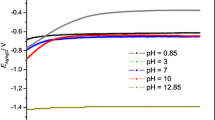
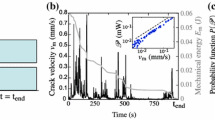
Conclusion
Corrosion, especially pitting corrosion, is a typical heterogeneous reaction composed of several processes. Usually, it is reduced to each elemental phenomenon, such as breakdown of passive film and substrate dissolution, which are treated separately to establish the theoretical and experimental bases of corrosion.
However, such a corrosion process forms a typical complex system, in which the reaction proceeds in a complicated fashion, remaining linked to each phenomenon in the process. Therefore, the nonequilibrium fluctuations discussed here have great significance since by using these fluctuations, the whole system can be described as it exists, without any reduction into elements.
Moreover, fluctuations of this kind are important, not only because they provide a useful method for describing such a complex system, but also because they actually exist in the reaction process. Thus it can be said that the corrosion reaction progresses according to the formation of nonequilibrium fluctuations. The most important point is that there is complete reciprocity between reactions and fluctuations; a reaction is controlled by the fluctuations, while the fluctuations are controlled by the reaction itself. Therefore, we can again point out that the reactivity in corrosion is determined, not by its distance from the reaction equilibrium, but by the growth process of the nonequilibrium fluctuations.
The author thanks Dr. Norio Sato, Professor Emeritus of Hokkaido University for stimulating, fruitful discussion, and also thanks Professor Asanuma at Gunma Polytechnic College and Dr. Shinohara at NRLM-JST for providing many data for this paper.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
R. Houbertz, U. Memmert, and R. J. Behm, Appl. Phys. Lett. 58 (1991) 1027.
S. Manne, P. K. Hansma, J. Massie, V. B. Elings, and A. A. Gewirth. Science 251 (1991) 183.
H. E. Stanley, Introduction of Phase Transitions and Critical Phenomena, Clarendon Press, Oxford, 1971.
P. Drazin and W. Reid, Hydrodynamic Stability, Cambridge University Press, London, 1981.
D. P. Woodruff, The Solid-Liquid Interface, Cambridge University Press, London, 1973.
N. Sato, K. Kudo, and T. Noda, Z. Phys. Chem. N. F. 98 (1975) 271.
N. Sato, J. Electrochem. Soc. 129 (1982) 260.
N. Sato, Electrochemistry, Chapter 3, Nittetsu Gijutsu Jyoho Center, Japan, 1993.
N. Sato, K. Kudo and R. Nishimura, J. Electrochem. Soc. 123 (1976) 1419.
R. Nishimura and N. Sato, J. Jpn. Inst. Metals 47 (1983) 1086.
C. L. Foley, J. Kruger, and C. J. Bechtold, J. Electrochem. Soc. 114 (1967) 994.
M. Sakashita and N. Sato, Corrosion 35 (1979) 351.
N. Sato, Corros. Sci. 27 (1987) 421.
M. Sakashita and N. Sato, Corros. Sci. 17 (1977) 473.
N. Sato, Corrosion 45 (1989) 354.
R. Nishimura and N. Sato, J. Jpn. Inst. Metals 47 (1983) 1086.
R. Nishimura and N. Sato, in Proc. 9th International Congress on Metallic Corrosion, Vol. 1, p. 96, National Research Council Canada, Toronto, Canada, 1984.
G. L. Griffin, J. Electrochem. Soc. 131 (1984) 18.
J. R. Galvele, in Passivity of Metals, R. P. Frankenthal and J. Kruger, eds., p. 285, Electrochemical Society, Pennington, NJ, 1978.
N. Sato, in Proc. of the First Soviet-Japanese Seminar on Corrosion and Protection of Metals, Ya. M. Kolotyrkin, ed., p. 258, Nauka, Moscow, 1979.
T. Shibata and T. Takeyama, Nature 260 (1976) 315.
T. Shibata and T. Takeyama, Corrosion 33 (1977) 243.
N. Sato, J. Electrochem. Soc. 123 (1976) 1197.
K. J. Vetter and H. H. Strehblow, Ber. Bunsenges. Phys. Chem. 74 (1970) 1024.
L. F. Lin, C. Y. Chao, and D. D. Macdonald, J. Electrochem. Soc. 128 (1981) 1187.
L. F. Lin, C. Y. Chao, and D. D. Macdonald, J. Electrochem. Soc. 128 (1981) 1194.
M. U-Macdonald and D. D. Macdonald, J. Electrochem. Soc. 134 (1987) 41.
D. D. Macdonald, J. Electrochem. Soc. 139 (1992) 3434.
N. Sato, Electrochim. Acta 16 (1971) 1683.
U. F. Frank, Z. Naturforsch. Teil A 4 (1949) 378.
T. Noda, K. Kudo, and N. Sato, Boshoku Gijutsu 20 (1971) 525.
H. Kaeshe, Z. Phys. Chem. N. F. 34 (1962) 87.
Y. Hisamatsu, Passivity and Its Breakdown on Iron and Iron Base Alloys, R. W. Steahle and H. Okada, eds., p. 99, National Association of Corrosion Engineers, Houston, TX, 1976.
S. Tsujikawa, Boshoku Gijutsu 31 (1982) 488.
U. F. Frank and K. Weil, Z. Elektrochem. 56 (1952) 814.
F. Flade, Z Phys. Chem. 76 (1911) 513.
J. Wojtowicz, in Modern Aspects of Electrochemistry, J. O’M Bockris and B. E. Conway, eds., No. 8, p. 51, Plenum Press, New York, 1972.
J. F. Cooper, R. H. Muller, and C. W. Tobias, J. Electrochem. Soc. 127 (1980) 1733.
L. Meunier, C. R. II Réunion du CITCE 1951, 242.
L Meunier, C. R. III Réunion du CITCE 1952, 247.
L. Meunier and G. Germain, C. R. III Réunion du CITCE 1952, 263.
R. S. Cooper and J. M. Bartlett, J. Electrochem. Soc. 105 (1958) 109.
A. J. Pearlstein, H. P. Lee, and K. Nobe, J. Electrochem. Soc. 132 (1985) 2159.
M. R. Bassett and J. L. Hudson, J. Phys. Chem. 92 (1988) 6963.
M. R. Bassett and J. L. Hudson, J. Electrochem. Soc. 137 (1990) 1815.
W. Li, X. Wang, and K. Nobe, J. Electrochem. Soc. 137 (1990) 1184.
W. Li, K. Nobe, and A. J. Pearlstein, J. Electrochem. Soc. 140 (1993) 721.
W. Li and K. Nobe, J. Electrochem. Soc. 140 (1993) 1642.
K. F. Bonhoeffcr and H. Gerischer, Z. Electrochem. 52 (1948) 149.
U. F. Frank and R. FitzHugh, Z. Elektrochem. 65 (1961) 156.
J. B. Talbot and R. A. Oriani, Electrochim. Acta 30 (1985) 1277.
J. B. Talbot, R. A. Oriani and M. J. DiCarlo, J. Electrochem. Soc. 132 (1985) 1545.
S. Chandrasekhar, Hydrodynamic and Hydmmagnetic Stability, Oxford University Press, London, 1961.
P. Glansdorff and I. Prigogine, Thermodynamic Theory of Structure, Stability and Fluctuations, Wiley-Interscience, New York, 1971.
I. Prigogine, From Being to Becoming, W. H. Freeman, San Francisco, 1980.
I. Prigogine and I. Stcngers, Order Out of Chaos, Bantam Books, New York, 1984.
W. W. Mullins and R. F. Sekerka, J. Appl. Phys. 34 (1963) 323.
R. F. Sekerka, J. Crystal Growth 3,4 (1968) 71.
R. T. Delves, J. Crystal Growth 3,4 (1968) 562.
D. T. J. Hurle, J. Crystal Growth 5 (1969) 162.
H. S. Chen and K. A. Jackson, J. Crystal Growth 8 (1971) 184.
R. F. Sekerka, 7. Crystal Growth 10 (1971) 239.
R. Aogaki, K. Kitazawa, Y. Rose and K. Fueki, Electrochim. Acta 25 (1980) 965.
R. Aogaki and T. Makino, Electrochim. Acta 26 (1981) 1509.
R. Aogaki, J. Electrochem. Soc. 129 (1982) 2442.
R. Aogaki, J. Electrochem. Soc. 129 (1982) 2447.
U. Landau, EPRI Report EM-2393, Electric Power Research Institute, Research Reports Center, Palo Alto, CA, 1982.
R. Aogaki and T. Makino, J. Electrochem. Soc. 131 (1984) 40.
R. Aogaki and T. Makino, J. Electrochem. Soc. 131 (1884) 46.
R. Aogaki and T. Makino, J. Chem. Phys. 81 (1984) 2154.
R. Aogaki and T. Makino, J. Chem. Phys. 81 (1984) 2164.
T. Makino and R. Aogaki, J. Chem. Phys. 81 (1984) 5137.
T. Makino and R. Aogaki, J. Chem. Phys. 81 (1984) 5145.
D. P. Barkey, R. H. Muller, and C. W. Tobias, J. Electrochem. Soc. 136 (1989) 2199.
D. P. Barkey, R. H. Muller, and C. W. Tobias, J. Electrochem. Soc. 136 (1989) 2207.
C.-P. Chen and J. Jorne, J. Electrochem. Soc. 138 (1991) 3305.
L.-G. Sundström and F. H. Bark, Electrochim. Acta 40 (1995) 599.
R. Aogaki, J. Electrochem. Soc. 142 (1995) 2954.
R. Aogaki, E. Yamamoto, and M. Asanuma, 7. Electrochem. Soc. 142 (1995) 2964.
R. Aogaki, J. Chem. Phys. 103 (1995) 8602.
R. Aogaki, A. Yamada, and A. Tadano, J. Chem. Phys. 103 (1995) 8616.
D. M. Mohilner, in Electroanalytical Chemistry, A. J. Bard, ed., Vol. 1, p. 292, Marcel Dekker, New York, 1966.
T. Okada, J. Electrochem. Soc. 132 (1985) 537.
N. Sato, Corrosion Science 37 (1995) 1947.
A. Tadano, M. Asanuma, and R. Aogaki, J. Crystal Growth 166 (1996) 1111.
A. Tadano and R. Aogaki, J. Chem. Phys. 106 (1997) 6126.
A. Tadano and R. Aogaki, J. Chem. Phys. 106 (1997) 6138.
A. Tadano and R. Aogaki, J. Chem. Phys. 106 (1997) 6146.
R. Aogaki and M. Asanuma, in Material Science Forum, Vols. 192–194, p. 101, Trans Tech Publications, Switzerland, 1995.
M. Asanuma and R. Aogaki, J. Chem. Phys. 106 (1997) 9930.
M. Asanuma and R. Aogaki, J. Chem. Phys. 106 (1997) 9938.
M. Asanuma and R. Aogaki, J. Chem. Phys. 106 (1997) 9944.
T. Imamura, Physics and Green Function, p. 52, Iwanami Syoten, Inc., Tokyo, 1978.
R. N. Bracewell, The Fourier Transform and Its Applications, p. 112, McGraw-Hill, Singapore, 1986.
B. MacDugall and M. J. Graham, J. Electrochem. Soc. 127 (1980) 789.
B. MacDugall and M. J. Graham, Electrochim. Acta 27 (1982) 1093.
U. Bertocci and F. Huet, Corrosion 51 (1995) 131.
R. Reigata, F. Sagués, and J. M. Costa, J. Chem. Phys. 101 (1994) 2329.
M. Asanuma and R. Aogaki, J. Electroanal. Chem. 396 (1995) 241.
R. Aogaki and M. Asanuma, Material Science Forum Vols. 289–292, p. 835, Trans Tech Publications, Zurich, Switzerland, 1998.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2002 Kluwer Academic Publishers
About this chapter
Cite this chapter
Aogaki, R. (2002). Nonequilibrium Fluctuations in the Corrosion Process. In: White, R.E., Bockris, J.O., Conway, B.E. (eds) Modern Aspects of Electrochemistry. Modern Aspects of Electrochemistry, vol 33. Springer, Boston, MA. https://doi.org/10.1007/0-306-46917-0_2
Download citation
DOI: https://doi.org/10.1007/0-306-46917-0_2
Publisher Name: Springer, Boston, MA
Print ISBN: 978-0-306-45968-9
Online ISBN: 978-0-306-46917-6
eBook Packages: Springer Book Archive