Abstract
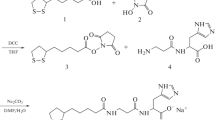
We designed isoprenomics-based prenylated biosynthetic intermediates of cohumulone and colupulone as Low-density lipoprotein (LDL)-targeting antioxidants. Syntheses of acylphloroglucinol (UTX-52), mono-prenylated acylphloroglucinol (UTX-53), and di-prenylated acylphloroglucinol (UTX-54) were performed according to our established aromatic prenylation method. The free radical-scavenging activity of UTX-53 was highest among all the compounds tested and its potency was about twice as International Calibration Extract as a hop extract. The inhibitory activity of UTX-53 on linoleic acid oxidation showed about four times higher than ICE-2. The inhibitory activity of UTX-52, UTX-53, and UTX-54 on human LDL oxidation showed almost the same inhibitory activity, and these compounds were weaker than that of ICE-2. According to these results, we suggest that the antioxidative activity of prenylated acylphloroglucinols strongly depends on the radical scavenging activity rather than the hydrophobicity of, e.g., artepillin C analogues.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Steinberg D (1995) Role of oxidized LDL and antioxidant in atherosclerosis. Adv Exp Med Biol 369:39–48
Witztum JL, Steinberg D (1991) Role of oxidized low density lipoprotein in atherogenesis. J Clin Invest 88:1785–1792
Larson AE, Yu RR, Lee OA et al (1996) Antimicrobial activity of hop extracts against Listeria monocytogenes in media and in food. Int J Food Microbiol 33:195–207
Segawa S, Takata Y, Kaneda H et al (2007) Effects of a hop water extract on the compound 48/80-stimulated vascular permeability in ICR mice and histamine release from OVA-sensitized BALB/c mice. Biosci Biotechnol Biochem 71:1577–1581
Hougee S, Faber J, Sanders A et al (2006) Selective inhibition of COX-2 by a standardized CO2 extract of Humulus lupulus in vitro and its activity in a mouse model of zymosan-induced arthritis. Planta Med 72:228–233
Tagashira M, Watanabe M, Uemitsu N (1995) Antioxidative activity of hop bitter acids and their analogues. Biosci Biotechnol Biochem 59:740–742
Uto Y, Ae S, Koyama D et al (2006) Artepillin C isoprenomics: design and synthesis of artepillin C isoprene analogues as lipid peroxidation inhibitor having low mitochondrial toxicity. Bioorg Med Chem 14:5721–5728
Uto Y, Ae S, Hotta A et al (2006) Artepillin C isoprenomics: design and synthesis of artepillin C analogues as antiatherogenic antioxidants. Adv Exp Med Biol 578:113–118
Zuurbier KWM, Fung SY, Scheffer JJC et al (1998) In-vitro prenylation of aromatic intermediates in the biosynthesis of bitter acids in Humulus lupulus. Phytochemistry 49:2315–2322
Blois MS (1958) Antioxidant determinations by the use of a stable free radical. Nature 181:1199
Liégeois C, Lermusieau G, Collin S (2000) Measuring antioxidant efficiency of wort, malt, and hops against the 2,2′-azobis(2-amidinopropane) dihydrochloride-induced oxidation of an aqueous dispersion of linoleic acid. J Agric Food Chem 48:1129–1134
Yagi K (1998) Simple assay for the level of total lipid peroxides in serum or plasma. Methods Mol Biol 108:101–106
Acknowledgments
We thank Ms. Maki Nakamura, Ms. Emiko Okayama and the staff of our faculty for measurement of NMR and elemental analyses.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2012 Springer Science+Business Media, LLC
About this paper
Cite this paper
Tanaka, R. et al. (2012). Prenylated Acylphloroglucinol Derivatives: Isoprenomics-Based Design, Syntheses and Antioxidative Activities. In: Wolf, M., et al. Oxygen Transport to Tissue XXXIII. Advances in Experimental Medicine and Biology, vol 737. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-1566-4_37
Download citation
DOI: https://doi.org/10.1007/978-1-4614-1566-4_37
Published:
Publisher Name: Springer, New York, NY
Print ISBN: 978-1-4614-1565-7
Online ISBN: 978-1-4614-1566-4
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)