioxidant and Anti-inflammatory Activities of Mucuna pruriens (Leaves) and Allium sativum (Bulbs). In: Jyothi, S., Mamatha, D., Satapathy, S., Raju, K., Favorskaya, M. (eds) Advances in Computational and Bio-Engineering. CBE 2019. Learning and Analytics in Intelligent Systems, vol 15. Springer, Cham. https://doi.org/10.1007/978-3-030-46939-9_14
Download citation
DOI: https://doi.org/10.1007/978-3-030-46939-9_14
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-46938-2
Online ISBN: 978-3-030-46939-9
eBook Packages: Mathematics and StatisticsMathematics and Statistics (R0)