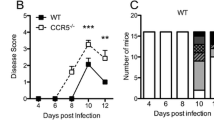
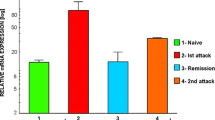
Abstract
Inflammation within the central nervous system (CNS) is critical in the development of the neuropathology associated with the human demyelinating disease multiple sclerosis (MS). Recent studies have identified a family of soluble proinflammatory molecules called chemokines that are able to direct leukocyte infiltration into the CNS in response to infection or injury. Identification of chemokines within and around demyelinating lesions in MS patients indicate a potential role for these molecules in contributing to the pathogenesis of MS. To address this issue, we have used mouse hepatitis virus (MHV) infection of the CNS to understand the dynamic interaction of chemokine expression as it relates to inflammation and neuropathology. Our results indicate that chemokine expression within the CNS results in persistent recruitment of both T lymphocytes and macrophages and results in subsequent myelin destruction. Herein, we demonstrate the complexity of the chemokine response to MHV infection of the CNS and the delicate balance that exists between host defense and development of disease.
Chapter PDF
Similar content being viewed by others
References
Lin MT, Hinton DR, Stohlman SA. Mouse hepatitis virus is cleared from the central nervous system in mice lacking perforin-mediated cytolysis. J Virol 1997 71:383–391.
Parra B, Hinton DR, Marten NW, Bergmann CC, Ln MT, Yang CS, Stohlman SA. IFN-gamma is required for viral clearance from central nervous system oligodendroglia. J Immunol 1999 162:1641–1647.
Wu GF, Dandekar AA, Pewe L, Perlman S. CD4 and CD8 T cells have redundant but not identical roles in virus-induced demyelination. J Immunol 2000 165:2278–2286.
Stohlman SA, Bergmann CC, Lin MT, Cua DJ, Hinton DR. CTL effector function within the central nervous system requires CD4+ T cells. J Immunol 1998 160:2896–2904.
Wang F, Stohlman SA, Fleming JO. Demyelination induced by murine hepatitis virus JHM (MHV-4) is immunologically mediated. J Neuroimmunol 1990 30:31–41.
Lane TE, Liu MT, Chen BP, Asensio VC, Samawi RM, Paoletti AD, Campbell IL, Kunkel SL, Fox HS, Buchmeier MJ. A central role for CD4+ T cells and RANTES in virus-induced central nervous inflammation and demyelination. J Virol 2000 74:1415–1424.
Lin MT, Hinton DR, Marten NW, Bergmann CC, Stohlman SA. Antibody prevents virus reactivation within the central nervous system. J Immunol 1999 162:7358–7368.
Wu GF, Perlman S. Macrophage infiltration, but not apoptosis, is correlated with immune-mediated demyelination following murine infection with a neurotropic coronavirus. J Virol 1999 73:8771–8780.
Miller SD, Vanderlugt CL, Begolka WS, Pao W, Yaunch RL, Neville KL, Katz-Levy Y, Carrizosa A, Kim BS. Persistent infection with Theiler’s virus leads to CNS autoimmunity via epitope spreading. Nat Med 1997 3:1133–1136.
Sun N, Grzybicki D, Castro R, Murphy S, Perlman S. Activation of astrocytes in the spinal cord of mice chronically infected with a neurotropic coronavirus. Virology 1995 213:482–493.
Luster AD. Chemokines-chemotactic cytokines that mediate inflammation. New Engl J Med 1998 338:436–445.
Clark-Lewis I, Kim K, Rajarathan K, Gong J, Dewald B, Moser B, Baggiolini M, Sykes B. Structure activity relationships of chemokines. J Leuk Biol 1995 57:703–711.
Taub DD, Oppenheim JJ. Chemokines, inflammation and the immune system. Ther Immunol 1994 1:229–246.
Cyster JG. Chemokines and cell migration in secondary lymphoid organs. Science 1999 286:2098–2102.
Cyster JG. Leukocyte migration: scent of the T zone. Cur Biol 2000 10:R30–33.
Huang D, Han Y, Rani MR, Glabinski A, Trebst C, Sorensen T, Tani M, Wang J, Chien P, O’Bryan S, Bielecki B, Zhou ZL, Majumder S, Ransohoff RM. Chemokines and chemokine receptors in inflammation of the nervous system: manifold role and exquisite regulation. Immunol Rev 2000 177:52–67.
Glabinski AR, Ransohoff RM. Chemokines and chemokine receptors in CNS pathology. J Neurovirol 1999 5:3–12.
Farber JM. Mig and IP-10: CXC chemokines that target lymphocytes. J Leukoc Biol 1997 61:246–257.
Piali L, Weber C, LaRosa G, Mackay CR, Springer TA, Clark-Lewis I, Moser B. The chemokine receptor CXCR3 mediates rapid and shear-resistant adhesion-induction of effector T lymphocytes by the chemokines IP-10 and Mig. Eur J Immunol 1998 28:961–972.
Liu MT, Chen BP, Oertel P, Buchmeier MJ, Armstrong D, Hamilton TA, Lane TE. Cutting Edge: the T cell chemoattractant IFN-inducible protein 10 is essential in host defense against viral-induced neurologic disease. J Immunol 2000 165:2327–2330.
Liu MT, Armstrong D, Hamilton TA, Lane TE. Expression of Mig (monokine induced by interferon-g) is important in T lymphocyte recruitment and host defense following viral infection of the central nervous system. J Immunol 2001 166:1790–1795.
Trifilo MJ, Bergmann CC, Kuziel WA, Lane TE. The CC chemokine ligand 3 (CCL3) regulates CD8+ T cell effector function and migration following viral infection. J Virol 2003 in press.
Chen BP, Kuziel WA, Lane TE. Lack of CCR2 results in increased mortality and impaired leukocte activation and trafficking following infection of the central nervous system with a neurotropic coronavirus. J Immunol 2001 167:4585–4592.
Glass WG, Liu MT, Kuziel WA, Lane TE. Reduced macrophage infiltration and demyelination in mice lacking the chemokine receptor CCR5 following infection with a neurotropic coronavirus. Virology 2001 288:8–17.
Murray PD, Krivacic K, Chernosky A, Wei T, Ransohoff RM, Rodriquez M. Biphasic and regionally-restricted chemokine expression in the central nervous system in the Theiler’s virus model of multiple sclerosis. J Neurovirol 2000 6:S44–52.
Ransohoff RM, Wei T, Pavelko KD, Lee JC, Murray PD, Rodriguez, M. Chemokine expression in the central nervous system of mice with a viral disease resembling multiple sclerosis: roles of CD4+ and CD8+ T cells and viral persistence. J Virol 2002 76:2217–2224.
Olszewski MA, Huffnagle GB, McDonald RA, Lindell DM, Moore BB, Cook DN, Toews GB. The role of macrophage inflammatory protein-1 alpha/CCL3 in regulation of T cell-mediated immunity to Cryptococcus neoformans infection. J Immunol 2000 165:6429–6436.
Huffnagle GB, Strieter RM, Standiford TJ, McDonald RA, Burdick MD, Kunkel SL, Toews GB. The role of monocytes chemotactic protein-1 (MCP-1) in the recruitment of monocytes and CD4+ T cells during a pulmonary Cryptococcus neoformans infection. J Immunol 1995 155:4790–4997.
Tanaka Y, Adams DH, Hubscher S, Hirano H, Siebenlist U, Shaw S. T-cell adhesion induced by proteoglcan-imobilized cytokine MIP-1 beta. Nature 1993 361:79–82.
Karpus WJ. Chemokines and central nervous system disorders. J Neurovirol 2001 7:493–500.
Liu MT, Keirstead HS, Lane TE. Neutralization of the chemokine CXCL10 reduces inflammatory cell invasion and demyelination and improves neurological function in a viral model of multiple sclerosis. J Immunol 2001 167:4091–4097.
Furie MB, Randolph GJ. Chemokines and tissue injury. Am J Pathol 1995 146:1287–1301.
Karpus WJ, Lukacs NW, Kennedy KJ, Smith WS, Hurst SD, Barrett TA. Differential CC chemokine-induced enhancement of T helper cell cycle cytokine production. J Immunol 1997 158:4129–4136.
Murphy P. The molecular biology of leukocyte chemoattractant receptors. Ann Rev Immunol 1994 12:593–633.
Kelvin D, Michiel D, Johnston J, Lloyd A, Sprenger H, Oppenheim J, Wang JM. Chemokines and serpentines: the molecular biology of chemokine receptors. J Leukocyte Biol 1993 54:604–612.
Horuk R. Molecular properties of the chemokine receptor family. Trends Pharmacol Sci 1994 15:159–165.
Horuk R. The interleukin-8-receptor family: from chemokines to malaria. Immunol Today 1994 15:169–174.
Ward SG, Bacon K, Westwick J. Chemokines and T lymphocytes: more than an attraction. Immunity 1998 9:1–11.
Horuk R, Chitnis CE, Darbonne WC, Colby TJ, Rybicki A, Hadley TJ, Miller H. A receptor for the malarial parasite Plasmodium vivax: the erythrocyte chemokine receptor. Science 1993 261:1182–1184.
Chaudhuri A, Polyakove J, Zbrzezna V, Williams K, Gulati S, Pogo A. Cloning of glycoprotein D cDNA, which encodes the major subunit of the Duffy blood group system and the receptor for Plasmodium vivax malaria parasite. Proc Nat Acad Sci USA 1993 90:10793–10797.
Chaudhuri A, Zbrzezna V, Polyakova J, Pogo A, Hesselgesser J, Horuk R. Expression of the Duffy antigen in K562 cells: evidence that it is the human chemokine erythrocyte receptor. J Biol Chem 1994 269:7835–7838.
Neote K, Darbonne W, Ogez J, Horuk R, Schall T. Identification of a promiscuous inflammatory peptide receptor on the surface of red blood cells. J Biol Chem 1993 268:12247–12249.
Horuk R, Colby T, Darbonne W, Schall T, Neote K. The human erythrocyte inflammatory peptide (chemokine) receptor. Biochemical characterization, solubilization, and development of a binding assay for the soluble receptor. Biochemistry 1993 32:5733–5738.
Ben-Baruch A, Michiel DF, Oppenheim JJ. Signals and receptors involved in recruitment of inflammatory cells. J Biol Chem 1995 270:11703–11706.
Sallusto F, Mackay CR, Lanzavecchia A. The role of chemokine receptors in primary, effector, and memory immune responses. Annu Rev Immunol 2000 18:593–620.
Hamann A, Klugewitz K, Austrup F, Jablonski-Westrich D. Activation induces rapid and profound alterations in the trafficking of T cells. Eur J Immunol 2000 30:3207–3218.
Potsch C, Vohringer D, Pircher H. Distinct migration patterns of naïve and effector CD8 T cells in the spleen: correlation with CCR7 receptor expression and chemokine reactivity. Eur J Immunol 1999 29:3562–3570.
Hvas J, Bernard CC. Molecular detection and quantitation of the chemokine RANTES mRNA in neurological brain. APMIS 1998 106:598–604.
Sorensen TL, Tani M, Jensen J, Pierce V, Lucchinetti C, Folcik VA, Qin S, Rottman J, Sellebjerg F, Strieter RM, Frederiksen JL, Ransohoff RM. Expression of specific chemokines and chemokine receptors in the central nervous system of multiple sclerosis patients. J Clin Invest 1999 103:807–815.
Baranzini SE, Elfstrom C, Chang SY, Butunoi C, Murray R, Higuchi R, Oksenberg JR. Transcriptional analysis of multiple sclerosis brain lesions reveals a complex pattern of cytokine expression. J Immunol 2000 165:6576–6582.
Lane TE, Asensio VC, Yu N, Paoletti AD, Campbell IL, Buchmeier MJ. Dynamic regulation of alpha and beta chemokine expression in the central nervous system during mouse hepatitis virus-induced demyelinating disease. J Immunol 1998 160:970–978.
Balashov KE, Rottman JB, Weiner HL, Hancock WW. CCR5(+) and CCR3(+) T cells are increased in multiple sclerosis and their ligands MIP-1-alpha and IP-10 are expressed in demyelinating brain lesions. Proc Natl Acad Sci USA 1999 96:6873–6878.
Glass WG, Lane TE. Functional expression of chemokine receptor CCR5 on CD4(+) T cells during virus-induced central nervous system disease. J Virol 2003 77: 191–198.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2005 Springer Science+Business Media, Inc.
About this chapter
Cite this chapter
Trifilo, M.J., Liu, M.T., Glass, W.G., Lane, T.E. (2005). Chemokines in Coronavirus-Induced Demyelination. In: Lavi, E., Constantinescu, C.S. (eds) Experimental Models of Multiple Sclerosis. Springer, Boston, MA. https://doi.org/10.1007/0-387-25518-4_45
Download citation
DOI: https://doi.org/10.1007/0-387-25518-4_45
Publisher Name: Springer, Boston, MA
Print ISBN: 978-0-387-25517-0
Online ISBN: 978-0-387-25518-7
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)