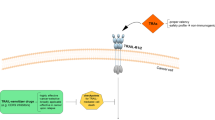
Abstract
The development of apoptosis resistance is a crucial step during the pathogenesis of malignant tumors. Thus, any treatment approach overcoming apoptosis resistance may be a valuable tool in oncology. Although a variety of treatments induce apoptosis, only very few specifically trigger programmed cell death. In this regard, the class of apoptosis inducing ligands may turn out to have a considerable potential in oncology. TNF-α-related apoptosis-inducing ligand (TRAIL/Apo2L) is the most promising candidate, either alone or in combination with established cancer therapies, since it induces apoptosis in a wide range of malignant cells while sparing most normal tissues.
Since death-receptor induced apoptosis is mainly mediated via nonmitochondrial death pathways, it is possible to induce apoptosis in cancer cell systems which mainly harbor defects within the mitochondrial death cascades.
Even more so it has been shown that conventional DNA damaging approaches reduced the killing threshold for receptor induced apoptosis, making TRAIL an ideal candidate for combined approaches. Thus, combined treatments might offer the chance to enhance therapeutic efficiency and overcome resistance. In combination, additive or synergistic apoptotic responses and substantially enhanced clonogenic cell kill has been documented. Furthermore, in several settings it has been shown that combined modality teatments were effective in malignant cells, which are highly resistant to either treatment, alone. Ionizing radiation is one of the most effective modalities in oncology. Thus, it is reasonable to test, how far combinations of TRAIL with ionizing radiation may increase the efficacy. Indeed, the combination of TRAIL with ionizing radiation in several in vitro settings as well as xenograft models resulted in highly increased rates of cell kill and long-term tumor control. No increase in the rate and severity of side effects has been documented, indicating that the combination really increases the therapeutic ratio. It is important to note that TRAIL and TRAIL receptor agonistic antibodies, either as single agent or in combination with cytotoxic drugs, were safe in human phase I trials. Ongoing phase II trials will hopefully document the clinical efficacy of this treatment approach.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Aldridge DR, Arends MJ, Radford R (1995) Increasing the susceptibility of the rat 208f fibroblast cell-line to radiation-induced apoptosis does not alter its clonogenic survival dose – response. Br J Cancer 71:571–577
Ashkenazi A (2008) Directing cancer cells to self-destruct with pro-apoptotic receptor agonists. Nat Rev Drug Discov 7:1001–1012
Ashkenazi A, Pai RC, Fong S, Leung S, Lawrence DA, Masters SA, Blackie C, Chang L, McMurtrey AE, Hebert A et al (1999) Safety and antitumor activity of recombinant soluble Apo2 ligand. J Clin Invest 104:155–162
Belka C, Budach W (2002) Anti-apoptotic Bcl-2 proteins: structure, function and relevance for radiation biology. Int J Radiat Biol 78:643–658
Belka C, Rudner J, Wesselborg S, Stepczynska A, Marini P, Lepple-Wienhues A, Faltin H, Bamberg M, Budach W, Schulze-Osthoff K (2000) Differential role of caspase-8 and BID activation during radiation- and CD95-induced apoptosis. Oncogene 19:1181–1190
Belka C, Schmid B, Marini P, Durand E, Rudner J, Faltin H, Bamberg M, Schulze-Osthoff K, Budach W (2001) Sensitization of resistant lymphoma cells to irradiation-induced apoptosis by the death ligand TRAIL. Oncogene 20:2190–2196
Buchsbaum DJ, Zhou T, Grizzle WE, Oliver PG, Hammond CJ, Zhang S, Carpenter M, LoBuglio AF (2003) Antitumor efficacy of TRA-8 anti-DR5 monoclonal antibody alone or in combination with chemotherapy and/or radiation therapy in a human breast cancer model. Clin Cancer Res 9:3731–3741
Chen XF, Kandasamy K, Srivastava RK (2003) Differential roles of RelA (p65) and c-Rel subunits of nuclear factor kappa B in tumor necrosis factor-related apoptosis-inducing ligand signaling. Cancer Res 63:1059–1066
Chinnaiyan AM, Prasad U, Shankar S, Hamstra DA, Shanaiah M, Chenevert TL, Ross BD, Rehemtulla A (2000) Combined effect of tumor necrosis factor-related apoptosis-inducing ligand and ionizing radiation in breast cancer therapy. Proc Natl Acad Sci U S A 97:1754–1759
Chow LQ, Eckhardt SG, Gustafson DL, O’Bryant C, Hariharan S, Diab S, Fox NL, Corey A, Padavic K, Brown M et al (2006) HGS-ETR1, an antibody targeting TRAIL-R1, in combination with paclitaxel and carboplatin in patients with advanced solid malignancies: results of a phase I and PK study. J Clin Oncol 24:103S
Chuntharapai A, Dodge K, Grimmer K, Schroeder K, Marsters SA, Koeppen H, Ashkenazi A, Kim KJ (2001) Isotype-dependent inhibition of tumor growth in vivo by monoclonal antibodies to death receptor 4. J Immunol 166:4891–4898
Danial NN, Korsmeyer SJ (2004) Cell death: critical control points. Cell 116:205–219
Debatin KM, Poncet D, Kroemer G (2002) Chemotherapy: targeting the mitochondrial cell death pathway. Oncogene 21:8786–8803
DegliEsposti MA, Smolak PJ, Walczak H, Waugh J, Huang CP, DuBose RF, Goodwin RG, Smith CA (1997) Cloning and characterization of TRAIL-R3, a novel member of the emerging TRAIL receptor family. J Exp Med 186:1165–1170
Di Pietro R, Secchiero P, Rana R, Gibellini D, Visani G, Bemis K, Zamai L, Miscia S, Zauli G (2001) Ionizing radiation sensitizes erythroleukemic cells but not normal erythroblasts to tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-mediated cytotoxicity by selective up-regulation of TRAIL-R1. Blood 97:2596–2603
Eskes R, Desagher S, Antonsson B, Martinou JC (2000) Bid induces the oligomerization and insertion of Bax into the outer mitochondrial membrane. Mol Cell Biol 20:929–935
Falschlehner C, Emmerich CH, Gerlach B, Walczak H (2007) TRAIL signalling: decisions between life and death. Int J Biochem Cell Biol 39:1462–1475
Finnberg N, Gruber JJ, Fei PW, Rudolph D, Bric A, Kim SH, Burns TF, Ajuha H, Page R, Wu GS et al (2005) DR5 knockout mice are compromised in radiation-induced apoptosis. Mol Cell Biol 25:2000–2013
Finnberg N, Klein-Szanto AJP, El-Deiry WS (2008) TRAU-R deficiency in mice promotes susceptibility to chronic inflammation and tumorigenesis. J Clin Invest 118:111–123
Fiveash JB, Gillespie GY, Oliver PG, Zhou T, Belenky ML, Buchsbaum DJ (2008) Enhancement of glioma radiotherapy and chemotherapy response with targeted antibody therapy against death receptor 5. Int J Radiat Oncol Biol Phys 71:507-516
Fuchs EJ, Paik JE, Engstrom LE, Bedi A (1997) CD95 (Fas/APO-1)-dependence of T cell apoptosis induced by DNA damage. Blood 90: 225
Ganten TM, Haas TL, Sykora J, Stahl H, Sprick MR, Fas SC, Krueger A, Weigand MA, Grosse-Wilde A, Stremmel W et al (2004) Enhanced caspase-8 recruitment to and activation at the DISC is critical for sensitisation of human hepatocellular carcinoma cells to TRAIL-induced apoptosis by chemotherapeutic drugs. Cell Death Differ 11:S86–S96
Georgakis GV, Li Y, Humphreys R, Andreeff M, O’Brien S, Younes M, Carbone A, Albert V, Younes A (2005) Activity of selective fully human agonistic antibodies to the TRAIL death receptors TRAIL-R1 and TRAIL-R2 in primary and cultured lymphoma cells: induction of apoptosis and enhancement of doxorubicin- and bortezomib-induced cell death. Br J Haematol 130:501–510
Goh HS, Yao J, Smith DR (1995) P53 point mutation and survival in colorectal-cancer patients. Cancer Res 55:5217–5221
Gong BD, Almasan A (2000) Apo2 ligand/TNF-related apoptosis-inducing ligand and death receptor 5 mediate the apoptotic signaling induced by ionizing radiation in leukemic cells. Cancer Res 60:5754–5760
Grassme H, Kirschnek S, Riethmueller J, Riehle A, von Kurthy G, Lang F, Weller M, Gulbins E (2000) CD95/CD95 ligand interactions on epithelial cells in host defense to Pseudomonas aeruginosa. Science 290:527–530
Griffith TS, Chin WA, Jackson GC, Lynch DH, Kubin MZ (1998) Intracellular regulation of TRAIL-induced apoptosis in human melanoma cells. J Immunol 161:2833–2840
Grosse-Wilde A, Voloshanenko O, Bailey SL, Longton GM, Schaefer U, Csernok AI, Schutz G, Greiner EF, Kemp CJ, Walczak H (2008) TRAIL-R deficiency in mice enhances lymph node metastasis without affecting primary tumor development. J Clin Invest 118:100–110
Guan BX, Yue P, Clayman GL, Sun SY (2001) Evidence that the death receptor DR4 is a DNA damage-inducible, p53-regulated gene. J Cell Physiol 188:98–105
Hamasu T, Inanami O, Asanuma T, Kuwabara M (2005) Enhanced induction of apoptosis by combined treatment of human carcinoma cells with X rays and death receptor agonists. J Radiat Res 46:103–110
Heinzelmann F, Jendrossek V, Lauber K, Nowak K, Eldh T, Boras R, Handrick R, Henkel M, Martin C, Uhlig S et al (2006) Irradiation-induced pneumonitis mediated by the CD95/CD95-ligand system. J Natl Cancer Inst 98:1248–1251
Herbst RS, Mendolson DS, Ebbinghaus S, Gordon MS, O’Dwyer P, Lieberman G, Ing J, Kurzrock R, Novotny W, Eckhardt G (2006) A phase I safety and pharmacokinetic (PK) study of recombinant Apo2L/TRAIL, an apoptosis-inducing protein in patients with advanced cancer. J Clin Oncol 24:124S
Herr I, Debatin KM (2001) Cellular stress response and apoptosis in cancer therapy. Blood 98:2603–2614
Ichikawa K, Liu WM, Zhao LM, Wang Z, Liu D, Ohtsuka T, Zhang HG, Mountz JD, Koopman WJ, Kimberly RP et al (2001) Tumoricidal activity of a novel anti-human DR5 monoclonal antibody without hepatocyte cytotoxicity. Nat Med 7:954–960
Ivanov N, Zhou HN, Hei TK (2007) Sequential treatment by ionizing radiation and sodium arsenite dramatically accelerates TRAIL-mediated apoptosis of human melanoma cells. Cancer Res 67:5397–5407
Johnstone RW, Ruefli AA, Lowe SW (2002) Apoptosis: a link between cancer genetics and chemotherapy. Cell 108:153–164
Kanzler S, Trarbach T, Heinemann V, Kohne CH, Sneller V, Bieber F, Galle PR, Seeber S (2005) Results of a phase 2 study of HGS-ETR1, a fully human agonistic monoclonal antibody to TRAIL Receptor 1, in subjects with relapsed or refractory colorectal cancer (CRC). EJC Suppl 3:178–178
Kim MR, Lee JY, Park MT, Chun YJ, Jang YJ, Kang CM, Kim HS, Cho CK, Lee YS, Jeong HY et al (2001) Ionizing radiation can overcome resistance to TRAIL in TRAIL-resistant cancer cells. FEBS Lett 505:179–184
Kolesnick R, Fuks Z (2003) Radiation and ceramide-induced apoptosis. Oncogene 22:5897–5906
Lacour S, Hammann A, Wotawa A, Corcos L, Solary E, Dimanche-Boitrel MT (2001) Anticancer agents sensitize tumor cells to tumor necrosis factor-related apoptosis-inducing ligand-mediated caspase-8 activation and apoptosis. Cancer Res 61:1645–1651
Lacour S, Micheau O, Hammann A, Drouineaud V, Tschopp J, Solary E, Dimanche-Boitrel MT (2003) Chemotherapy enhances TNF-related apoptosis-inducing ligand DISC assembly in HT29 human colon cancer cells. Oncogene 22:1807–1816
Li HL, Zhu H, Xu CJ, Yuan JY (1998) Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94:491–501
Liu XS, Kim CN, Yang J, Jemmerson R, Wang XD (1996) Induction of apoptotic program in cell-free extracts: requirement for dATP and cytochrome c. Cell 86:147–157
Lowe SW, Ruley HE, Jacks T, Housman DE (1993) P53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74:957–967
Lowe SW, Bodis S, McClatchey A, Remington L, Ruley HE, Fisher DE, Housman DE, Jacks T (1994) P53 status and the efficacy of cancer-therapy in-vivo. Science 266:807–810
Luo X, Budihardjo I, Zou H, Slaughter C, Wang XD (1998) Bid, a Bcl2 interacting protein, mediates cytochrome c release from mitochondria in response to activation of cell surface death receptors. Cell 94:481–490
Maduro JH, de Vries EGE, Meersma GJ, Hougardy BMT, van der Zee AGJ, De Jong S (2008) Targeting pro-apoptotic trail receptors sensitizes HeLa cervical cancer cells to irradiation-induced apoptosis. Int J Radiat Oncol Biol Phys 72:543–552
Marini P, Jendrossek V, Durand E, Gruber C, Budach W, Belka C (2003) Molecular requirements for the combined effects of TRAIL and ionising radiation. Radiother Oncol 68:189–198
Marini P, Schmid A, Jendrossek V, Faltin H, Daniel PT, Budach W, Belka C (2005) Irradiation specifically sensitises solid tumour cell lines to TRAIL mediated apoptosis. BMC Cancer 5:5
Marini P, Denzinger S, Schiller D, Kauder S, Welz S, Humphreys R, Daniel PT, Jendrossek V, Budach W, Belka C (2006) Combined treatment of colorectal tumours with agonistic TRAIL receptor antibodies HGS-ETR1 and HGS-ETR2 and radiotherapy: enhanced effects in vitro and dose-dependent growth delay in vivo. Oncogene 25:5145–5154
Marsters SA, Sheridan JP, Pitti RM, Huang A, Skubatch M, Baldwin D, Yuan J, Gurney A, Goddard AD, Godowski P et al (1997) A novel receptor for Apo2L/TRAIL contains a truncated death domain. Current Biology 7:1003–1006
Merino D, Lalaoui N, Morizot A, Schneider P, Solary E, Micheau O (2006) Differential inhibition of TRAIL-mediated DR5-DISC formation by decoy receptors 1 and 2. Mol Cell Biol 26:7046–7055
Miyashita T, Reed JC (1995) Tumor-suppressor p53 is a direct transcriptional activator of the human bax gene. Cell 80:293–299
Mom CH, Sleijfer S, Gietema JA, Fox NL, Piganeau C, Lo L, Uges DRA, Loos W, de Vries EGE, Verweij J (2006) Mapatumumab, a fully human agonistic monoclonal antibody that targets TRAIL-R1, in combination with gemcitabine and cisplatin: a phase 1 study in patients with advanced solid malignancies. EJC Suppl 4:202
Nagane M, Cavenee WK, Shiokawa Y (2007) Synergistic cytotoxicity through the activation of multiple apoptosis pathways in human glioma cells induced by combined treatment with ionizing radiation and tumor necrosis factor-related apoptosis-inducing ligand. J Neurosurg 106:407–416
Nakano K, Vousden KH (2001) PUMA, a novel proapoptotic gene, is induced by p53. Mol Cell 7:683–694
Ni R, Tomita Y, Matsuda K, Ichihara A, Ishimura K, Ogasawara J, Nagata S (1994) FAS-mediated apoptosis in primary cultured mouse hepatocytes. Exp Cell Res 215:332–337
Norbury CJ, Zhivotovsky B (2004) DNA damage-induced apoptosis. Oncogene 23:2797–2808
Oda E, Ohki R, Murasawa H, Nemoto J, Shibue T, Yamashita T, Tokino T, Taniguchi T, Tanaka N (2000) Nora, a BH3-only member of the Bcl-2 family and candidate mediator of p53-induced apoptosis. Science 288:1053–1058
Ogasawara J, Watanabefukunaga R, Adachi M, Matsuzawa A, Kasugai T, Kitamura Y, Itoh N, Suda T, Nagata S (1993) Lethal effect of the anti-Fas antibody in mice. Nature 364:806–809
Pacey S, Plummer RE, Attard G, Bale C, Calvert AH, Blagden S, Fox NL, Corey A, de Bono JS (2005) Phase I and pharmacokinetic study of HGS-ETR2, a human monoclonal antibody to TRAIL R2, in patients with advanced solid malignancies. J Clin Oncol 23: 205S
Pan GH, Ni J, Wei YF, Yu GL, Gentz R, Dixit M (1997a) An antagonist decoy receptor and a death domain-containing receptor for TRAIL. Science 277:815–818
Pan GH, Orourke K, Chinnaiyan AM, Gentz R, Ebner R, Ni J, Dixit M (1997b) The receptor for the cytotoxic ligand TRAIL. Science 276:111–113
Pukac L, Kanakaraj P, Humphreys R, Alderson R, Bloom M, Sung C, Riccobene T, Johnson R, Fiscella M, Mahoney A et al (2005) HGS-ETR1, a fully human TRAIL-receptor 1 monoclonal antibody, induces cell death in multiple tumour types in vitro and in vivo. Br J Cancer 92:1430–1441
Ramp U, Caliskan E, Mahotka C, Krieg A, Heikaus S, Gabbert HE, Gerharz CD (2003) Apoptosis induction in renal cell carcinoma by TRAIL and gamma-radiation is impaired by deficient caspase-9 cleavage. Br J Cancer 88:1800–1807
Rezacova M, Vavrova J, Vokurkova D (2008) Ionizing radiation sensitizes leukemic MOLT-4 cells to TRAIL-induced apoptosis. Acta Medica (Hradec Kralove) 51:101–105
Rudner J, Jendrossek V, Lauber K, Daniel PT, Wesselborg S, Belka C (2005) Type I and type II reactions in TRAIL-induced apoptosis – results from dose-response studies. Oncogene 24:130–140
Scaffidi C, Fulda S, Srinivasan A, Friesen C, Li F, Tomaselli KJ, Debatin KM, Krammer PH, Peter ME (1998) Two CD95 (APO-1/Fas) signaling pathways. EMBO J 17:1675–1687
Schneider P, Bodmer JL, Thome M, Hofmann K, Holler N, Tschopp J (1997) Characterization of two receptors for TRAIL. FEBS Lett 416:329–334
Shankar S, Singh TR, Chen XF, Thakkar H, Firnin J, Srivastava RK (2004a) The sequential treatment with ionizing radiation followed by TRAIL/Apo-2L reduces tumor growth and induces apoptosis of breast tumor xenografts in nude mice. Int J Oncol 24:1133–1140
Shankar S, Singh TR, Srivastava RK (2004b) Ionizing radiation enhances the therapeutic potential of TRAIL in prostate cancer in vitro and in vivo: intracellular mechanisms. Prostate 61:35–49
Shankar S, Chen XF, Srivastava RK (2005) Effects of sequential treatments with chemotherapeutic drugs followed by TRAIL on prostate cancer in vitro and in vivo. Prostate 62:165–186
Sheridan JP, Marsters SA, Pitti RM, Gurney A, Skubatch M, Baldwin D, Ramakrishnan L, Gray CL, Baker K, Wood WI et al (1997) Control of TRAIL-induced apoptosis by a family of signaling and decoy receptors. Science 277:818–821
Singh TR, Shankar S, Chen WF, Asim M, Srivastava RK (2003) Synergistic interactions of chemotherapeutic drugs and tumor necrosis factor-related apoptosis-inducing ligand/Apo-2 ligand on apoptosis and on regression of breast carcinoma in vivo. Cancer Res 63:5390–5400
Sprick MR, Rieser E, Stahl H, Grosse-Wilde A, Weigand MA, Walczak H (2002) Caspase-10 is recruited to and activated at the native TRAIL and CD95 death-inducing signalling complexes in a FADD-dependent manner but can not functionally substitute caspase-8. EMBO J 21:4520–4530
Suliman A, Lam A, Datta R, Srivastava RK (2001) Intracellular mechanisms of TRAIL: apoptosis through mitochondrial-dependent and -independent pathways. Oncogene 20:2122–2133
Takahashi M, Yasui H, Ogura A, Asanuma T, Kubota N, Tsujitan M, Kuwabara M, Inanami O (2008) X irradiation combined with TNF alpha-related apoptosis-inducing ligand (TRAIL) reduces hypoxic regions of human gastric adenocarcinoma xenografts in SCID mice. J Radiat Res 49:153–161
Tolcher AW, Mita M, Meropol NJ, von Mehren M, Patnaik A, Padavic K, Hill M, Mays T, McCoy T, Fox NL et al (2007) Phase I pharmacokinetic and biologic correlative study of mapatumumab, a fully human monoclonal antibody with agonist activity to tumor necrosis factor-related apoptosis-inducing ligand receptor-1. J Clin Oncol 25:1390–1395
Verbrugge I, de Vries E, Tait SWG, Wissink EHJ, Walczak H, Verheij M, Borst J (2008) Ionizing radiation modulates the TRAIL death-inducing signaling complex, allowing bypass of the mitochondrial apoptosis pathway. Oncogene 27:574–584
Vogelstein B, Kinzler KW (2004) Cancer genes and the pathways they control. Nat Med 10:789–799
von Haefen C, Gillissen B, Hemmati PG, Wendt J, Guner D, Mrozek A, Belka C, Dorken B, Daniel PT (2004) Multidomain Bcl-2 homolog Bax but not Bak mediates synergistic induction of apoptosis by TRAIL and 5-FU through the mitochondrial apoptosis pathway. Oncogene 23:8320–8332
Wagner KW, Punnoose EA, Januario T, Lawrence DA, Pitti RM, Lancaster K, Lee D, von Goetz M, Yee SF, Totpal K et al (2007) Death-receptor O-glycosylation controls tumor-cell sensitivity to the proapoptotic ligand Apo2L/TRAIL. Nat Med 13:1070–1077
Walczak H, Miller RE, Ariail K, Gliniak B, Griffith TS, Kubin M, Chin W, Jones J, Woodward A, Le T et al (1999) Tumoricidal activity of tumor necrosis factor related apoptosis-inducing ligand in vivo. Nat Med 5:157–163
Wang SL, El-Deiry WS (2004) Inducible silencing of KILLER/DR5 in vivo promotes bioluminescent colon tumor xenograft growth and confers resistance to chemotherapeutic agent 5-fluorouracil. Cancer Res 64:6666–6672
Wang K, Yin XM, Chao DT, Milliman CL, Korsmeyer SJ (1996) BID: a novel BH3 domain-only death agonist. Genes Dev 10:2859–2869
Williams BA, Makrigiannis AP, Blay J, Hoskin DW (1997) Treatment of the P815 murine mastocytoma with cisplatin or etoposide up-regulates cell-surface Fas (CD95) expression and increases sensitivity to anti-Fas antibody-mediated cytotoxicity and to lysis by anti-CD3-activated killer-T cells. Int J Cancer 73:416–423
Wissink EHJ, Verbrugge I, Vink SR, Schader MB, Schaefer U, Walczak H, Borst J, Verheij M (2006) TRAIL enhances efficacy of radiotherapy in a p53 mutant, Bcl-2 overexpressing lymphoid malignancy. Radiother Oncol 80:214–222
Wu GS, Burns TF, McDonald ER, Jiang W, Meng R, Krantz D, Kao G, Gan DD, Zhou JY, Muschel R et al (1997) KILLER/DR5 is a DNA damage-inducible p53-regulated death receptor gene. Nat Genet 17:141–143
Yang J, Liu XS, Bhalla K, Kim CN, Ibrado AM, Cai JY, Peng TI, Jones DP, Wang XD (1997) Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 275:1129–1132
Yin XM, Wang K, Gross A, Zhao YG, Zinkel S, Klocke B, Roth KA, Korsmeyer SJ (1999) Bid-deficient mice are resistant to Fas-induced hepatocellular apoptosis. Nature 400:886–891
Yoshida T, Maeda A, Tani N, Sakai T (2001) Promoter structure and transcription initiation sites of the human death receptor 5/TRAIL-R2 gene. FEBS Lett 507:381–385
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2009 Springer-Verlag Berlin Heidelberg
About this chapter
Cite this chapter
Niemöller, O., Belka, C. (2009). Targeting Death-Receptors in Radiation Therapy. In: Kalthoff, H. (eds) Death Receptors and Cognate Ligands in Cancer. Results and Problems in Cell Differentiation, vol 49. Springer, Berlin, Heidelberg. https://doi.org/10.1007/400_2009_17
Download citation
DOI: https://doi.org/10.1007/400_2009_17
Published:
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-03044-4
Online ISBN: 978-3-642-03045-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)