Abstract
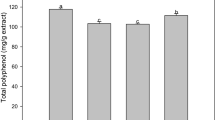
The antioxidant and anti-inflammatory potential of Heritiera fomes ethanolic extract was evaluated.Query Ethanolic extraction of dried and grinded plant leaves and bark was performed. Antioxidant activity was evaluated by 1, 1-diphenyl-2-picryl-hydrazil (DPPH) free radical scavenging assay, ferric reducing antioxidant power (FRAP) and β-carotene bleaching inhibition assay. Total phenolic and flavonoid content were also determined. Anti-inflammatory activity was evaluated by in vitro human RBC membrane stabilizing assay and in vivo mice paw edema test. Ethanolic bark and leaf extracts of H. fomes (S1 and S2, respectively) showed significant scavenging effect on DPPH scavenging 68.51 µg/ml for S1 and 108.64 µg/ml for S2. Potent reduction potential (FRAP) was observed in S1 (83.75 mM of ferrous equivalent) while the FRAP of standard ascorbic acid was found 86.66 μM of ferrous equivalent. S1 also exhibited 87.43% β-carotene bleaching inhibition as comparable to standard butylated hydroxytoluene with 98.85% inhibition. Substantial phenolic and flavonoid content was also observed in S1 (136.83 μg gallic acid equivalent/g of extract and 76.67 μg quercetin equivalent/g of extract, respectively). S1 could provide 61.73% and 53.27% protection of RBC membrane in hypotonicity and heat induced lysis of RBC, respectively while the standard diclofenac sodium provided 91.66% and 86.66% protection, respectively. Furthermore, 44.16% reduction in mice paw edema at 40 μg/kg bw concentration was observed in S1 as compared to standard indomethacine (53.54% in 10 μg/kg bw concentration). The results revealed that the bark extract (S1) of H. fomes possesses significant antioxidant and anti-inflammatory activity.



Similar content being viewed by others
References
Abe H, Katada K, Orita M, Nishikibe M (1991) Effects of calcium antagonists on the erythrocyte membrane. J Pharm Pharmacol 43:22–26
Droge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82:47–95. https://doi.org/10.1152/physrev.00018.2001
Fu L, Xu BT, Xu XR, Qin XS, Gan RY, Li HB (2010) Antioxidant capacities and total phenolic contents of 56 wild fruits from South China. Molecules 15:8602–8617. https://doi.org/10.3390/molecules15128602
Fu L, Xu B-T, Xu X-R, Gan R-Y, Zhang Y, Xia E-Q, Li H-B (2011a) Antioxidant capacities and total phenolic contents of 62 fruits. Food Chem 129:345–350
Fu L, Xu BT, Gan RY, Zhang Y, Xu XR, Xia EQ, Li HB (2011b) Total phenolic contents and antioxidant capacities of herbal and tea infusions. Int J Mol Sci 12:2112–2124. https://doi.org/10.3390/ijms12042112
Hossain M, Saha S, Asadujjaman M, Kahan S (2013a) Analgesic, antioxidant and antibacterial activity of Smilax zeylanica Linn. (family-Smilacaceae). Pharmacologyonline 1:244–250
Hossain MA, Panthi S, Asadujjaman M, Khan SA, Ferdous F, Sadhu SK (2013b) Phytochemical and pharmacological assessment of the ethanol leaves extract of Heritiera fomes Buch. Ham. (family-Sterculiaceae). J Pharmacogn Phytochem 2:95–101
Jenab M et al (2006) Plasma and dietary vitamin C levels and risk of gastric cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC-EURGAST). Carcinogenesis 27:2250–2257. https://doi.org/10.1093/carcin/bgl096
Ksiksi T, Hamza AA (2012) Antioxidant, lipoxygenase and histone deacetylase inhibitory activities of Acridocarpus orientalis from Al Ain and Oman. Molecules 17:12521–12532. https://doi.org/10.3390/molecules171112521
Kumar V, Tyagi D (2013) Phytochemical screening and free-radical scavenging activity of Bergenia stracheyi. J Pharmacogn Phytochem 2:175–180
Li AN, Li S, Zhang YJ, Xu XR, Chen YM, Li HB (2014) Resources and biological activities of natural polyphenols. Nutrients 6:6020–6047. https://doi.org/10.3390/nu6126020
Lim YY, Quah EPL (2007) Antioxidant properties of different cultivars of Portulaca oleracea. Food Chem 103:734–740. https://doi.org/10.1016/j.foodchem.2006.09.025
Mahdi-Pour B, Jothy SL, Latha LY, Chen Y, Sasidharan S (2012) Antioxidant activity of methanol extracts of different parts of Lantana camara. Asian Pac J Trop Biomed 2:960–965. https://doi.org/10.1016/S2221-1691(13)60007-6
Manach C, Scalbert A, Morand C, Remesy C, Jimenez L (2004) Polyphenols: food sources and bioavailability. Am J Clin Nutr 79:727–747. https://doi.org/10.1093/ajcn/79.5.727
Martindale JL, Holbrook NJ (2002) Cellular response to oxidative stress: signaling for suicide and survival. J Cell Physiol 192:1–15. https://doi.org/10.1002/jcp.10119
Peng C et al (2014) Biology of ageing and role of dietary antioxidants. Biomed Res Int 2014:831841. https://doi.org/10.1155/2014/831841
Pidgeon GP, Lysaght J, Krishnamoorthy S, Reynolds JV, O’Byrne K, Nie D, Honn KV (2007) Lipoxygenase metabolism: roles in tumor progression and survival. Cancer Metastasis Rev 26:503–524. https://doi.org/10.1007/s10555-007-9098-3
Rebaya A et al (2014) Total phenolic, total flavonoid, tannin content, and antioxidant capacity of Halimium halimifolium (Cistaceae). J Appl Pharm Sci 5:52–57
Sarkhel S (2016) Evaluation of the anti-inflammatory activities of Quillaja saponaria Mol. saponin extract in mice. Toxicol Rep 3:1–3. https://doi.org/10.1016/j.toxrep.2015.11.006
Shinde U, Phadke A, Nair A, Mungantiwar A, Dikshit V, Saraf M (1999) Membrane stabilizing activity—a possible mechanism of action for the anti-inflammatory activity of Cedrus deodara wood oil. Fitoterapia 70:251–257
Simlai A, Roy A (2013) Biological activities and chemical constituents of some mangrove species from Sundarban estuary: an overview. Pharmacogn Rev 7:170–178. https://doi.org/10.4103/0973-7847.120518
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Viticult 16:144–158
Szollosi R, Varga IS (2002) Total antioxidant power in some species of Labiatae (adaptation of FRAP method). Acta Biologica Szegediensis 46:125–127
Takada H, Kokubo K, Matsubayashi K, Oshima T (2006) Antioxidant activity of supramolecular water-soluble fullerenes evaluated by beta-carotene bleaching assay. Biosci Biotechnol Biochem 70:3088–3093. https://doi.org/10.1271/bbb.60491
Talukder C, Saha S, Adhikari S, Mondal HK, Islam MK, Anisuzzman M (2012) Evaluation of antioxidant, analgesic and antidiarrhoeal activity of Flacourtia jangomas (Lour.) Raeusch. leaves. Pharmacologyonline 3:20–28
Ueno H, Yamakura S, Arastoo RS, Oshima T, Kokubo K (2014) Systematic evaluation and mechanistic investigation of antioxidant activity of fullerenols using β-carotene bleaching assay. J Nanomater 2014:7. https://doi.org/10.1155/2014/802596
Umapathy E, Ndebia E, Meeme A, Adam B, Menziwa P, Nkeh-Chungag B, Iputo J (2010) An experimental evaluation of Albuca setosa aqueous extract on membrane stabilization, protein denaturation and white blood cell migration during acute inflammation. J Med Plants Res 4:789–795
Wangensteen H, Dang HC, Uddin SJ, Alamgir M, Malterud KE (2009) Antioxidant and antimicrobial effects of the mangrove tree Heritiera fomes. Nat Prod Commun 4:371–376
Winter CA, Risley EA, Nuss GW (1962) Carrageenin-induced edema in hind paw of the rat as an assay for anti-inflammatory drugs. In: Proceedings of the society for experimental biology and medicine society for experimental biology and medicine, New York, vol 111, pp 544–547
Wojdyło A, Oszmiański J, Czemerys R (2007) Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chem 105:940–949. https://doi.org/10.1016/j.foodchem.2007.04.038
Xu D-P et al (2017) Natural antioxidants in foods and medicinal plants: extraction assessment and resources. Int J Mol Sci 18:96. https://doi.org/10.3390/ijms18010096
Zheng J, Zhou Y, Li Y, Xu DP, Li S, Li HB (2016) Spices for prevention and treatment of cancers. Nutrients 8:495. https://doi.org/10.3390/nu8080495
Zhishen J, Mengcheng T, Jianming W (1999) The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem 64:555–559. https://doi.org/10.1016/S0308-8146(98)00102-2
Zhou Y, Zheng J, Li Y, Xu DP, Li S, Chen YM, Li HB (2016) Natural polyphenols for prevention and treatment of cancer. Nutrients. https://doi.org/10.3390/nu8080515
Acknowledgements
Experimental facilities were provided by Biochemistry and Molecular Biology Laboratory, Khulna University, Bangladesh and Bangladesh Council of Scientific and Industrial Research (BCSIR). We acknowledge the Sundarbans east division, Ministry of Environment, Forest and Climate change, Government of Bangladesh for necessary administrative and logistic support in collecting plant sample material.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical statement
Ethical ideologies and rules for Scientific Experiments on Animals originated by the Swiss Academy of Sciences and the Helsinki Declaration in 2000 and the animal ethics and regulation of Khulna University, Bangladesh (Research Ref. No. KUAEC-2017/08/15) were strictly followed. To draw human blood (RBC membrane stabilizing assay), procedure and rules of Bangladesh Medical Research Council was strictly obeyed.
Conflict of interest
Md. Emdadul Islam has no conflict of interest. Kazi Mohammed Didarul Islam has no conflict of interest. Md. Morsaline Billahhas has no conflict of interest. Rana Biswas has no conflict of interest. Md. Hossain Sohrabhas has no conflict of interest. S. M. Mahbubur Rahmanhas has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Islam, M.E., Islam, K.M.D., Billah, M.M. et al. Antioxidant and anti-inflammatory activity of Heritiera fomes (Buch.-Ham), a mangrove plant of the Sundarbans. ADV TRADIT MED (ADTM) 20, 189–197 (2020). https://doi.org/10.1007/s13596-019-00401-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13596-019-00401-0