Abstract
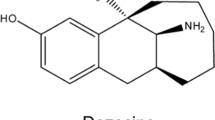
Cebranopadol is a novel first-in-class analgesic with highly potent agonistic activity at nociceptin/orphanin FQ peptide (NOP) and opioid receptors. It is highly potent and efficacious across a broad range of preclinical pain models. Its side effect profile is better compared to typical opioids. Mechanistic studies have shown that cebranopadol’s activity at NOP receptors contributes to its anti-hyperalgesic effects while ameliorating some of its opioid-type side effects, including respiratory depression and abuse potential. Phase II of clinical development has been completed, demonstrating efficacy and good tolerability in acute and chronic pain conditions.
This article focusses on reviewing data on the preclinical in vitro and in vivo pharmacology, safety, and tolerability, as well as clinical trials with cebranopadol.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Asth L, Ruzza C, Malfacini D et al (2016) Beta-arrestin 2 rather than G protein efficacy determines the anxiolytic-versus antidepressant-like effects of nociceptin/orphanin FQ receptor ligands. Neuropharmacology 105:434–442
Benredjem B, Dallaire P, Pineyro G (2017) Analyzing biased responses of GPCR ligands. Curr Opin Pharmacol 32:71–76
Bird MF, Lambert DG (2015) Simultaneous targeting of multiple opioid receptor types. Curr Opin Support Palliat Care 9:98–102
Bohn LM, Lefkowitz RJ, Gainetdinov RR et al (1999) Enhanced morphine analgesia in mice lacking beta-arrestin 2. Science 286:2495–2498
Bohn LM, Gainetdinov RR, Lin FT et al (2000) Mu-opioid receptor desensitization by beta-arrestin-2 determines morphine tolerance but not dependence. Nature 408:720–723
Bologna Z, Teoh JP, Bayoumi AS et al (2017) Biased G protein-coupled receptor signaling: new player in modulating physiology and pathology. Biomol Ther 25:12–25
Calo’ G, Lambert DG (2018) Nociceptin/orphanin FQ receptor ligands and translational challenges: focus on cebranopadol as an innovative analgesic. Br J Anaesth 121:1105–1114
Camarda V, Fischetti C, Anzellotti N et al (2009) Pharmacological profile of NOP receptors coupled with calcium signaling via the chimeric protein G alpha qi5. Naunyn Schmiedeberg’s Arch Pharmacol 379:599–607
Chang SD, Brieaddy LE, Harvey JD et al (2015a) Novel synthesis and pharmacological characterization of NOP receptor agonist 8-[(1S,3aS)-2,3,3a,4,5,6-hexahydro-1H-phenalen-1-yl]-1-phenyl-1,3,8-triazaspiro[4.5]decan-4-one (Ro 64-6198). ACS Chem Neurosci 6:1956–1964
Chang SD, Mascarella SW, Spangler SM et al (2015b) Quantitative signaling and structure-activity analyses demonstrate functional selectivity at the nociceptin/orphanin FQ Ooioid receptor. Mol Pharmacol 88:502–511
Charlton SJ, Vauquelin G (2010) Elusive equilibrium: the challenge of interpreting receptor pharmacology using calcium assays. Br J Pharmacol 161:1250–1265
Christoph T, Kögel B, Strassburger W et al (2007) Tramadol has a better potency ratio relative to morphine in neuropathic than in nociceptive pain models. Drugs R D 8:51–57
Christoph A, Eerdekens MH, Kok M et al (2017) Cebranopadol, a novel first-in-class analgesic drug candidate: first experience in patients with chronic low back pain in a randomized clinical trial. Pain 158:1813–1824
Christoph T, Raffa R, De Vry J et al (2018) Synergistic interaction between the agonism of cebranopadol at nociceptin/orphanin FQ and classical opioid receptors in the rat spinal nerve ligation model. Pharmacol Res Perspect. https://doi.org/10.1002/prp2.444
Chung S, Pohl S, Zeng J et al (2006) Endogenous orphanin FQ/nociceptin is involved in the development of morphine tolerance. J Pharmacol Exp Ther 318:262–267
Ciccocioppo R, Angeletti S, Sanna PP et al (2000) Effect of nociceptin/orphanin FQ on the rewarding properties of morphine. Eur J Pharmacol 404:153–159
Comer SD, Ashworth JB, Sullivan MA et al (2009) Relationship between rate of infusion and reinforcing strength of oxycodone in humans. J Opioid Manag 5:203–212
Courteix C, Coudoré-Civiale MA, Privat AM et al (2004) Evidence for an exclusive antinociceptive effect of nociceptin/orphanin FQ, an endogenous ligand for the ORL1 receptor, in two animal models of neuropathic pain. Pain 110:236–245
Cremeans CM, Gruley E, Kyle DJ et al (2012) Roles of μ-opioid receptors and nociceptin/orphanin FQ peptide receptors in buprenorphine-induced physiological responses in primates. J Pharmacol Exp Ther 343:72–81
de Guglielmo G, Matzeu A, Kononoff J et al (2017) Cebranopadol blocks the escalation of cocaine intake and conditioned reinstatement of cocaine seeking in rats. J Pharmacol Exp Ther 362:378–384
Dahan A, Yassen A, Bijl H et al (2005) Comparison of the respiratory effects of intravenous buprenorphine and fentanyl in humans and rats. Br J Anaesth 94:825–834
Dahan A, Boom M, Sarton E et al (2017) Respiratory effects of the nociceptin/orphanin FQ peptide and opioid receptor agonist, cebranopadol, in healthy human volunteers. Anesthesiology 126:697–707
Ding H, Czoty PW, Kiguchi N et al (2016) A novel orvinol analog, BU08028, as a safe opioid analgesic without abuse liability in primates. Proc Natl Acad Sci 113:E5511–E5518
Ding H, Kiguchi N, Yasuda D et al (2018) A bifunctional nociceptin and mu opioid receptor agonist is analgesic without opioid side effects in nonhuman primates. Sci Transl Med 10:eaar3483
Eerdekens M, Koch ED, Kok M et al (2016) Cebranopadol, a novel first-in-class analgesic: efficacy, safety, tolerability in patients with pain due to diabetic peripheral neuropathy. Postgrad Med 128(Suppl 2):25
Eerdekens MH, Kapanadze S, Koch ED et al (2018) Cancer related chronic pain: investigation of the novel analgesic drug candidate cebranopadol in a randomized, double blind, noninferiority trial. Eur J Pain. https://doi.org/10.1002/ejp.1331
Fantinati A, Bianco S, Guerrini R et al (2017) A diastereoselective synthesis of cebranopadol, a novel analgesic showing NOP/mu mixed agonism. Sci Rep 7:2416
Göhler K, Sokolowska M, Schoedel K et al (2018) Assessment of the abuse potential of cebranopadol in non-dependent recreational opioid users: a phase 1 randomized controlled study. J Clin Psychopharmacol. https://doi.org/10.1097/JCP.0000000000000995
Higgins GA, Grottick AJ, Ballard TM et al (2001) Influence of the selective ORL1 receptor agonist, Ro64-6198, on rodent neurological function. Neuropharmacology 41:97–107
Hu E, Calò G, Guerrini R et al (2010) Long-lasting antinociceptive spinal effects in primates of the novel nociceptin/orphanin FQ receptor agonist UFP-112. Pain 148:107–113
Journigan VB, Polgar WE, Khroyan TV et al (2014) Designing bifunctional NOP receptor-mu opioid receptor ligands from NOP-receptor selective scaffolds. Part II. Bioorg Med Chem 22:2508–2516
Khroyan TV, Polgar WE, Cami-Kobeci G et al (2011) The first universal opioid ligand, (2S)-2-[(5R,6R,7R,14S)-N-cyclopropylmethyl-4,5-epoxy-6,14-ethano-3-hydroxy-6-methoxymorphinan-7-yl]-3,3-dimethylpentan-2-ol (BU08028): characterization of the in vitro profile and in vivo behavioral effects in mouse models of acute pain and cocaine-induced reward. J Pharmacol Exp Ther 336:952–961
Khroyan TV, Cippitelli A, Toll N et al (2017) In vitro and in vivo profile of PPL-101 and PPL-103: mixed opioid partial agonist analgesics with low abuse potential. Front Psych 8:52
Kleideiter E, Piana C, Wang S et al (2018) Clinical pharmacokinetic characteristics of cebranopadol, a novel first-in-class analgesic. Clin Pharmacokinet 57:31–50. Erratum in: Clin Pharmacokinet 57:1057–1058
Ko MC, Naughton NN (2009) Antinociceptive effects of nociceptin/orphanin FQ administered intrathecally in monkeys. J Pain 10:509–516
Kotlińska J, Suder P, Legowska A et al (2000) OrphaninFQ/nociceptin inhibits morphine withdrawal. Life Sci 66:PL119–PL123
Kotlinska J, Wichmann J, Rafalski P et al (2003) Non-peptidergic OP4 receptor agonist inhibits morphine antinociception but does not influence morphine dependence. Neuroreport 14:601–604
Lambert DG, Bird MF, Rowbotham DJ (2015) Cebranopadol: a first in-class example of a nociceptin/orphanin FQ receptor and opioid receptor agonist. Br J Anaesth 114:364–366
Linz K, Christoph T, Tzschentke TM et al (2014) Cebranopadol: a novel potent analgesic nociceptin/orphanin FQ peptide and opioid receptor agonist. J Pharmacol Exp Ther 349:535–548
Linz K, Schröder W, Frosch S et al (2017) Opioid-type respiratory depressant side effects of cebranopadol in rats are limited by its nociceptin/orphanin FQ peptide receptor agonist activity. Anesthesiology 126:708–715
Lutfy K, Hossain SM, Khaliq I et al (2001) Orphanin FQ/nociceptin attenuates the development of morphine tolerance in rats. Br J Pharmacol 134:529–534
Malfacini D, Ambrosio C, Gro’ MC et al (2015) Pharmacological profile of nociceptin/orphanin FQ receptors interacting with G-proteins and β-arrestins 2. PLoS One 10:e0132865
Meert TF, Vermeirsch HA (2005) A preclinical comparison between different opioids: antinociceptive versus adverse effects. Pharmacol Biochem Behav 80:309–326
Micheli L, Lucarini E, Corti F et al (2018) Involvement of the N/OFQ-NOP system in rat morphine antinociceptive tolerance: are astrocytes the crossroad? Eur J Pharmacol 823:79–86
Murphy NP, Lee Y, Maidment NT (1999) Orphanin FQ/nociceptin blocks acquisition of morphine place preference. Brain Res 832:168–170
Piana C, Wang S, Bursi R (2016) A novel model-based methodology for the evaluation of abuse potential. https://www.page-meeting.org/pdf_assets/2662-Poster_PAGE_2016_final.pdf. Accessed 30 Aug 2018
Podlesnik CA, Ko MC, Winger G et al (2011) The effects of nociceptin/orphanin FQ receptor agonist Ro 64-6198 and diazepam on antinociception and remifentanil self-administration in rhesus monkeys. Psychopharmacology 213:53–60
Raehal KM, Walker JK, Bohn LM (2005) Morphine side effects in beta-arrestin 2 knockout mice. J Pharmacol Exp Ther 314:1195–1201
Raffa RB, Burdge G, Gambrah J et al (2017) Cebranopadol: novel dual opioid/NOP receptor agonist analgesic. J Clin Pharm Ther 42:8–17
Reiss D, Wichmann J, Tekeshima H et al (2008) Effects of nociceptin/orphanin FQ receptor (NOP) agonist, Ro64-6198, on reactivity to acute pain in mice: comparison to morphine. Eur J Pharmacol 579:141–148
Rizzi A, Malfacini D, Cerlesi MC et al (2014) In vitro and in vivo pharmacological characterization of nociceptin/orphanin FQ tetrabranched derivatives. Br J Pharmacol 171:4138–4153
Rizzi A, Cerlesi MC, Ruzza C et al (2016) Pharmacological characterization of cebranopadol a novel analgesic acting as mixed nociceptin/orphanin FQ and opioid receptor agonist. Pharmacol Res Perspect 4:e00247
Rizzi A, Ruzza C, Bianco S et al (2017) Antinociceptive action of NOP and opioid receptor agonists in the mouse orofacial formalin test. Peptides 94:71–77
Rutten K, De Vry J, Bruckmann W et al (2010) Effects of the NOP receptor agonist Ro65-6570 on the acquisition of opiate- and psychostimulant-induced conditioned place preference in rats. Eur J Pharmacol 645:119–126
Rutten K, De Vry J, Bruckmann W et al (2011) Pharmacological blockade or genetic knockout of the NOP receptor potentiates the rewarding effect of morphine in rats. Drug Alcohol Depend 114:253–256
Rutten K, Schröder W, Christoph T et al (2018) Selectivity profiling of NOP, MOP, DOP and KOP receptor antagonists in the rat spinal nerve ligation model of mononeuropathic pain. Eur J Pharmacol 827:41–48
Ruzza C, Rizzi A, Malfacini D et al (2014) Pharmacological characterization of tachykinin tetrabranched derivatives. Br J Pharmacol 171:4125–4137
Ruzza C, Holanda VA, Gavioli EC et al (2018) NOP agonist action of cebranopadol counteracts its liability to promote physical dependence. Peptides 112:101–105
Sałat K, Jakubowska A, Kulig K (2015) Cebranopadol: a first-in-class potent analgesic agent with agonistic activity at nociceptin/orphanin FQ and opioid receptors. Expert Opin Investig Drugs 24:837–844
Salat K, Furgala A, Salat R (2018) Evaluation of cebranopadol, a dually acting nociceptin/orphanin FQ and opioid receptor agonist in mouse models of acute, tonic, and chemotherapy-induced neuropathic pain. Inflammopharmacology 26:361–374
Schiene K, De Vry J, Tzschentke TM (2011) Antinociceptive and antihyperalgesic effects of tapentadol in animal models of inflammatory pain. J Pharmacol Exp Ther 339:537–544
Schiene K, Schröder W, Linz K et al (2018a) Inhibition of experimental visceral pain in rodents by cebranopadol. Behav Pharmacol. https://doi.org/10.1097/FBP.0000000000000420
Schiene K, Schröder W, Linz K et al (2018b) Nociceptin/orphanin FQ opioid peptide (NOP) receptor and micro-opioid peptide (MOP) receptors both contribute to the anti-hypersensitive effect of cebranopadol in a rat model of arthritic pain. Eur J Pharmacol 832:90–95
Scholz A, Bothmer J, Kok M et al (2018) Cebranopadol: a novel, first-in-class, strong analgesic: results from a randomized phase IIa clinical trial in postoperative acute pain. Pain Physician 21:E193–E206
Schröder W, Lambert DG, Ko MC et al (2014) Functional plasticity of the N/OFQ-NOP receptor system determines analgesic properties of NOP receptor agonists. Br J Pharmacol 171:3777–3800
Schunk S, Linz K, Hinze C et al (2014) Discovery of a potent analgesic NOP and opioid receptor agonist: cebranopadol. ACS Med Chem Lett 5:857–862
Shen Q, Deng Y, Ciccocioppo R et al (2017) Cebranopadol, a mixed opioid agonist, reduces cocaine self-administration through nociceptin opioid and mu opioid receptors. Front Psych 8:234
Spagnolo B, Calo G, Polgar WE et al (2008) Activities of mixed NOP and mu-opioid receptor ligands. Br J Pharmacol 153:609–619
Sukhtankar DD, Lagorio CH, Ko MC (2014) Effects of the NOP agonist SCH221510 on producing and attenuating reinforcing effects as measured by drug self-administration in rats. Eur J Pharmacol 745:182–189
Tian JH, Xu W, Fang Y et al (1997) Bidirectional modulatory effect of orphanin FQ on morphine-induced analgesia: antagonism in brain and potentiation in spinal cord of the rat. Br J Pharmacol 120:676–680
Toll L (2013) The use of bifunctional NOP/mu and NOP receptor selective compounds for the treatment of pain, drug abuse, and psychiatric disorders. Curr Pharm Des 19:7451–7460
Toll L, Khroyan TV, Polgar WE et al (2009) Comparison of the antinociceptive and antirewarding profiles of novel bifunctional nociception receptor/mu-opioid receptor ligands: implications for therapeutic applications. J Pharmacol Exp Ther 331:954–964
Toll L, Bruchas MR, Calo’ G et al (2016) Nociceptin/orphanin FQ receptor structure, signaling, ligands, functions, and interactions with opioid systems. Pharmacol Rev 68:419–457
Tzschentke TM, Rutten K (2018) Mu-opioid peptide (MOP) and nociceptin/orphanin FQ peptide (NOP) receptor activation both contribute to the discriminative stimulus properties of cebranopadol in the rat. Neuropharmacology 129:100–108
Tzschentke TM, De Vry J, Terlinden R et al (2006) Tapentadol HCl: analgesic, μ opioid receptor (MOR) agonist, noradrenaline reuptake inhibitor. Drugs Future 31:1053–1061
Tzschentke TM, Christoph T, Kögel B et al (2007) (−)-(1R,2R)-3-(3-dimethylamino-1-ethyl-2-methyl-propyl)-phenol hydrochloride (tapentadol HCl): a novel mu-opioid receptor agonist/norepinephrine reuptake inhibitor with broad-spectrum analgesic properties. J Pharmacol Exp Ther 323:265–276
Tzschentke TM, Jahnel U, Kögel B et al (2009) Tapentadol hydrochloride: a next-generation, centrally acting analgesic with two mechanisms of action in a single molecule. Drugs Today 45:483–496
Tzschentke TM, Kögel BY, Frosch S et al (2017a) Limited potential of cebranopadol to produce opioid-type physical dependence in rodents. Addict Biol. https://doi.org/10.1111/adb.12550
Tzschentke TM, Linz K, Frosch S et al (2017b) Antihyperalgesic, antiallodynic, and antinociceptive effects of cebranopadol, a novel potent nociceptin/orphanin FQ and opioid receptor agonist, after peripheral and central administration in rodent models of neuropathic pain. Pain Pract 17:1032–1041
Ueda H, Yamaguchi T, Tokuyama S et al (1997) Partial loss of tolerance liability to morphine analgesia in mice lacking the nociceptin receptor gene. Neurosci Lett 237:136–138
Ueda H, Inoue M, Takeshima H et al (2000) Enhanced spinal nociceptin receptor expression develops morphine tolerance and dependence. J Neurosci 20:7640–7647
Walentiny DM, Wiebelhaus JM, Beardsley PM (2018) Nociceptin/orphanin FQ receptors modulate the discriminative stimulus effects of oxycodone in C57BL/6 mice. Drug Alcohol Depend 187:335–342
Winger G, Hursh SR, Casey KL et al (2002) Relative reinforcing strength of three N-methyl-D-aspartate antagonists with different onsets of action. J Pharmacol Exp Ther 301:690–697
Winter L, Nadeson R, Tucker AP et al (2003) Antinociceptive properties of neurosteroids: a comparison of alphadolone and alphaxalone in potentiation of opioid antinociception. Anesth Analg 97:798–805
Yassen A, Olofsen E, Romberg R et al (2007) Mechanism-based PK/PD modeling of the respiratory depressant effect of buprenorphine and fentanyl in healthy volunteers. Clin Pharmacol Ther 81:50–58
Zaveri NT (2011) The nociceptin/orphanin FQ receptor (NOP) as a target for drug abuse medications. Curr Top Med Chem 11:1151–1156
Zaveri NT, Jiang F, Olsen C et al (2013) Designing bifunctional NOP receptor-mu opioid receptor ligands from NOP receptor-selective scaffolds. Part I. Bioorg Med Chem Lett 23:3308–3313
Zaveri NT, Journigan VB, Polgar WE (2015) Discovery of the first small-molecule opioid pan antagonist with nanomolar affinity at mu, delta, kappa, and nociception opioid receptors. ACS Chem Neurosci 6:646–657
Acknowledgments
Thanks are given to Stefanie Frosch and Marielle Eerdekens for careful revision of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Tzschentke, T.M., Linz, K., Koch, T., Christoph, T. (2019). Cebranopadol: A Novel First-in-Class Potent Analgesic Acting via NOP and Opioid Receptors. In: Ko, MC., Caló, G. (eds) The Nociceptin/Orphanin FQ Peptide Receptor. Handbook of Experimental Pharmacology, vol 254. Springer, Cham. https://doi.org/10.1007/164_2019_206
Download citation
DOI: https://doi.org/10.1007/164_2019_206
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-20185-2
Online ISBN: 978-3-030-20186-9
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)