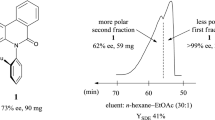
Abstract
The present review generalizes results of a comprehensive experimental study (by means of PXRD, SCXRD, TRPXRD, DSC, IR) of six chiral systems. These are enantiomeric systems of a single substance: (1) the system with a eutectic point (S- and R-E3ClMA), (2) the system forming a binary compound (l- and d-phenylglycine), and (3) the system forming equimolar and non-equimolar discrete compounds (S- and R-malic acid). Also these are diastereomeric systems and a system composed of enantiomers of different substances: (4) the diastereomeric system with continuous solid solutions (l- and l-allo-threonine), and (5) the diastereomeric system with a eutectic point (d- and l-allo-threonine), and (6) the system of enantiomers of different substances (l-valine and l-isoleucine) which contains a non-equimolar discrete compound. Crystal structures of discrete equimolar and non-equimolar homomolecular and heteromolecular compounds found in the systems under discussion are described. Also, polymorph diversity and the limits of solid solutions in such binary systems are discussed.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Notes
- 1.
The designations represent the relative (l and d) and absolute (S and R) configurations of the levorotatory and dextrorotatory enantiomers, respectively.
- 2.
A chiral center can take place not only at a stereogenic atom, but also in a “space”, for example, in the molecule center of gravity. Not only a chiral center, but also a chiral axis, and a chiral plane can be a source of chirality.
- 3.
References
Aakeröy CB, Cooke TI, Nieuwenhuyzen M (1996) The crystal structure of the molecular cocrystal L-malic acid L-tartaric acid (1/1). Supramol Chem 7(2):153–156
Andersson M, Fredga A, Jerslev B (1966) Anomalous racemates of malic acid. Acta Chem Scand 20:1060–1063
Anischenko LA, Shanina SN (2010) Amino acids in the depths: the results of the study and the prospects for the study. Bull Inst Geol, Komi SC UB RAS 9:34–35 (in Russian)
Bada JL, Schroeder RA (1975) Amino acid racemization reactions and their geochemical implications. Naturwissenschaften 62(2):71–79
Bergmann M, Lissitzin M (1930) Die überzähligen Stereoisomeren der γ-Amino-β-oxy-buttersäure. Ber Dtsch Chem Ges 63(2):310–313
Binev D, Taratin N, Kotelnikova E, Seidel-Morgenstern A, Lorenz H (2014) Solubility equilibria and crystallographic characterization of the L-threonine/L-allo-threonine system. Part 1: solubility equilibria in the threonine diastereomeric system. Cryst Growth Des 14:367–373
Bredikhin AA, Bredikhina ZA, Zakharychev DV, Samigullina AI, Gubaidullin AT (2015) 4-benzoylamino-3-hydroxybutyric acid, historically first “Anomalous Racemate”: reinvestigation. Cryst Growth Des 15(3):1362–1373
CCDC files: 1903255 (L-phenylglycine), 1904158 (compound I3L), 1903257 (compound V3L)
CSD files (identifiers): FIVGEW (DL-phenilglycine), COFRUK10 (S-malic acid), DLMALC11 (RSI-malic acid), DLMALC (RSII-malic acid), LVALIN01 (L-valine) and LISLEU02 (L-isoleucine)
Dalhus B, Görbitz CH (1999) Molecular aggregation in crystalline 1:1 complexes of hydrophobic D- and L-amino acids. I. The L-isoleucine series. Acta Cryst B55(3):424–431
De Torres T, Ortiz JE, Arribas I, Delgado A, Julia R, Martin-Rubi JA (2010) Geochemistry of Persististrombus latus Gmelin from the Pleistocene Iberian Mediterranean realm. Lethaia 43:149–163
Drozdova TV (1977) Geochemistry of amino acids. Nauka, Moscow (in Russian)
Eddleston MD, Arhangelskis M, Fričšić T, Jones W (2012) Solid state grinding as a tool to aid enantiomeric resolution by cocrystallization. Chem Commun 48:11340–11342
Fredga A (1940) Über den konfigurativen Zusammenhang zwischen optisch aktiver Milchsäure und Thiomilchsäure. Arkiv Kemi Mineral Geol 14B(12):1–10
Fredga A (1944) Proceedings of the Theodor Svedberg. Uppsala and Stockholm, Sweden, p 261
Galan K, Eicke MJ, Elsner MP, Lorenz H, Seidel-Morgenstern A (2015) Continuous preferential crystallization of chiral molecules in single and coupled mixed-suspension mixed-product-removal crystallizers. Cryst Growth Des 15(4):1808–1818
Gubaidullin AT, Samigullina AI, Bredikhina ZA, Bredikhin AA (2014) Crystal structure of chiral ortho-alkyl phenyl ethers of glycerol: true racemic compound, normal, false and anomalous conglomerates within the single five-membered family. CrystEngComm 16:6716–6729
Isakov AI, Kotelnikova EN, Kryuchkova LY, Lorenz H (2013a) Effect of crystallization conditions on polymorphic diversity of malic acid RS-racemate. Trans Tianjin Univ 19(2):86–91
Isakov AI, Kotelnikova EN, Kryuchkova LY, Lorenz H (2013b) Isomorphism and polymorphism in the system of malic acid enantiomers on the basis of X-ray and high-temperature X-ray diffractometry. In: Qu H, Rantanen J, Malwade C (eds) Proceedings of the 20th international workshop on industrial crystallization (BIWIC-2013), vol 20. Odense, Denmark, pp 395–402
Isakov AI, Kotelnikova EN, Lorenz H (2015) Non-equimolar discrete phases formed in the system of malic acid enantiomers. Chem Eng Technol 38(6):1047–1052
Isakov AI, Kotelnikova EN, Bocharov SN, Zolotarev AA Jr, Lorenz H (2016a) Thermal deformations of the crystal structures of L-valine, L-isoleucine and discrete compound V2I. In: Lorenz H, Buchholz H (eds) Proceedings of the 23rd international workshop on industrial crystallization (BIWIC-2016), vol 23. Cuvillier Verlag, Göttingen, pp 7–12
Isakov AI, Kotelnikova EN, Muenzberg S, Bocharov SN, Lorenz H (2016b) Solid phases in the system L-valine–L-isoleucine. Cryst Growth Des 16:2653–2661
Isakov AI, Kotelnikova EN, Lorenz H (2018) Limits of solid solutions and crystal morphology in the system of L-valine–L-leucine enantiomers. In: Proceedings of the VI international symposium on “Biogenic – abiogenic interactions in natural and anthropogenic systems”. St. Petersburg, Russia, pp 121–123
Jacques J, Collet A, Wilen SH (1981) Enantiomers, racemates and resolutions, 1rd edn. Wiley, New York
Janczak J, Zobel D, Luger P (1997) L-threonine at 12 K. Acta Cryst C53:1901–1904
Kaemmerer H (2012) New concepts for enantioselective crystallization. Shaker Verlag, Aachen
Kotelnikova EN, Isakov AI, Lorenz H (2017) Non-equimolar discrete compounds in binary chiral systems of organic substances (highlight). CrystEngComm 19(14):1851–1869
Kotelnikova EN, Isakov AI, Lorenz H (2018) Thermal deformations of crystal structures formed in the systems of malic acid enantiomers and L-valine–L-isoleucine enantiomers. CrystEngComm 20(18):2562–2572
Koolman HC, Rousseau RW (1996) Effects of isomorphic compounds on the purity and morphology of L-isoleucine crystals. AIChE J 42(1):147–153
Kryuchkova LY, Kotelnikova EN, Lorenz H, Zolotarev AA Jr, Bocharov SN (2018a) Solid phases in the chiral phenylglycine system according to PXRD and SCXRD data. In: Cartigny Y, Couvrat N (eds) Proceedings of the 25th international workshop on industrial crystallization (BIWIC-2018), vol 25. Rouen, France, pp 226–231
Kryuchkova LY, Kotelnikova EN, Zolotarev AA Jr, Lorenz H (2018b) Limits of solid solutions in the L-leucine—isoleucine system according to PXRD and SCXRD data. In: Cartigny Y, Couvrat N (eds) Proceedings of the 25th international workshop on industrial crystallization (BIWIC-2018), vol 25. Rouen, France, p 225
Kurosawa I, Teja AS, Rousseau RW (2005) Solubility measurements in the l-Isoleucine + l-Valine + Water System at 298 K. Eng Chem Res 44:3284–3288
Liu Z-S, Xu Y-L, Wang H, Yan C, Gao R-Y (2004) Chiral separation of binaphthol enantiomers on molecularly imprinted polymer monolith by capillary electrochromatography. Anal Sci 20:673–678
Lin G-Q, Zhang J-G, Cheng J-F (2011) Overview of chirality and chiral drugs. In: Lin G-Q, You Q-D, Cheng J-F (eds) Chiral drugs: chemistry and biological action. Wiley, New York, Hoboken (NJ)
Lorenz H, Seidel-Morgenstern A (2014) Processes to separate enantiomers. Angew Chem Int Ed 53:1218–1250
Murakami H (2007) From racemates to single enantiomers–chiral synthetic drugs over the last 20 years. Top Curr Chem 269:273–299
Nogradi M (1981) Stereochemistry. Basic concepts and applications. Pergamon press, Oxford
Oches EA, McCoy WD (2001) Historical developments and recent advances in amino acid geochronology applied to loess research: examples from North America, Europe, and China. Earth Sci Rev 54(1–3):173–192
Robins J, Jones M, Matisoo-Smith E (2001) Amino acid racemization dating in New Zealand: an overview and bibliography. Auckland University, Auckland
Sapoundjiev D, Lorenz H, Seidel-Morgenstern A (2006) Solubility of chiral threonine species in water/ethanol mixtures. J Chem Eng Data 51(5):1526–1566
Shoemaker DP, Donohue J, Schomaker V, Corey RB (1950) The crystal structure of L threonine. J Am Chem Soc 72:2328–2349
Swaminathan P, Srinivasan R (1975) Studies in molecular structure, symmetry and conformation. VIII. Crystal and molecular structure of L-allothreonine. Acta Cryst B31:217–221
Taratin NV, Lorenz H, Kotelnikova EN, Glikin AE, Galland A, Dupray V, Coquerel G, Seidel-Morgenstern A (2012) Mixed crystals in chiral organic systems: a case study on (R)- and (S)-ethanolammonium 3-chloromandelate. Cryst Growth Des 12:5882–5888
Taratin NV, Binev D, Lorenz H, Seidel-Morgenstern A, Kotelnikova EN (2013) Characterization and limits of solid solutions in binary systems of threonine diastereomers according to X-ray diffraction data. In: Bekker TB, Litasov KD, Sobolev NV (eds) Proceedings of the III international conference on crystallogenesis and mineralogy. Novosibirsk, Russia, pp 341-I–341-II
Taratin N, Lorenz H, Binev D, Seidel-Morgenstern A, Kotelnikova E (2015) Solubility equilibria and crystallographic characterization of the L-threonine/L-allo-threonine system. Part 2: crystallographic characterization of solid solutions in the threonine diastereomeric system. Cryst Growth Des 15:137–144
Torii K, Iitaka Y (1970) The crystal structure of L-valine. Acta Cryst B26:1317–1326
Torii K, Iitaka Y (1971) The crystal structure of L-isoleucine. Acta Cryst B27:2237–2246
Xie R, Chu L-Y, Deng J-G (2008) Membranes and membrane processes for chiral resolution. Chem Soc Rev 37:1243–1263
Acknowledgements
The investigations were performed using equipment of the Resource Centre for X-ray Diffraction Studies of Saint Petersburg State University and equipment of the MPI Magdeburg PCF lab. The authors thank Dr. N. Taratin, Dr. D. Binev, Dr. S. Muenzberg, Dr. M. G. Krzhizhanovskaya, Prof. A. Seidel-Morgenstern and Prof. G. Coquerel for their contributions over the years. The authors appreciate the financial support provided by the Russian Foundation for Basic Research (Projects: 18-35-00183 mol_a and 16-29-11-727 ofi_m).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this paper
Cite this paper
Kotelnikova, E.N., Isakov, A.I., Kryuchkova, L.Y., Zolotarev, A.A., Bocharov, S.N., Lorenz, H. (2020). Acids with Chiral Molecules as Essential Organic Compounds of Biogenic–Abiogenic Systems. In: Frank-Kamenetskaya, O., Vlasov, D., Panova, E., Lessovaia, S. (eds) Processes and Phenomena on the Boundary Between Biogenic and Abiogenic Nature. Lecture Notes in Earth System Sciences. Springer, Cham. https://doi.org/10.1007/978-3-030-21614-6_37
Download citation
DOI: https://doi.org/10.1007/978-3-030-21614-6_37
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-21613-9
Online ISBN: 978-3-030-21614-6
eBook Packages: Earth and Environmental ScienceEarth and Environmental Science (R0)